{ DOWNLOAD AS PDF }
ABOUT AUTHORS
Kajal V. Kosankar*, Dr. A.N. Aher
MVP samaj college of pharmacy, Gangapur road Nashik, Maharashtra, India
ABSTRACT
Ficus racemosa it belongs to family Moraceae syn. Ficus glomerata Roxb. is the major group of angiosperm (flowering plant). They are widely distributed throughout Asia, Africa, America, Australia, Himalayan ranges, Punjab, Khasia Mountain, Bihar, Orissa, Rajasthan, Deccan, China, Indonesia and Malaysia. Udumber is used from ancient time for treating the many diseases. This tree is used traditionally for public health. It is also called yajnodumbara and grows all over the India at different areas. The common name is ‘Audumbar’ and ‘Umbar’ and used in Unani, Ayurveda and Homeopathy. The leaves tender are used in kalasha prokshana it is sprinkle water to different places from a scared copper mug and the bark is used in Homa. In review macroscopically studies, microscopically studies and chemical constituents are studied on different parts of the plant. Traditional uses and pharmacological uses are described, the extraction of leaves, fruit, stem, bark and latex are done due to this the important constituents are isolated in respective solvent like ethanol, methanol, acetone, petroleum ether, ethyl acetate and water. The more information is described below and the people are moving towards ayurvedic preparations.
[adsense:336x280:8701650588]
Reference Id: PHARMATUTOR-ART-2631
|
PharmaTutor (Print-ISSN: 2394 - 6679; e-ISSN: 2347 - 7881) Volume 6, Issue 12 Received On: 06/11/2018; Accepted On: 20/11/2018; Published On: 01/12/2018 How to cite this article: Kosankar, K. and Aher, A.N. 2018. The Phytoconstituents and Pharmacological Actions of Ficus racemosa Linn (Family: Moraceae) - An updated review. PharmaTutor. 6, 12 (Dec. 2018), 55-63. DOI:https://doi.org/10.29161/PT.v6.i12.2018.55 |
INTRODUCTION
In our country many plants are used traditionally, among that few are used for treatment of several diseases. Moraceae family- consists of fifty genera and 1400 species. Over world 850 species of Ficus are found. About 80% of population use herbal medicines for primary healthcare estimated by the WHO. India is rich in traditional and medicinal plant used for public healthcare. People are moving more towards traditional plant rather than allopathic medicines.
Ficus racemosa also called as ‘Audumbar tree’ & ‘Umbar tree’ grown in Maharashtra. It spelled as ‘Cluster fig’ in English. (Paarakh et al., 2008) Common name is ‘Goolar’. The importance of indigenous medicines is coming from generation to generation. India is one of rich country for plants resource and they are valuable medicines used in Ayurveda, Unani, Siddha and other system of medicines.
By isolating the important components from different parts of plant like: roots, stems, leaves, barks, flowers and fruits formulation can be made and used for many diseases like: Roots- hydrophobia, Barks- galactogogue, Fruits- blood disorders, dry cough, burning sensation, leprosy, menorrhagia, Leaves- astringent, bronchitis. The leaf of F. racemosa contains Flavonoids, triterpinoids, alkaloid and tannins. The fruit have glauanol acetate, tiglic acid, taraxasterol, lupeol acetate and many more constituents are present in other part of plant.
They are widely distributed throughout Asia, Africa, America, Australia, Himalayan ranges, Punjab, Khasia Mountain, Bihar, Orissa, Rajasthan, Deccan, China, Indonesia and Malaysia. The reproductive system is unique because it involving synconia fig and specialized pollinator wasps.
VERNACULAR NAMES (Shah et al., 2016)
Sanskrit – Udumbara
Hindi – Goolar
Marathi – Umbar
Bengali – Jagnadumbar
Telugu – Paidi
Oriya – Dimri
Malayalam – Atti
Nepali – Gular, Dumri
Kannada – Rumadi
Tamil - Atti
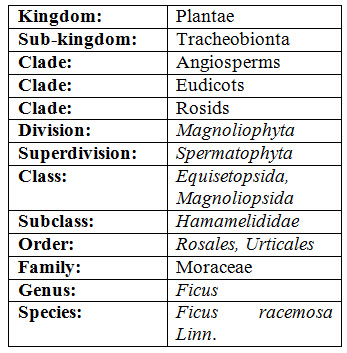
TAXONOMICAL CLASSIFICATION
“The study of the identification, taxonomy and nomenclature of organisms, including the classification of living things with regard to their natural relationships and study of variation and the evolution of taxa”. Classification of Ficus racemosa is given below: (Shah et al., 2016)
HISTORY
It is a one of the herb mentioned in all ancient scriptures of Ayurveda. Udumbara is considered sacred to God Dattaguru, has various synonyms like yajnanga, yajniya, yajnayoga and yajnyasara suggesting the use in ritual sacrifice. The ksiri viksa produced on cutting or plucking the leaf, latex oozes out. The plant is from a group, called pancavalkala. Maharishi Charaka has categorized Udumbara as anti-diuretic herb (mutra sangrahaniya). Susruta has described the properties of the plant, like astringent, promotes callus healing in fractures (bhagna sandhaniya) and useful in vaginal disorders. The plant was located on south-western coast (middle-earth) of the Bay of Belfalas,

Fig.1 Fruit of Ficus racemosa Linn.
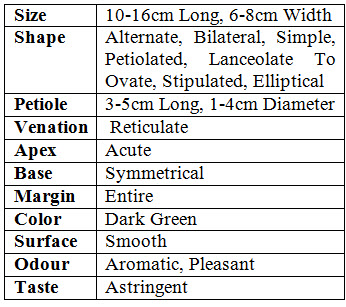
MACROSCOPICAL STUDY
MICROSCOPICAL STUDY (Ahmed et al., 2010)
• Cork: It contains polygonal or rectangular cells. The cork is made up of polygonal or rectangular cells. The phellogen contains 1-2 layers of thin walled cells.
• Phelloderm: It contains compact tissue of parenchymatous cells or small groups of sclereids and it is lignified with simple pits. Several parenchymatous cells which contains single prism of calcium oxalate brownish content.
• Cortex: It contains numerous sclereids they are rectangular or isodiametric and pitted thick walled and cortical cell contains resinous mass. Prismatic crystals of calcium oxalate are present in cells. Phloem contains sieve tubes, companion cells, phloem parenchyma, sclereids, phloem fibers and medullary rays. Starch grains they are present in ovoid to spherical. Laticiferous vessels are brown granular present in phloem region. Cambium present in 2-3 layer of tangentially elongated thin walled cells.
• Leaf: It shows dorso-ventral characters with single layer of palisade cells at upper epidermis. Lower and upper epidermis showed abundant and uniserate, unicellular, thin walled covering trichromes on the upper epidermis.
• Mesophyll is middle part between lower and upper epidermis of the leaf, contains sclerenchymatous cells covering to vascular bundle. Collenchymatous cells are present in mesophyll between vascular bundles and lower epidermis.
• Vascular bundle contains xylem and phloem arrangement at middle of mid rib.

Fig2. Tree of Ficus racemosa Linn.
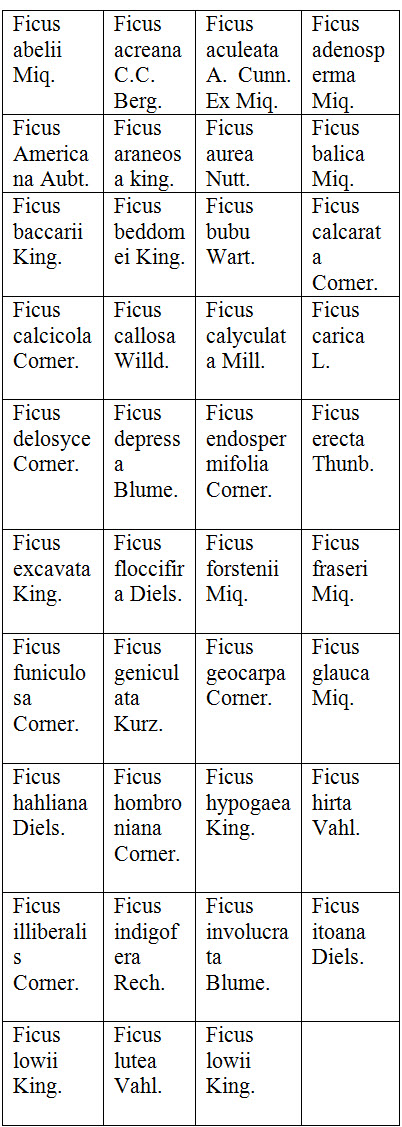
SPECIES OF FICUS
The different species of ficus belong to family Moraceae are given below:
CHEMICAL CONSTITUENTS:
The racemosa Linn species contains primary and secondary metabolites like, carbohydrates, mucilage, alkaloids, flavonoids, steroids, tannins, terpinoids, phenolic substance, glycosides, saponins, coumarins, triterpenoids, α phenolics, bergapten, bergaptol, lanosterol, stigmasterol, lupen-3-one, β-sitosterol-d-glucoside and vitamin K₁, α hydroxyl ursolic acid, protocatechuic acid, oleanolic acid, rusolic acid, maslinic acid also used for treating many diseases.
The non-enzymatic constituents like phenolic components flavonoids, vitamin C and the enzymatic constituents like ascorbate oxidase, ascorbate peroxidase, catalase peroxidase.quercitin-3-glucoside, rutin and methyl esters of leucoanthocyanins are obtained from leaves.
Bark – Tannin, wax, saponin, β-sitosterol, steroids, alkaloids, gluanol acetate, leucopelargonidin-3-O-α-L-rhamnopyranoside, leucopelargonidin-3-O–β-D-glucopyranoside, leucocyanidin-3-O-β-D-glucopyrancoside, (Ahmed et al., 2010) lupeol, ceryl behenate, lupeol acetate, α-amyrin acetate, leucocyanidin and leucoanthocyanin, kaempferol, coumarin, ellagic acid, α-amyrin, stigmasterol, quercetin, bergenin and racemosic acid.
Leaves - Campestrol, arabinose, bergapten, psoralenes, ficusin, (Baruah et al., 1992), stimasterol, isofucosterol, α-amyrin, lupeol, tannic, arginine, serine, aspartic acid, glycine, threonine, alanine, proline, tryptophan, tyrosine, methionine, valine, isoleucine, leucine, n-nonacosane, n-hentricontanen, hexa-cosanol, n-octacosan gluanol acetate, racemosic acid, quercitin-3-glucoside, rutin and methyl esters of leucoanthocyanins. (Mandal et al., 1999)
Fruits - The phenolic compounds like gallic acid and ellagic acid. Furanocoumarins like psoralen, bergapten, β- sitosterol tetracyclic triterpene, glaunol acetate, Glucanol, tiglic acid, taraxasterol, lupeol acetate, friedelin, hydrocarbons, sterols, glycosides, carbohydrates, hentriacontane and α-amyrin.
Latex - Euphol, isoeuphorbol, β-sitosterol, 4-deoxyphorbol, cycloartenol and cycloeuphordenol.
TRADITIONAL USES:
Ficus racemosa all parts of plant are used traditionally from longer time to cure the diseases, different parts uses are given below: (Ahmed et al., 2010)
Bark: Used for multiple purpose like Galactagogue and gynecological disorders. Decoction of the bark used in piles, ulcerative colitis, diarrhea and dysentery, diabetes, astringent, menorrhea, hemoptysis, wash wounds, abortions, uropathy, piles and asthma.
Sap of bark: Diabetes, burns, swelling, leucorrhea, dysentery and diarrhea
Roots: Hydrophobia, dysentery, pectoral, complications, diabetes and mumps. Fruits: Active against leprosy, menorrhagia, leucorrhoea, burns, intestinal worms, astringent, stomachic, menorrhea, hemoptysis, visceral obstruction, diarrhea, constipation dry cough, urinary tract infection and blood disorders, more potent for diabetes treatment.
Leaves: Bronchitis, bowel syndrome, piles, bilious infections, douche in dysmenorrhea, ulcers. Leaf juice used to prevent hair splitting. The decoction of leaves used in wound washing and healing.
Latex- Externally applied on wounds so that it shows anti- inflammatory activities, aphrodisiac, diarrhea, dysentery, hemorrhoids, stomachache, cholera, mumps pain, edema, boils, blisters, measles, promote healing and most important is adhesive.
Sap of root: Gonorrhea, diabetes, mumps, heat stroke, chronic wounds and malaria in child.
