About Authors:
CHIRAG S. RAJPARA*1, JAWED AKHTAR1, AMIT KHANDHAR2
1. Department of Quality Assurance, School of Pharmaceutical Sciences, Jaipur National University, Jagatpura, Jaipur-302025, Rajasthan, India.
2. Zydus cadila pharma, moraiya,ahmedabad.
*chiragrajpara2601@gmail.com
ABSTRACT
* A simple, specific, accurate and stability-indicating reversed phase high performance liquid chromatographic method was developed for simultaneous estimation of Tadalafil and Dapoxetine HCL, Water Symmetry C-18 (150 x 4.6 mm),5 µ and a mobile phase composed of Buffer : Acetonitrile (65:35)
* The retention time of Tadalafil and Dapoxetine HCL were found to be10.08 and 4.45 min respectively. Linearity was established for Tadalafil and Dapoxetine HCL in the range of 8.0-60 μg/ml and 24.0-180.0 μg/ml respectively. The percentage recovery of Tadalafil and Dapoxetine HCL were found to be in the range of 99.7-100.6% and 98.07-99.09% respectively. The drug was subjected to acid and alkali hydrolysis, oxidation, dry heat and photolytic degradation. The degradation studies indicated, Tadalafil showed degradation in acid and alkali while it was found stable in H2O2, photolytic and in presence of dry heat and Dapoxetine showed degradation in thermal and peroxide while it was found stable in rest of parameters . The degradation products of Tadalafil and Dapoxetine HCL in acidic, alkaline conditions were well resolved from the pure drug with significant differences in their retention time values. This method can be successfully employed for the quantitative analysis of Tadalafil and Dapoxetine HCL in bulk drugs and formulations.
[adsense:336x280:8701650588]
Reference Id: PHARMATUTOR-ART-1387
1. INTRODUCTION
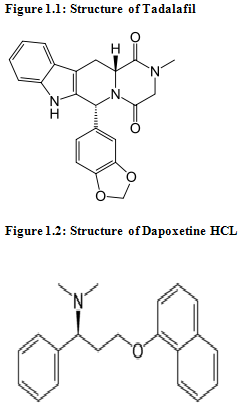
Tadalafil (6R-trans)-6-(1,3-benzodioxol-5-yl)- 2,3,6,7,12,12a-hexahydro- 2-methy pyrazino [1', 2':1,6] pyrido[3,4-b]indole - 1,4-dione(fig. 1.1), is a white powder, freely soluble in acetonitrile. It is A long acting PDE5 Inhibitor for the management of erectile dysfunction. It is indicated for the treatment of erectile dysfunction in men.[10, 11, 12].
Dapoxetine S)-N,N-dimethyl-3-(naphthalen-1- yloxy)-1-phenylpropan-1-amine(fig. 1.2), is white crystalline powder, Freely soluble in water and ethanol and Acetonitrile. It is Short acting serotonine reuptake inhibitor. It indicated for the treatment of premature ejaculation in men 18 to 64 years of age.[13,14]
Literature survey indicated that determination of Tadalafil and Dapoxetine HCL were done by RP-HPLC method with UV detection for routine analysis of Dapoxetine HCl in a pharmaceutical formulation. [8]The literature survey does not reveal any stability indicating assay method for simultaneous estimation of Tadalafil and Dapoxetine HCL in bulk and Pharmaceutical dosage form which gives information about the degradation products as well as separation of degradation products. The literature survey indicated that no stability indicating RP-HPLC method was proposed for Tadalafil and Dapoxetine HCL .
The International Conference on Harmonization (ICH) guideline entitled “Stability testing of new drug substances and products” requires that stress testing be carried out to elucidate the inherent stability characteristics of the active substance [4]. An ideal stability-indicating method is one that resolves the drug and its degradation products efficiently. Consequently, the implementation of an analytical methodology to determine Tadalafil and Dapoxetine HCL in presence of its degradation products is rather a challenge for pharmaceutical analyst. Therefore, it was thought necessary to study the stability of Tadalafil and Dapoxetine HCL under acidic, alkaline, oxidative, photolytic and dry heat conditions. This paper reports validated stability-indicating HPLC method for simultaneous determination of Tadalafil and Dapoxetine HCL in presence of their degradation products. The proposed method is simple, accurate, reproducible, stability-indicating and suitable for routine determination of Tadalafil and Dapoxetine HCL in its dosage form. The method was validated in compliance with ICH guidelines. [5, 6]
[adsense:468x15:2204050025]
2. MATERIALS AND METHODS:
Tadalafil and Dapoxetine HCL of working standard grade was kindly supplied as gift sample by Zydus Cadila Healthcare Ltd., Ahmedabad, India and was certified to contain 99.30% (w/w) and 99.40%(w/w) respectively, on as is basis. Tadalafil and Dapoxetine HCL Active Pharmaceutical Ingredient (API) was kindly supplied as gift sample by MSN Laboratories, Hyderabad, India and was certified to contain 99.30% (w/w) and 99.40%(w/w) respectively, on as is basis. Acetonitrile and Methanol used were of HPLC grade and were purchased from Spectrochem Pvt. Ltd. Mumbai, India. The tablet formulation containing 20 mg of Tadalafil and 60 mg of Dapoxetine HCL were procured from local market and used for analysis of marketed formulation. After several trials taken by changing mobile phase, columns, flow rate, column temperature, the following chromatographic conditions were finalised.
The HPLC system (Shimadzu Corporation, Japan), model Shimadzu VP, consisted of a system controller (CLASS-VP), on-line degasser (LC 2010C, Shimadzu), low pressure gradient valve (LC 2010C, Shimadzu), solvent delivery module (LC 2010C, Shimadzu), auto injector (LC 2010C, Shimadzu), column oven (LC 2010C, Shimadzu), and CLASS – VP software version = SPI, binary pump, auto injector (SIL-10AD VP, Shimadzu), column oven (CTO-10AS VP, Shimadzu) and PDA detector (PDA-SPD-M10A VP, Shimadzu Diode Array Detector) and Chem station (software).
The wavelength of detection chosen was 285 nm for both the drug. Water Symmetry C-18 (150 x 4.6 mm),5 µ was used for the analysis. The mobile phase comprising of a mixture of-Butyl ammonium hydrogen sulphate and NaH2PO4 with organic mixture in ratio of 65: 35 % v/v. The buffer pH adjusted to 2.5 with H3PO4 .The acetonitrile was used as a organic mixture. The flow rate of mobile phase is 1.0 ml/min. The injection volume was 10 μl.
In addition, an electronic balance (Mettler Toledo), a pH meter (Metrohm), a sonicator (Flexit jour lab pvt. Ltd., model UCB 45), a hot air oven (Labhosp) was used in this study.
2.1 Preparation of Mobile Phase:
Dissolve 0.7 gm of Tetra-Butyl ammonium hydrogen sulphate and 3.7 gm NaH2PO4 in 2000 ml Milli-Q water and then adjust ph 2.5 with H3PO4. Mix the buffer solution and acetonitrile in proportion of 65:35 and sonicate it properly and filter the mobile phase through 0.45 µ HVLP milli-pore filter.
2.2 Preparation of Standard Solution:
The standard stock solution was prepared by transferring 40 mg of Tadalafil (A) and 60 mg of Dapoxetine (B) in a 100 ml volumetric flask separately. Then pipette out 5ml (A) and 10ml (B) in to 50ml volumetric flask and make up the volume up to 50mlwith diluents which is having final concentration 40 µg/ml and 12040 µg/ml. The HPLC analysis was performed on reversed-phase high-performance liquid chromatographic system with isocratic elution mode using a mobile phase of Buffer: organic mixture (65:35 %v/v), on Water Symmetry C-18 (150 x 4.6 mm),5 µ with 1.0 ml/min flow rate at 285 nm using UV detector.
2.2 Analysis of Marketed Formulations:
Take tablets in to mortar pestle and crused it to make a fine powder and then take a powder drug equivalent to 2.5 tablets (50 mg tadalafil and 150 mg of dapoxetine) and transfer both drugs in to 250 ml of volumetric flask separately and make up the volume up to 100 ml with diluent and sonicated them for 20 min. then make up the volume up to 250 ml with diluent. Then pipette out 5 ml of both drugs and transfer in to 25ml of volumetric flask separately and make up the volume up to 25 ml with diluent. These give final concentration 40 μg /ml and 120 μg /ml of tadalafil and dapoxetine respectively. The solution was further filtered using 0.45μ PVDF filter.
2.4 Method Validation:
The method of analysis was validated as per the recommendations of ICH [7] and USP [3] for the parameters like accuracy, linearity, precision, specificity, detection limit, quantitation limit ruggedness, robustness and solution stability.
2.4.1 Linearity:
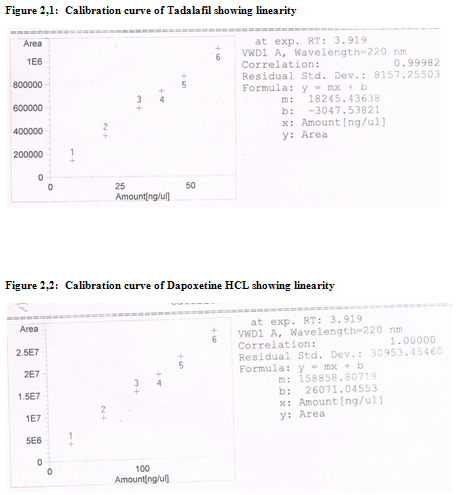
Every 10 μl of the working standard solution of Tadalafil and Dapoxetine HCL in the mass concentration range of 8.0-60.0 μg/ml and 24.0-180 μg/ml respectively, were injected into the chromatographic system. The chromatograms were developed and the peak area was determined for each concentration of the drug solution. Calibration curve of Silodosin was obtained by plotting the peak area ratio versus the applied concentrations of Tadalafil and Dapoxetine HCL (fig. 2.1 & 2.2).
2.4.2 Accuracy:
The accuracy of the method was determined by calculating percentage recovery of Tadalafil and Dapoxetine HCL . Recovery studies were carried out by applying the spiking method in which known amount of Tadalafil and Dapoxetine corresponding to 50, 100 and 150% of label claim had been added (standard addition method) placebo. At each level of the amount three determinations were performed and the results obtained were compared.
2.4.3 Precision:
The method precision was done by preparing six different sample preparations by one analyst under the same conditions and analysing them.
2.4.4 LOD and LOQ:
The limit of detection (LOD) and limit of quantitation (LOQ) were calculated using following formulae: LOD= 3.3(SD)/S and LOQ= 10 (SD)/S, where SD=standard deviation of response (peak area) and S= average of the slope of the calibration curve.
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT articles@pharmatutor.org
Subscribe to PharmaTutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE
2.4.5 System Suitability:
System suitability tests are an integral part of chromatographic method which are used to verify reproducibility of the chromatographic system. To ascertain its effectiveness, certain system suitability test parameters were checked by repetitively injecting the drug solution at the concentration level 40 μg/ml of Tadalafil and 120 μg/ml of Dapoxetine HCL to check the reproducibility of the system.
2.4.6 Specificity:
To carryout specificity test injection of placebo solution was injected to check whether there is no carry over in blank solution as well as the placebo does not interferes with the drug peak. The drug peak was also checked for its peak purity in presence of excipients.
2.4.7 Robustness:
For robustness evaluation of HPLC method a few parameters like flow rate, column oven temperature, percentage of organic mixture in the mobile phase and pH of buffer were deliberately changed. One factor was changed at one time to estimate the effect. Each factor selected was changed at three levels (-1, 0, +1) with respect to optimized parameters. Robustness of the method was done at the concentration level of Tadalafil and 120 μg/ml of Dapoxetine HCL.
2.4.8 Ruggedness:
Ruggedness was determined between two different analysts, instruments and columns to see whether the method provides reproducible results with the changes.
2.4.9 Solution Stability:
The standard and sample solutions were injected at different time intervals for 36 hrs to observe % difference in chromatographic responses relative to freshly prepared solutions.
2.5Forced degradation studies:
Forced degradation studies of the drugs were carried out under conditions of acid-alkali hydrolysis, dry heat, oxidation and photolysis. Tadalafil and Dapoxetine were weighed (20.0 mg) and (60.0mg) respectively and transferred into 100 ml volumetric flasks. To this, pipette out 5ml of Tadalafil and 10ml of Dapoxetine HCL to 50 ml volumetric flask to get final conc. 40 μg/ml and 120 μg/ml respectively. For sample powdered tablets weighed equivalent to 50 mg of Tadalafil and 150mg of Dapoxetine were transferred to 250 ml volumetric flask and further diluted both by taking 5ml to 25ml volumetric flask . Three such sets were prepared for acid, alkali and peroxide degradation.
Ø Forced degradation in basic media was performed by adding 5 ml of 5 N NaOH to above prepared mixtures in one set of flasks and these mixtures were heated for up to 2 h at 80°C.Then the solution was cooled and neutralized with 5 ml 5N HCl.
Ø Forced degradation in acidic media was performed by adding 5 ml of 5 N HCl to above prepared mixtures in one set of flasks and these mixtures were heated for up to 2 h at 80°C. Then the solution was cooled and neutralized with 5 ml 5N NaOH.
Ø Forced degradation in peroxide was performed by adding 5 ml of 10% H2O2 to above prepared mixtures in one set of flasks and these mixtures were heated for up to 2 h at 80°C.
Ø For dry heat degradation, solid drugs (20+60 mg Std. and Tablet powder equivalent to 20+60 mg) were kept in Petri dish in oven at 100°C for 24 h.
Ø For photo stability, solid drugs (20+60) mg Std. and Tablet powder equivalent to 20+60 mg) were kept in Petri dish in direct sunlight in summer days for 5 h on a wooden plank.
For HPLC analysis, all the degraded sample solutions were diluted with diluent Water:ACN (50:50) to obtain final concentration of 40μg/ml of Tadalafil and 120μg/ml of Dapoxetine.
Besides, solutions containing 40μg/ml of Tadalafil and 120μg/ml of Dapoxetine separately were also prepared without being performing the degradation of both the drugs. Then 10 μl solution of above solutions were injected into HPLC system and analyzed under the chromatographic condition described earlier.
3. RESULTS AND DISCUSSION:
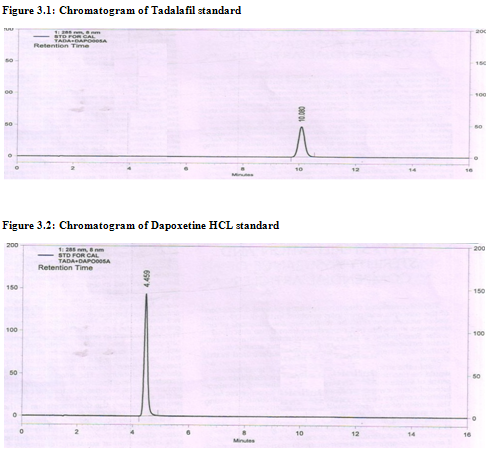
The mobile phase consisting of Buffer:ACN (65: 35, %v/v), 1.0ml/min flow rate was optimised which gave sharp peak with minimum tailing factor for Tadalafil and Dapoxetine (fig. 3.1 & 3.2). The retention time for Tadalafil and Dapoxetine were 10.08 and 4.4 min respectively. UV spectra of Tadalafil and Dapoxetine showed the λmax at common wavelength 285 nm, so this wavelength was selected as the detection wavelength.
Ø The calibration curve for Tadalafil and Dapoxetine were found to be linear over the range of 8.0-60.0 μg/ml and 24.0-180 μg/ml respectively. The graph gave a regression coefficient of 0.99982 and 1.0000 respectively. The data of regression analysis of the calibration curves is shown in Table 1.1 &1.2 respectively.
Ø The accuracy results show that the method is accurate to practically achieve added amount at three different levels within 98.08-101.9 and 97.09-101.09 respectively with RSD less than 2%. The average %recovery were found to be 99.08 and 99.09% respectively. The results are shown in Table 3.1 and 3.2.
Ø The method precision gave results obtained within 2% RSD suggesting the method is precise. The results are shown in Table 4.
Ø The LOD for Tadalafil and Dapoxetine were found to be 1.47 μg/ml and 0.643 while LOQ were 4.47 μg/ml and 1.948 μg/ml respectively.
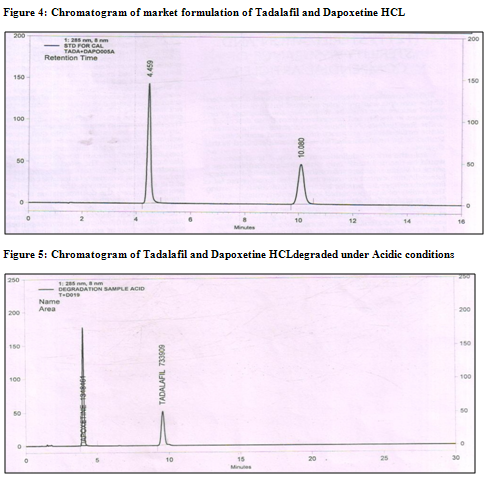
Ø The developed method was also found to be specific, since there was no interference of excipients and the purity of the peak was >0.999(fig. 4).
Ø The results for ruggedness evaluation give RSD below 2% suggest the method is rugged to changes. The results are shown along with method precision in Table 4.
Ø The results for system suitability parameters are shown in Table 5.
Ø Results for robustness evaluation for drug are presented in Table 6. Insignificant differences in peak areas and less variability in retention times were observed. The results mentioned are of area obtained.
Ø The results for solution stability are shown in Table 7. The standard and sample solutions were found to be stable up to 36 hrs.
Ø The proposed method was successfully applied to the determination of Silodosin in its dosage form. The results for the combination were comparable with the corresponding labelled amounts.
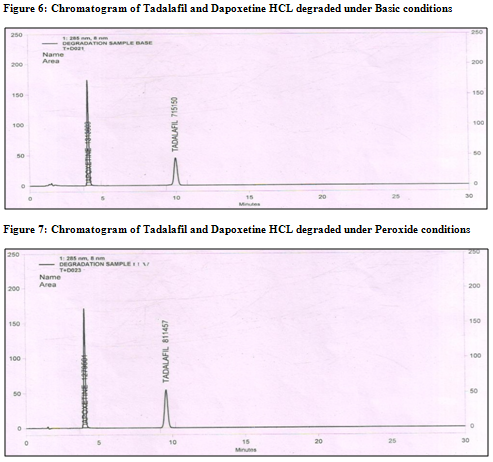
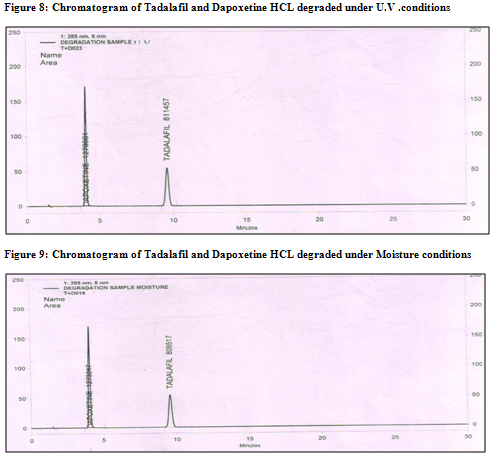
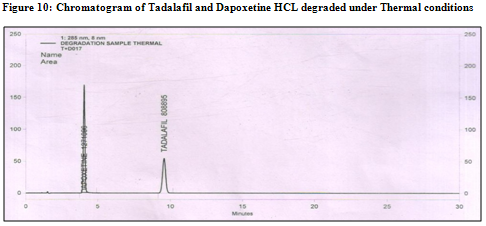
Ø The degradation study indicated that Tadalafil and Dapoxetine were susceptible to acid, base and peroxide while it was stable in direct sunlight and dry heat under experimental conditions (fig. 7.1,8.1,9.1 and fig7.2,8.2,9.2) respectively., while the drug showed no degradation at 0 h in all the degradation conditions.
4. CONCLUSION:
In the proposed study, stability-indicating HPLC method was developed for the simultaneous estimation of Tadalafil and Dapoxetine HCL and validated as per ICH guidelines. Statistical analysis proved that method was accurate, precise, and repeatable. The developed method was found to be simple, sensitive and selective for analysis of Silodosin without any interference from the excipients. The method was successfully used for determination of drugs in a pharmaceutical formulation. Assay results for combined dosage form using proposed method showed 99.3±0.40 % and 101.8±0.8%of Tadalafil and Dapoxetine HCL respectively. The results indicated the suitability of the method to study stability of Tadalafil and Dapoxetine HCL under various forced degradation conditions viz. acid, base, peroxide, dry heat and photolytic degradation. It can be concluded that the method separates the drugs from their degradation products; it may be employed for analysis of stability samples of Tadalafil and Dapoxetine HCL.
5. ACKNOWLEDGEMENTS:
The authors thank Zydus Cadila Healthcare Ltd., Ahmedabad and MSN Laboratories, Hyderabad, India for providing Silodosin as gift sample for this work. They also thank the management of Jaipur National University for providing required facilities to carry out this research work.
6. REFERENCES:
1. Ewing GW. Instrumental Methods of Chemical Analysis. 5th ed. New York: McGraw-Hill Book Company; 1985. p. 1-7.
2. Mendham J, Denney RC, Baraes JD, Thomas MJK. Vogel’s Textbook of Quantitative Chemical Analysis. 5th ed. Singapore: Pears Edjucation; 1991.p.5-11.
3. Swarbrick J, Boyal JC. Encyclopedia of Pharmaceutical Technology. Vol. I. New York: Marcel Dekker Inc.; 1998. p. 217-24.
4. Meyer VR. Practical High Performance Liquid Chromatography. 2nd ed. London: John Wiley and Sons; 1993. p. 26-7, 40, 222, 246 and 256.
5. Safarinejad MR (2007). "Safety and Efficacy of Dapoxetine in the Treatment of Premature Ejaculation: A Double-Blind, Placebo-Controlled, Fixed-Dose, Randomized Study". Neuropsychopharmacology 33 (6): 1259. doi:10.1038/sj.npp.1301500. PMID 17625501.
6. Ahuja S. Modern Pharmaceutical Analysis: An Overview. In: Ahuja S, Scypinski S, editors. Handbook of Modern Phamaceutical Analysis. Vol. III. United States of America: Academic Press; 2001. p. 349.
7. Mendham J, Denney RC, Baraes JD, Thomas MJK. Vogel’s Textbook of Quantitative Chemical Analysis. 5th ed. Singapore: Pears Edjucation; 1991. p. 217-35.
8. Szepei Gabor. HPLC in Pharmaceutical Analysis. Vol. I, 1990. p. 101-73.
9. Kendirci M, Salem E, Hellstrom WJ (June 2007). "Dapoxetine, a novel selective serotonin transport inhibitor for the treatment of premature ejaculation". Ther Clin Risk Manag 3 (2): 277–89. doi:10.2147/tcrm.2007.3.2.277. PMC 1936309. PMID 18360636.
TABLES:
Table1.1: Data of calibration curve for Tadalafil
|
Linearity Range |
Stock solution to be taken in ml |
Dilute to volume (ml)with diluent |
Final concentration in µg/ml Tadalafil |
Area |
|
20% |
1.0 |
50 |
8.001 |
141708.00 |
|
50% |
2.5 |
50 |
20.00 |
353242.00 |
|
80% |
4.0 |
50 |
32.00 |
588258.00 |
|
100% |
5.0 |
50 |
40.00 |
734313.00 |
|
120% |
6.0 |
50 |
48.00 |
861502.00 |
|
150% |
7.5 |
50 |
60.01 |
1094695.00 |
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT articles@pharmatutor.org
Subscribe to PharmaTutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE
Table1.2: Data of calibration curve for Dapoxetine HCL
|
Linearity Range |
Stock solution to be taken in ml |
Dilute to volume (ml)with diluent |
Final concentration in µg/ml Dapoxetine |
Area |
|
20% |
2.0 |
50 |
24.0 |
386707.00 |
|
50% |
5.0 |
50 |
60.0 |
954853.00 |
|
80% |
8.0 |
50 |
96.0 |
152914.0 |
|
100% |
10.0 |
50 |
120.0 |
190658.0 |
|
120% |
12.0 |
50 |
144.0 |
229436.0 |
|
150% |
15.0 |
50 |
180.0 |
285941.0 |
Table 2.1: Data for linearity of Tadalafil
|
Linearity Range |
Stock solution to be taken in ml |
Dilute to volume (ml)with diluent |
Final concentration in µg/ml Tadalafil |
Area |
|
20% |
1.0 |
50 |
8.001 |
141708.00 |
|
50% |
2.5 |
50 |
20.00 |
353242.00 |
|
80% |
4.0 |
50 |
32.00 |
588258.00 |
|
100% |
5.0 |
50 |
40.00 |
734313.00 |
|
120% |
6.0 |
50 |
48.00 |
861502.00 |
|
150% |
7.5 |
50 |
60.01 |
1094695.00 |
Table 2.2: Data for linearity of Dapoxetine HCL
|
Linearity Range |
Stock solution to be taken in ml |
Dilute to volume (ml)with diluent |
Final concentration in µg/ml Dapoxetine |
Area |
|
20% |
2.0 |
50 |
24.0 |
386707.00 |
|
50% |
5.0 |
50 |
60.0 |
954853.00 |
|
80% |
8.0 |
50 |
96.0 |
152914.0 |
|
100% |
10.0 |
50 |
120.0 |
190658.0 |
|
120% |
12.0 |
50 |
144.0 |
229436.0 |
|
150% |
15.0 |
50 |
180.0 |
285941.0 |
The calibration curve was plotted as Area vs. Concentration
Table 3.1: Results of Accuracy Data of Tadalafil
|
For Tadalafil |
|||||
|
Level |
Amount of Drug added (mg ) |
Amount of Drug recovered (mg) |
Recovery (%) |
Mean ± SEM (%) |
% RSD
|
|
50 % |
25.50 |
25.02 |
98.08 |
100.6 ± 0.3551 |
1.6 |
|
25.10 |
25.39 |
101.9 |
|||
|
25.30 |
25.36 |
101.0 |
|||
|
100 % |
50.40 |
50.02 |
99.09 |
99.7 ±0.2160 |
0.5 |
|
50.60 |
50.32 |
100.1 |
|||
|
50.81 |
50.01 |
99.01 |
|||
|
150 % |
75.80 |
75.16 |
99.9 |
100.4 ± 0.0849 |
0.4 |
|
75.60 |
75.51 |
100.6 |
|||
|
75.30 |
75.21 |
100.6 |
|||
Table 3.2: Results of Accuracy Data of Dapoxetine HCL
|
For Dapoxetine |
|||||
|
Level |
Amount of Drug added (mg) |
Amount of Drug recovered (mg) |
Recovery (%) |
Mean ± SEM (%) |
% RSD
|
|
50 % |
74.90 |
75.87 |
101.09 |
99.08 ± 0.5959 |
2.0 |
|
75.80 |
75.02 |
99.06 |
|||
|
75.60 |
73.55 |
97.09 |
|||
|
100 % |
150.10 |
148.07 |
99.02 |
98.07 ± 0.0849 |
0.5 |
|
150.80 |
147.78 |
98.06 |
|||
Table 4: Method precision of Tadalafil and Dapoxetine HCL
|
Sr. No. |
Parameters (n= 5) |
Tadalafil |
Dapoxetine |
|
1 |
Retention Time (min) |
4.46 |
10.08 |
|
2 |
Theoritical Plates |
8660.77 |
4335.33 |
|
3 |
Asymmetry |
0.99 |
1.07 |
|
4 |
Capacity Factor |
33.90 |
153.40 |
|
5 |
% RSD |
0.2 |
0.1 |
Table 5: System suitability parameter of Tadalafil and Dapoxetine HCL
|
Sr. No. |
Parameters (n= 5) |
Tadalafil |
Dapoxetine |
|
1 |
Retention Time (min) |
4.46 |
10.08 |
|
2 |
Theoritical Plates |
8660.77 |
4335.33 |
|
3 |
Asymmetry |
0.99 |
1.07 |
|
4 |
Capacity Factor |
33.90 |
153.40 |
|
5 |
% RSD |
0.2 |
0.1 |
Table 6: Robustness of Tadalafil and Dapoxetine HCL
|
Compound |
% RSD (n= 5) |
||
|
Normal Condition |
Changed Condition |
||
|
Temperature |
Normal |
(-5°C) |
(+5°C) |
|
Tadalafil |
0.8 |
0.7 |
0.7 |
|
Dapoxetine |
0.8 |
0.6 |
0.7 |
|
pH |
Normal |
(-0.2 unit) |
(+0.2 unit) |
|
Tadalafil |
0.8 |
1.0 |
0.1 |
|
Dapoxetine |
0.8 |
0.9 |
0.2 |
|
Flow Rate |
Normal |
(-10%) |
(+10%) |
|
Tadalafil |
0.8 |
0.1 |
0.2 |
|
Dapoxetine |
0.8 |
0.1 |
0.1 |
|
Mobile phase ratio |
Normal |
(-2%) |
(+2%) |
|
Tadalafil |
0.8 |
0.1 |
0.2 |
|
Dapoxetine |
0.8 |
0.1 |
0.1 |
Table 7.1: Solution Stability of Tadalafil and Dapoxetine HCL API
|
Time (hour) |
Area |
% Difference |
||
|
Tadalafil |
Dapoxetine |
Tadalafil |
Dapoxetine |
|
|
0 (Initial) |
12544881 |
18997230 |
== |
== |
|
4 |
12517727 |
18982958 |
0.5 |
-0.2 |
|
8 |
12565888 |
18985192 |
0.4 |
0.1 |
|
12 |
12564030 |
18996924 |
0.8 |
-0.1 |
|
16 |
12540320 |
18940989 |
0.4 |
-0.3 |
|
20 |
12559102 |
18922468 |
0.9 |
-0.4 |
|
24 |
12543807 |
18956786 |
0.9 |
-0.7 |
|
% Mean RSD |
0.55 |
-0.22 |
||
|
± SD |
0.5678 |
-0.2436 |
||
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT articles@pharmatutor.org
Subscribe to PharmaTutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE
Table 7.2:Solution Stability of Tadalafil and Dapoxetine HCL Sample
|
Time (hour) |
Area |
% Difference |
||
|
Tadalafil |
Dapoxetine |
Tadalafil |
Dapoxetine |
|
|
0 (Initial) |
12568238 |
17736559 |
== |
== |
|
4 |
12558705 |
17727597 |
0.7 |
0.2 |
|
8 |
12551218 |
17716739 |
0.9 |
0.3 |
|
12 |
12540626 |
17719009 |
1.0 |
0.1 |
|
16 |
12575018 |
17691385 |
0.7 |
-0.1 |
|
20 |
12580299 |
17694692 |
0.8 |
-0.5 |
|
24 |
12571286 |
17745569 |
1.6 |
0.3 |
|
% Mean RSD |
0.68 |
0.04 |
||
|
± SD |
0.6914 |
0.0487 |
||
Table 8: Summary of Validation Parameter of RP-HPLC for estimation of Tadalafil and Dapoxetine HCL
|
Parameter |
Acceptance Crieteria |
Tadalafil |
Dapoxetine |
|
Linearity Range Correlation Coefficient |
Correlation coefficient r2 > 0.999 or 0.995 |
8.0 – 60.0 µg/ml r2 = 0.99982 |
24-180 µg/ml r2 = 1.00000 |
|
LOD |
S/N > 2 or 3 |
1.47 µg/ml |
0.643 µg/ml |
|
LOQ |
S/N > 10 |
4.47 µg/ml |
1.948 µg/ml |
|
Precision |
RSD < 2% |
%RSD = 1.1 |
%RSD = 0.4 |
|
Intermediate Precision |
RSD < 2% |
%RSD = 0.8 |
% RSD =1.3 |
|
Accuracy |
Recovery 98- 102% (individual) |
% recovery= 98.08-101.9 |
% recovery = 97.09-101.09 |
|
Specificity |
1) No intereference from blank, placebo and other degradation products with the main peak. 2) The peak purity index > 0.999 |
No intereference. Peak purity 1)Test sample = 0.99997 2)Spiked sample = 0.99967 3) Acid degradation = 0.99995 4) Alkali degradation = 0.99574 5) UV degradation = 0.99995 6) Thermal degradation = 0.99993 7) Humidity degradation = 0.99991 8)Peroxide degradation = 0.99941 |
No intereference. Peak purity 1)Test sample = 0.99928 2) Spiked sample = 0.9998 3) Acid degradation = 0.99761 4) Alkali degradation = 0.99844 5) UV degradation = 0.99986 6) Thermal degradation = 0.99863 7) Humidity degradation = 0.99886 8) Peroxide degradation = 0.99817 |
|
Solution Stability |
> 12 hour |
Stable up to 24 hour %RSD = 0.55 |
Stable up to 24 hour %RSD = -0.22 |
|
Robustness |
RSD NMT 2% in modified condition |
Complies |
Complies |
Table 9: Summary of Degradation Studies for Tadalafil and Dapoxetine HCL
|
Degradation Condition |
Time (hrs.) |
% Degradation |
tR a(min) of degradation products |
Peak Purity of Silodosin |
|||
|
API |
Sample |
API |
Sample |
API |
Sample |
||
|
Acid, 5N HCl |
2 hr, 80°C |
27.8% |
31.6% |
6.087 & 9.445 |
6.805 & 9.983 |
0.9999 |
0.9999 |
|
Base, 5N NaOH |
2 hr, 80°C |
12% |
13.6% |
-----** |
------ ** |
0.9999 |
0.9999 |
|
Oxidative, 10% H2O2 |
2 hr, 80°C |
ND b |
ND b |
------- |
------- |
0.9999 |
0.9999 |
|
Dry Heat (100°C) |
24 hr |
ND b |
ND b |
------- |
------- |
0.9999 |
0.9999 |
|
Photo Degrn. (SUNTEST-XLS+) |
12 hr |
ND b |
ND b |
------- |
------- |
0.9999 |
0.9999 |
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT articles@pharmatutor.org
Subscribe to PharmaTutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE









