{ DOWNLOAD AS PDF }
ABOUT AUHTOR
HardikRana*, BhargavVaghasiya, MansiDholakia
Department of Pharmaceutics,
Anand Pharmacy College, Anand, 388 001, Gujarat, India.
*hardikrana1439@gmail.com
ABSTRACT
The objective of present study was to enhance the solubility of poorly soluble drug Ibuprofen using spherical crystallization technique.The potential agglomerates were prepared by addition of different concentrationof polymer selected on the basis of Phase solubility study. Spherical agglomerates were prepared using diethyl ether as bridging liquid by neutralizing technique, spherical agglomeration technique, Quasi emulsion solvent diffusion technique. Spherical agglomerates were evaluated for morphology, production yield, drug content, particle size and dissolution behaviour compared with pure drug.The result of phase solubility studies revealed that there is enhancement of solubility by PEG 4000. Rod shaped crystals of pure drug converted to spherical was confirmed by optical microscopy. The dissolution of agglomerates of optimum batch exhibited 88.24% release compare to 47.18% of pure drug within 60 minute. This study demonstrated that spherical crystallization technique can be consider as a suitable alteration of granulation. Ibuprofen spherical agglomerates can be prepared with PEG 4000. It exhibited excellent physicochemical, solubility, dissolution rate in comparison with pure drug. Among other spherical crystallization technique, QESD proved to be excellent techniquefor enhancement of solubility and dissolution.
[adsense:336x280:8701650588]
REFERENCE ID: PHARMATUTOR-ART-2471
|
PharmaTutor (ISSN: 2347 - 7881) Volume 5, Issue 3 Received On: 20/08/2016; Accepted On: 23/11/2016; Published On: 01/03/2017 How to cite this article: Rana H, Vaghasiya B, Dholakia M;Solubility Enhancement of poorly Water Soluble Drug by Spherical Crystallization Technique; PharmaTutor; 2017; 5(3); 31-36 |
INTRODUCTION:
Aqueous solubility and dissolution are two of the crucial factors influencing drug absorption from the gastrointestinal tract (GIT). However, many newly discovered compounds exhibit low oral bioavailability. More than 40% of active substances during formulation development by the pharmaceutical industry are poorly water soluble.[1] Poor water solubility, which is associated with poor dissolution characteristics. As a result, much research has been conducted into methods of improving drug solubility and dissolution rates to increase the oral bioavailability of hydrophobic drugs. One way of improving dissolution involves the reduction of particle size and/or increasing saturation solubility. One of the most common approaches used to reduce particle size is milling, a mechanical micronization process. Milling is a well-established technique which is relatively cheap, fast and easy to scale-up. However, milling has several disadvantages, the main one being the limited opportunity to control important characteristics of the final particle such as size, shape, morphology, surface properties and electrostatic charge. In addition, milling is a high energy process which causes disruptions in the drug’s crystal lattice, resulting in the presence of disordered or amorphous regions in the final product. These amorphous regions are thermodynamically unstable and are therefore susceptible to recrystallization upon storage, particularly in hot and humid conditions.[2]
The alteration of the surface properties also changes the milled product’s saturation solubility as well as blending and flow properties, which in turn, have an impact on the formulation process. Furthermore, milled particles often show aggregation and agglomeration which results in poor wettability and thus poor dissolution.[3]An alternative to milling involves growing the particle from a solution to the desired size range under controlled conditions, for example by spray drying, emulsion solvent-diffusion[4] and supercritical fluid technology.[5, 6] One of the advantages of these methods is the possibility of designing in certain beneficial characteristics such as enhancing dissolution rate by incorporating different polymers. The spherical crystallization technique has been successfully applied now days to improve the micromeritic properties of drug substances. [7] In the most common case, this technique is reputed to improve the wettability and dissolution rate of different drugs. [8, 9, 10, 11] Some drugs have also been recrystallized by the spherical agglomeration technique using polymeric materials to modify their release. [12, 13]
There are two main methods for spherical crystallization: spherical agglomeration (SA method) and emulsion solvent diffusion (ESD method). [14] In the SA method, a quasi-saturated solution of the drug, in a solvent in which it is very soluble, is poured into a poor solvent of the drug. Provided that the good and the poor solvents are freely miscible and interaction (binding force) between the solvents is stronger than drug interaction with the good solvent, crystals precipitate immediately. A suitable amount of a third solvent, which is not miscible with the poor solvent and which preferentially wets the precipitated crystals, is added to the system while stirring. This third solvent, which is called a ‘bridging liquid’, can collect the crystals suspended in the system by forming liquid bridges between the crystals due to capillary negative pressure and interfacial tension between the interface of solid and liquid. When interaction between the drug and the good solvent is stronger than that of the good and poor solvents, the good solvent drug solution is dispersed in the poor solvent, producing quasi emulsion droplets, even if the solvents are normally miscible. This is due to an increase in the interfacial tension between good and poor solvent. Then the good solvent diffuses gradually out of the emulsion droplet into the outer poor solvent phase. The counter diffusion of the poor solvent into the droplet induces the crystallization of the drug within the droplet due to the decreasing solubility of the drug in the droplet containing the poor solvent. This process is known as the emulsion solvent diffusion (ESD) process.[15]
Ibuprofen is a non-steroidal anti-inflammatory drug which is widely used as a pain killer. It has a low solubility in water and gastric fluids, which determines a low dissolution rate and hence inter individual variability on its bioavailability. [16]The aim of the present investigation is to enhance solubility hence dissolution rate by Spherical crystallization technique.
MATERIALS AND METHODS
2.1. Materials:
Ibuprofen was supplied as a gift sample by Baroque Pharmaceuticals Pvt. Ltd, Khambhat. PEG 4000 was purchased from S.D. fine chemicals, Baroda. Deionized double-distilled water was used throughout the study.
2.2.Preparation of Ibuprofen agglomerates
The polymer according to specific concentration was dissolved along with 250 mg of drug (Ibuprofen) in the mixture of good solvent (ethanol= 3 ml) and bridging liquid (Dimethyl formamide= 1 ml) in the different drug: polymer mass ratio a shown in Table 1. The solution was poured into 100 ml distilled water (poor solvent) with an optimized stirring rate (800 rpm) using propeller type of agitator (Remi Motors Ltd., Mumbai, India) at room temperature. After agitating the system for 30 min, the prepared agglomerates were collected by filtration through Whatman filter paper no. 1.The spherical crystals were washed with distilled water and placed at room temperature for drying for 24 h and stored in desiccators.[17]
Table 1: Composition of Solid Dispersion
|
Ingredients |
F-1 |
F-2 |
F-3 |
|---|---|---|---|
|
Ibuprofen (mg) |
250 |
250 |
250 |
|
Ethanol (ml) |
3 |
3 |
3 |
|
DMF (ml) |
1 |
1 |
1 |
|
PEG 4000 (mg) |
22 |
36.6 |
62 |
|
Water (ml) |
100 |
100 |
100 |
|
Stirring Speed (rpm) |
800 |
800 |
800 |
2.3. Particle size determination
Particle size was determined using the optical microscopy. Optical microscopy was used to observe the crystal habit (size and shape) in this study. Optical microscopy was the simplest instrument to capture images of crystal habits, especially for wet samples or samples in solution. Optical microscopy (OM) can be divided into eye-piece, objective lens, object holder and stage.[18, 19]
2.4. Drug content
Drug content was determined by dissolving 100mg equivalent Ibuprofen powder in 100ml methanol and if necessary making further dilution using methanol. Amount of drug dissolved was analyzedspectrophotometrically (Shimadzu 1650PC) at wavelength of 221nmand drug content find out using calibration equation. All studies were carried out in triplicates.[20]
2.5.Saturation Solubility study
Excess amounts of Ibuprofen agglomerates and pure TBM drug powder was dispersed in 20mL distilled water. The dispersion was shaken at 100 rpm at 370C for 24 hrs. usingshaker thermostatic cabinet (Remi, RI’S-24BL, Mumbai).After 24 hours the dispersion was filtered through a whatman filter paper (grade 1, circles, 90 mm diameter), followed by dilution. The filtered sample solutions were analyzed using a UV–visible spectrophotometer (shimadzu 1650PC) at 221 nm. The mean results of triplicate measurements and the standard deviation were reported.[21]
The Gibbs-free energy of change (ΔG˚tr°) of tolbutamide occurred during formation of agglomerates from untreated TBM powder was calculated using equation (1).
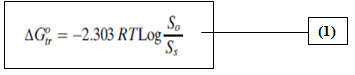
Where S0/Ss, is the ratio of the molar solubility of Ibuprofen agglomerates in distilled water to that of the untreated TBM powder. R is gas constant, its value is 8.31 J K-1 mol-1 and T is temperature in Kelvin.
2.6. In vitro Dissolution study
The dissolution study was carried out by using a USP apparatus II (Electrolab, TDT-08L) using 900 ml of phosphate buffer pH 7.4 at a temperature of 37±10C at 75 rpm. At regular time intervals (5 min), suitable amount of sample was withdrawn and same amount replaced by fresh medium. Samples were suitably diluted and filtered through syringe filter (Axiva SFCA25X, 0.45µm). Drug amount released was obtained spectrophotometrically (shimadzu 1650PC) at wavelength of 221nm. All studies were carried out in triplicates.[21]
RESULT and DISCUSSION
3.1. Selection of Solvents
The choice of the best solvent was done based on the available literature on solubility of tolbutamide and miscibility of the solvents.[22]Ethanol, N, N-Dimethyl formamide (DMF) and water were selected as a good solvent, bridging liquid and poor solvent, respectively. Selection of bridging liquid should be such that it should be immiscible with the poor solvent i.e. water and the drug should have slight solubility in it. [23] Agglomerates were formed by agitating the crystals in a liquid suspension and adding bridging liquid which preferentially wets the crystal surface to cause binding. The addition of bridging liquid (DMF) promotes the formation of liquid bridges between the drug crystals to form spherical agglomerates. The spherically agglomerated crystals are formed by coalescence of these dispersed crystals.
3.2. Effect of the Drug: Polymer Ratio
The agglomerated crystals prepared by incorporating water-soluble polymers can improve solubility and in vitro drug release. Addition of PEG water-soluble polymer increased the solubility of Ibuprofen in water as well as in the dissolution medium.
Table 2: Results of different Evaluation parameter
|
Formulation |
Drug: Polymer mass ratio |
Stirring Speed in rpm |
Mean particle size (µm) a |
Percentage yield (%) |
Drug content (%) |
Appearance of the agglomerated crystals |
|---|---|---|---|---|---|---|
|
PEG 4000 |
1: 0.03 |
800 |
92.84 |
88.6± 0.91 |
98.18± 0.45 |
Less agglomeration |
|
|
1: 0.05 |
800 |
113.89 |
90.13± 0.42 |
98.64± 0.91 |
Large agglomerates |
|
|
1: 0.08 |
800 |
153.45 |
94± 0.8 |
98.64± 0.91 |
Large and more number of agglomerates |
|
Pure TBM |
- |
- |
95.6 |
- |
100.30± 0.26 |
Rod shaped crystals |
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT editor-in-chief@pharmatutor.org
Subscribe to Pharmatutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE
3.3. Particle Size, Percentage yield and Drug content
Untreated Ibuprofen powder showed higher 95.6 µm particle size and rod shaped crystal habit. The mean particle size, percentage yield and drug content of Ibuprofen agglomerates were shown in the table . The size of prepared agglomerates was found between 92.84 µm to 153.45 µm which is significantly different from that of pure drug. The presence of PEG in spherical agglomerates influenced the particle size of resultant agglomerates as shown in table. As the concentration of the PEG increased, the size of the agglomerates increased. The yield of the agglomerates was found satisfactory and ranged from 88 to 94 % while drug content was in the range 98 to 99%.
3.4. Saturation Solubility Study
The results of solubility studies indicate that the pure Ibuprofen possesses a very low solubility in water (0.026±0.0005 mg/mL, n=3). The drug solubility from the spherical crystals increased significantly demonstrating that the incorporation of hydrophilic polymers PEG 4000 which enhances the drug solubility by improving wettability. The use of the polymer PEG in agglomerate formation increases the solubility. This may be due to the improved porosity, decreased particle size and partial amorphization of drug in agglomerates. The solubility also increases due to adsorption of PEG on to the drug particle surface during agglomeration. Addition of polyethylene glycol water-soluble polymer increases the solubility of TBM in water.
Table 3: Average Solubility of Ibuprofen and its agglomerates
|
Polymer |
Drug: Polymer ratio |
Average Solubility (mg/ml) ± SD |
Gibbs free energy change (ΔG˚tr) (J K-1Mol-1) |
|---|---|---|---|
|
PEG 4000 |
1: 0.03 |
0.322±0.0021# |
-6071.27 |
|
|
1: 0.05 |
0.353±0.0035# |
-6308.22 |
|
|
1: 0.08 |
0.492±0.0015# |
-7164.08 |
|
Pure Ibuprofen |
- |
0.026±0.0005 |
4.28 |
All the values are expressed as mean± STD, n=3.
The ΔG˚tr values provide the information about whether the reaction condition is favourable or unfavourable for drug solubilisation in the aqueous carrier solution. Negative Gibbs free energy values indicate favourable conditions. Thus there was a significant improvement in solubility.
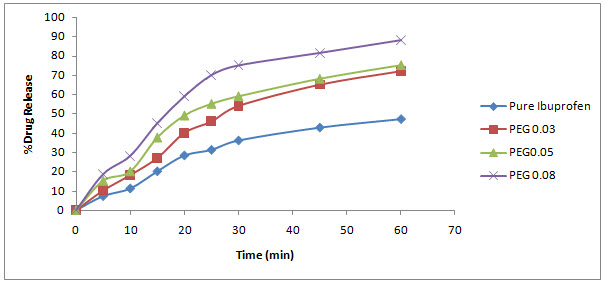
3.5. In vitro dissolution studies
The dissolution profiles of pure ibuprofen and its agglomerates are shown in figure. Dissolution rate was influenced by crystal habit of drug powder. The amount of drug dissolved depends on the spherical crystal faces exposed to the dissolution medium. Spherical agglomerates of different polymers gave higher dissolution compared to pure Ibuprofen powder. In dissolution study, PEG agglomerates showed 88.24%% cumulative drug release in 60 min compared with Ibuprofen (47.18%). The reason for this faster dissolution was linked to the better wettability of the spherically agglomerated crystals. Incorporating the water-soluble polymer like PEG into the agglomerates of poorly soluble crystals improved the dissolution rate followed by, incorporating the hydrophilic bursting polymer chitosan which showed an improved dissolution rate as compared with pure drug. The increase of the dissolution rates of the agglomerates is also attributed to the much better uniformity, the decreased crystallinity, and the reduction of particle.
CONCLUSION
In this study, the attempt was made to develop spherical crystals of poorly water soluble drug Ibuprofen by using emulsion solvent diffusion (ESD) technique of Spherical crystallization. The present study showed that spherically agglomerated crystals of Ibuprofen with PEG prepared by ESD technique exhibited an improvement in the solubility and dissolution rate as compared with pure Ibuprofen. Spherical crystals prepared with ESD technique produce agglomerates with smaller mean particle size, highest crystal yield and higher aqueous solubility. This method of spherical crystallization changes the crystal habit from rod type (Untreated Ibuprofen) to spherical crystals which were clearly seen in microscopy. The in vitro drug release from agglomerates was found to be 2 times higher than that of pure drug. The enhanced dissolution rates and aqueous solubility attributed to the reduction of the particle size, change in crystal habit, formation of hydrophilic surface and the increased wettability due to adsorption of stabilizing agents, and partial amorphization of Ibuprofen during spherical crystallization. The manufacturing of the spherical crystals by emulsion solvent diffusion technique requires simple and common equipment only and is able to perform in a single process step. The aforementioned technique is a promising tool for effective agglomerate formation during pharmaceutical development in order to achieve appropriate dissolution behaviour of poorly water soluble active ingredient. This technique may be applicable for producing oral solid dosage forms of poorly soluble drugs with improved dissolution rate and solubility.
REFERENCES
[1]Martinez M, Augsburger L, Johnston T, Jones W;Applying the biopharmaceutics classification system to veterinary pharmaceutical products. Part I. Biopharmaceutics and formulation considerations; Adv Drug Deliv Rev.; 2002; 54; 805–824.
[2] Ward G.H., Schultz, R.K.; Process induced crystallinity changes in Albuterolsulfate and its effect on powder physical stability;Pharm. Res.;1995; 12; 773–779.
[3]Tur K.M., Chang H.S., Baie, S.; Use of bioadhesive polymer to improve the bioavailability of griseofulvi;. Int. J. Pharm.; 1997;148; 63–71.
[4] Quintanar-Guerrero D., Allemann E., Fessi, H., Doelker E.; Preparation techniques and mechanisms of formation of biodegradable nanoparticles from preformed polymers;Drug Dev. Ind. Pharm,;1998; 24; 1113–1128.
[5] Hu J., Johnston K.P., Williams R.O.; Spray freezing into liquid (SFL) particle engineering technology to enhance dissolution of poorly soluble drugs: organic solvent versus organic/aqueous co-solvent systems;Eur. J. Pharm. Sci.; 2003; 20; 295–303.
[6] Moneghini M., Kikic I., Voinovich D., Perissutti B., Alessi P., Cortesi A., Princivalle F., Solinas D.; Study of the solid state of carbamazepine after processing with gas anti-solvent technique;Eur. J. Pharm. Biopharm.; 2003; 56; 281–289.
[7] Kawashima Y., Cui F., Takeuchi H., Niwa T., Hino T.,Kiuchi K.; Improvements in flowability and compressibility of pharmaceutical crystals for direct tabletting by spherical crystallization with a two-solvent system;Powder Technol;1994b; 78; 151–157.
[8] Kawashima Y., Handa T., Takeuchi H., Okumura M.,Katou H., Nagata O.; Crystal modification of phenytoin with polyethylene glycol for improving mechanical strength, dissolution rate and bioavailability by a spherical crystallization technique;Chem. Pharm. Bull;, 1986; 34; 3376–3383.
[9] Sano A., Kuriki T., Kawashima Y., Takeuchi H., HinoT., Niwa T.; Particle design of tolbutamide by spherical crystallization technique. V. Improvement of dissolution and bioavailability of direct compressed tablets prepared using tolbutamide agglomerated crystals;Chem. Pharm. Bull;, 1992; 40; 3030–3035.
[10] Guillaume F.,Guyot-Hermann A.M., Guyot J.C.; Spherical crystallization of meprobamate II; Farmaco.; 1993; 48; 473–485.
[11] Di Martino P., Barthelemy C., Piva F., Joiris E., Palmieri G.F., Martelli S.; Improved dissolution behavior of fenbufen by spherical crystallization;Drug Dev. Ind. Pharm.; 1999; 25; 1073–1081.
[12] Akbuga J.; Preparation and evaluation of controlled release furosemide microspheres by spherical crystallization;Int. J. Pharm.; 1989; 53; 99–105.
[13] Ribardiere A., Tchoreloff P., Couarraze G., Puisieux F.; Modification of ketoprofen bead structure produced by the spherical crystallization technique with a two-solvent system;Int. J. Pharm.;1996; 144; 195–207.
[14] Kawashima Y.; Part 1. New processes.Application of spherical crystallization to particulate design of pharmaceuticals for direct tabletting and coating and new drug delivery system. In: Chulia, D., Deleuil, M., Pourcelot, Y. (Eds.);Powder Technology and Pharmaceutical Process; Elsevier; 1994a; 493–512.
[15]Morishima K et al ;Micromeritic characteristics and agglomeration mechanisms in the spherical crystallization of bucillamine by the spherical agglomeration and emulsion solvent diffusion method; Powder Technology; 1993; 76 (1); 57-64.
[16] Miralles, M.J., McGinity, J.W. and Martin, A.; Combined water-soluble carriers for coprecipitates of tolbutamide;J. Pharm. Sci.;1982; 71; 302-304.
[17] Kawashima Y et al; Parameters determining the agglomeration behavior and the micromeritic properties of spherically agglomerated crystals prepared by the spherical crystallization technique with miscible solvent system; international journal of Pharmaceutics; 1995; 119(2); 139-147.
[18] Paradkar A. R., Pawar A.P. et al; Spherical crystallization of Celecoxib; Drug Development and Industrial Pharmacy; 2002; 28(10).
[19] Martino P et al; Improved dissolution behavior of Fenbufen by spherical crystallization Drug Development and Industrial Pharmacy; 1999; 25(10).
[20] Kachrimanis K et al; Spherical crystal agglomeration of ibuprofen by the solvent change technique in the presence of methacrylic polymers; Journal of Pharmaceutical Sciences; 2000;89 (2); 250-259.
[21] Jbilou M. et al; Ibuprofen agglomerates preparation by phase separation; Drug Development and Industrial Pharmacy; 1999; 25(3).
[22] Keraliya R, Soni T, Thakkar V, Gandhi T.; Effect of solvent on Crystal Habit and Dissolution behaviour of Tolbutamide by Initial Solvent Screening;Dissolution Technologies; 2010; 16-21.
[23] Chourasia M.K., Jain A., Jain N., Vaidya S, Jain S.K.; Utilization of spherical crystallization for preparation of directly compressible materials; Indian Drugs;2004; 41; 319–329.
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT editor-in-chief@pharmatutor.org
Subscribe to Pharmatutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE