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT editor-in-chief@pharmatutor.org
Subscribe to Pharmatutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE
PHARMACOLOGICAL USES
• Hypoglycemia activity
• Antioxidant activity
• Hepatoprotective activity
• Antitussive activity
• Antiulcer activity
• Wound healing
• Anthelmintic activity
• Antidiuretic activity
• Antidiarrheal activity
• Chemo-preventive activity
• Anticancer activity
• Antibacterial activity
• Anti-inflammatory activity
• Memory enhancing activity
• Hypolipidemic activity
• Renal & testicular protective action
• Analgesic activity
• Antifungal activity
• Angiotensin converting enzyme inhibitor activity
• Cardioprotective activity
• Gastroprotective activity
• Antifilarial activity
• Antifertility activity
• Larvicidal activity
• Antipyretic activity
Hypoglycemic activity: (Ushir et al., 2009)
Ficus racemosa shows hypoglycemic activity, to evaluate the study of hypoglycemia activity ethanol extract is used (250mg/kg/day, p.o.) lowering of blood glucose level was determined in 2 weeks. Alloxan diabetic albino rats they conforms hypoglycemic activity. Methanolic extract of stem bark also show glucose lowering activity at dose 200-400mg/kg, p.o. this test is done on normal and alloxan induced diabetic rats. Whereas, this activity was compared with standard antidiabetic agent glibenclamide at dose 10mg/kg it shows antidiabetic activity. Β-sitosterol isolated from stem bark it is more potent when compared with other isolated compound. Methanol extract of fruit given in 1,2,3, and 4g/kg it lowers the blood glucose level in normal and alloxan induced diabetic rabbits. α-amyrin acetate is important constituent isolated form fruits and the dose is given at 100gm/kg which lowers the blood glucose level in 5 to 24hrs gives result at 18.4% and 17.0% in sucrose when compared with streptoratocin induced diabetic rat model.
Antioxidant activity: (Sultana et al., 2013)
The 1,1-diphenyl-2-picrylhydrazyl radical (DPPH) used to evaluate antioxidant activity by free radical scavenging method. DPPH free radical reduced when hydrazine react with hydrogen donors, it forms stable DPPH molecules through donation of hydrogen. Due to this method it is easy to determine antiradical power of an antioxidant activity by decrease in absorbance of DPPH at 519nm. Color change from purple to yellow, there is discolorations of DPPH when absorbance of methanolic extract of stem and leaves was measured at 517nm. The extract of stem and leaves was compared with standard butylated hydroxytoluene (BHT) by free radical scavenging method the extract show antioxidant activity.
Hepatoprotective activity: (Deep et al., 2013).
The leaves extract and stem bark extract shows hepatoprotective activity in rats by inducing chronic liver damage by subcutaneous injection of 50% v/v carbon tetrachloride in liquid paraffin at dose of 3ml/kg, it is given in alternate days for 4 weeks. Stem bark extract dose given in 250 and 500mg/kg, all biochemical parameters like SGOP, SGPT, serum bilirubin and alkaline phosphate was evaluated and it is compared with standard silymarin.
Antitussive activity: (Ahmed et al., 2010) Methanol extract (200mg/kg) was compared with standard codeine phosphate (10mg) that is evaluated in sulfur dioxide gas-induced cough in mice. Maximum activity was seen at 90 min after administration of the bark extract, maximum inhibition is 56.9%.
Antiulcer activity: (Yadav et al., 2015)
50% of ethanolic extract of fruit used as antiulcer and reduce oxidative damage at mucosal lining of stomach. The studies are done on animal models of rats at dose 50, 100 and 200mg/kg, twice a day for 5 days. For evaluating the antiulcer activity the models are used like ethanol, pylorus ligation and cold strain-induced ulcer.
Wound healing activity: (Biswas et al., 2003)
Ointment was prepared by using powder of F. racemosa with petroleum jelly 15%w/w. The study was done on Charles Foster strain rats by using 8mm of full-thickness punch wound model. It heals highly significant generation of tissue DNA, RNA and total protein was observed by using ointment.
Anthelmintic activity: (Chandrashekhar et al., 2008)
Earthworms lost their activity when it comes in contact with ethanolic bark extract at 5, 10, and 50 mg/ml given dose-dependently. There is loss of mortality and cause death. The higher concentration of bark extract shows paralytic effect and death time is shorter and it is compared with standard piperazine citrate 3% within 90 min.
Antidiuretic activity: (Deep et al., 2013)
Decoction of Ficus racemosa bark given at dose at 250, 500 or 1000 mg/kg body weight it is given for 5 hrs at 1 hr of interval. It shows reduction in Na+ level and Na+/K+ ratio and increase urinary osmolarity.
Antidiarrheal activity: (Deep et al., 2013)
Ethanol extract of stem bark was stuied on different experimental models of diarrhea in rats. The inhibitory activity was observed against castor oil induced diarrhea, PGE2 induced enteropooling and charcoal meal tests this was done in rats. By using this extract on rats it shows antidiarrheal activity.
Chemo-preventive activity: (Ahmed et al., 2009)
The is treatment given orally to the rats by two methods, incidence tumors in ferric nitrilotriacetate (Fe-NTA) it induced chemotoxicity in rats and potassium bromate- induced nephrotoxicityin in rats. Ficus racemosa extract (PM Paarakh, 2009) was given at 200 and 400mg/kg BW it results in decrease of xanthin oxidase, γ-glutamyl tranpeptidase, lipid peroxidation and H2O2. It also shows recovery in blood urea nitrogen, serum creatinine, renal OCD activity and DNA synthesis, by evaluating decrease in all this parameters it shows chemopreventive activity.
Anticancer activity: (Yadav et al., 2015)
Methanolic extract shows cytotoxic effects on more than one hepatic cancer cell lines like, HL-60, HepG2, NCI-H23 and HEK-293T. Among this evaluation more cytotoxic activity was observed in HL-60 and HepG2 cells at 50% inhibitory concentration (IC50).
Antibacterial activity: (Ahemed et al., 2010)
Ficus racemosa extract at different solvent used as antibacterial, like stem bark ethanolic extract found active against Pseudomonas aeruginosa, Proteus mirabilis, Staphylococcus aureus, Bacillus cereus, Alcaligenes faecalis and Salmonella typhimurium. Aq. Extract shows activity against Streptococcus faecalis. Methanolic extract shows effect against Bacillus subtilis. Petroleum ether (Paarakh et al., 2009) shows effect against E coli, Bacillus pumitis, Bacillus subtilis, Pseudomonas aeruginosa and Staphylococcus aureus. Hydro alcoholic (Deep et al., 2013) extract of leaves shows effect against Actinomyces vicosus at minimum inhibitory concentration of 0.08mg/ml.
Anti-inflammatory activity: (Ahmed et al., 2010)
Petroleum ether of leaves shows anti-inflammatory activity against inflammation producing histamine and serotonin. It reduces edema induced by dextran and it is mediated histamine and serotonin. The ethanol extract of bark inhibits COX-1, 5LOX, phospholipase A2 and biosynthesis of PGE2. The petroleum ether of leaves at 200-400mg/kg bw it shows activity on carrageenan, serotonin, histamine and dextran-induced at rat hind limb paw edema. Maximum effect was shown at 400mg/kg in chronic test, when it was compared with phenylbutazone and non- steroidal anti-inflammatory agent (Mandal et al., 2000).
Memory enhancing activity: (Devansh et al., 2013)
It shows activity on Alzheimer disease which is caused by decrease in acetylcholine level and it is an extrapyramidal disease.
Hypolipidemic activity: (Deep et al., 2013)
Ethanolic extract of bark was given at dose 100-500mg/kg bw by inducing alloxan in diabetic rats. It shows hypolipidemic activity when compared with standard glibenclamide and it also shows hypocholesterolemic effect when rats were induced by dietary fibre content fruits in diet. The evaluation results shows increased in fecal extraction of cholesterol and bile acids.
Renal and testicular protective activity: (Yadav et al., 2015)
Acetone extract of bark was given at 250-500mg/kg it shows effect against doxorubicin induced renal and testicular toxicity. The extract was given at 500mg/kg it shows sperm count and spermatogenesis at higher level when it compared with normal control dose and antioxidant activity also observed at same dose.
Analgesic activity: (Zulfiker et al., 2010)
Ethanol extract of bark and leaves was given and analgesic activity was evaluated by using hat plate and tail immersion method. The dose was given at 300mg/kg I.P. The leaf extract shows increase in latency time and giving protection about 40.1% the bark extract shows increase in latency time and giving protection about 35%. The analgesic activity observed due to presences of important constituent. (Malairajan et al., 2006) The decoction of leaves of F. racemosa gives analgesic activity was evaluated when acetic acid writhing test was done in mouse. Due to this there is decrease in number of writhes. This extract was given to mice and the result was evaluated by hat plate test the analgesic activity till 3hrs was observed. (Ahmed et al., 2010) The ethanol extract of bark and leaves was administered dose dependently the analgesic activity was observed by analgesiometer at 100, 300 & 500mg/ml. (Paarath et al., 2009)
Antifungal activity:
The evaluation of antifungal activity different species of fungi were used Trichophyton mentagrophytas, Trichophyton rubrum, Trichophyton soundanense, Candida albicans, Candida krusei & Torulopsis glabrata. (Shah 2016 t al., 2016) For the evaluation of anti-fungal 50% methylene chloride in hexane flash activity column fraction extract of leaves were used for the examination. The extract inhibits growth of given pathogens like Curvularia sp, Colletotrichum gloeosporioides, Alternaria sp, Corynespora cassiicola & Fecsarium sp, due to presences of psoralen in extract. (Joseph et al., 2010)
Angiotensin converting enzyme inhibitor activity: (Ahmed et al., 2010)
Cold aqueous extract (FRC) and hat aqueous extract (FRH) given close dependently & shows inhibition activity against porcine kidney & rabbit lung ACE. As compare to cold aqueous extract hat aqueous extract shows higher angiotensin activity both extract shows higher inhibition on porcine kidney and rabbit lung ACE. FRH lower IC50 values at concentration 1.36 &1.91 µg/ml for porcine kidney and rabbit lung ACE when compared with FRC at concentration 128 and 291 µg/ml. Angiotensin converting enzyme inhibition studied by radial scavenging activity. The both extract was given dose dependently it shows maximum inhibition about 87% and 75% for both extract FRC and FRH at concentration 25 µg/ml FRH at 10.8 µg/ml shows lower IC50 value compared with 15.8 µg/ml & 16.5 µg/ml concentration of FRC and BHT (butylated hydroxytoluene)
Cardiopratective activity: (Voumik et al., 2010)
Acetone extract of F-racemes Linn bark it shows cardio protective activity against doxorubicin-induced cardio toxicity in rats the cardio protective activity was evaluated by biochemical parameters are CK-MB, LDH, AST, ALT, troponin I, thiobarbituric acid and glutathione.
Gastroprotective activity:
Ethanolic extract of fruits, bark & leaves given dose dependently which shows inhibition of ulcer index in pylorus ligation, ethanol, cold resistant stress-induced ulcers, lipid peroxidation, superoxide dismutase, H+ K+ AT pose and increase the activity of enzyme catalase it also shows gastric mucosal protection this activity was observed due to administration of fruit extract gastroprotective activity was evaluate by against acidity ulcer index in aspirin, pylorus ligation-induced gastric ulcer, HCL-ethanol mixture, water immersion stress-induced ulcer it shows reduction in gastric lesion and gastric mucous protection administration of 50% ethanolic extract of unripe fruit of Ficus racemasa at100, 200, & 300 mg/kg in rats gives gastroprotective effect when compared with ethanol 4 hrs pylorus ligation-induced gastric ulcer more effective at dose 300 mg/kg compared with sucarlfate 250mg/kg administration of methanolic extract of unripe fruit was given at does 100, 200 & 400 mg/kg. The gastric ulcer was induced by aspirin and cold restrainst stress. (Sangameswaran et al., 2008)
Anti-filarial activity: (Mishra et al., 2005)
Alcoholic and aqueous extract of fruit shows inhibition of spontaneous motility of worm and nerve muscle of setaria cervi and increase in amplitude and due to concentration microfilarial causes death at 21 and 35 ng/ml for alcohol extract and 27 and 42 ng/ml at aqueous extract due too in vitro study at LC50 and LC90.
Antifertility activity: (Dheeraj et al., 2011).
The extract shows anti fertility about 70% reduction in sperm count, motility, viability & abnormal morphology was determined reduction in weight of reproductive organ and the level of sialic acid in epididymis and fructose in seminal vesicle the bark extract shows 80% of vaginal contraception.
Larvicidal activity:
The hexane, ethyl acetate, petroleum ether, acetone & methanol extracts of leaf & bark for evaluation of toxicity against fourth instar larval of culex quinquefas ciatus after 24hrs motility and all extracts shows moderate larvicidal effects the acetone extract of bark shows higher larvicidal activity & also separation & identification of gluanol acetate due to bioassay guided fractionation method it also identify new mosquito larvicidal compound. Antilarvicidal activity against fourth-instar larval of Aedes aegypti (LC50 14.55 & LC90 64.99ppm) Anopheles stephensi (LC50 28.50 & LC90 106.50 ppm) & C. quinquefasciatus (LC50 41.42 & LC90 192.77 ppm) it is observed due to important constituents gluanol acetate an (Rahuman et al., 2008).
Antipyretic activity:
Methanolic extract of bark was given at dose 200 and 300 mg/kg dose dependently antipyretic effect was observed that body temperature reduces in both induced pyrexia in albino rats the evaluation of antipyretic activity the extract was compared with standard drug i.e paracetamol the aqueous and petroleum ether extract of leaves shows antipyretic activity when compared with indomethacin against yeast induced pyrexia introduced in rats.
Other uses:
The unripe fruit of F. racemose are used for culinary purpose, pickles, chutney, sambar, curry & sabji.
SIDE EFFECTS
1. F. racemose is coolant tree, precaution is taken while using kapha dominant person with recurrent allergic rhinitis, cough & cold.
2. Ripe fruit avoided in use of culinary it cause or worsen the intestinal worm infestation.
3. Safety is taken during pregnancy.
SUMMARY
1. Overall study gives information about whole plant.
2. The different parts of plant i.e. fruit, leaves, stem bark is used on various diseases.
3. The medicinal plants are used traditionally form ancient time.
4. Due to use of Ayurvedic drug there is less toxicity in chemotherapy so, the people are moving towards traditional medicines.
5. The information in taken form many review articles and research paper it gives details about many uses.
6. The evaluation parameters are given for the determination of activity of the various extract.
7. By using various concentrations of leaves, bark stem, fruit, latex extract it shows antimicrobial activity.
8. The morphological studies & microscopically studies are done by comparing the leaves with monographs given in I.P. & B.P.
9. Overview on many papers it shows curing of many diseases.
10. Chemical constitutions studies are also done on different part of plants.
11. The side effect of F. racemosa is given, so that before using the extract the precaution can be taken.
12. Decoction of the leaves and bark is cost effect and shows treatment on many diseases.
CONCLUSION
Umber is use for public health because the studies proved the umber can be used in day to day life. The medicinal plants are widely used by medicinal practitioner for the treatment of the diseases. The extract of different part of plant like leaves, latex, fruits & bark shows pharmacological action. The important constituents are isolated from plant and it is responsible for curing the diseases like anti-diabetic, anti-diarrheal, anti-bacterial, anti-fungal, anti-ulcer, anti-wormicidal, anti-hypoglycemic, hypolipidemic, anti-diuretic and anti-inflammatory. It is popular indigenous system of medicines like Ayurveda, Siddha, Unani and Homeopathy. It takes vital place in traditional system. The study explores the potential of plant and signifies its importance in the pharmaceutical industries. The plant is widely available all over the India. It has antibiotic and antibacterial properties due to this the formulation can be prepared like lotion and cream and has antioxidant property due to this it is helpful in chemoprotection and radioprotection. It is used in food preparation but the safety must be taken while giving in pregnancy.
REFERANCES:
1. Ahmed F and Urooj A (2010); Traditional uses, medicinal properties, and phytopharmacology of Ficus racemosa: a review; Pharm.Biol; 48(6);672-81.
2. Ahmed F and Urooj A (2011); Pharmacognostical studies on Ficus racemosa stem bark; PHCOG J; 3(19);19-24.
3. Bhalerao S.A., Verma D.P., Teli N.C., Dinwana V.S and Thakur S.S (2014); Ficus racemosa Linn: Acomprehensive review; JOAC; 3(4);1423-31.
4. Bheemachari J., Ashok K., Joshi N.H., Suresh D.K and Gupta V.R.M (2007); Antidiarrhoeal evaluation of Ficus racemosa Linn. Latex; Acta Pharm Sci; 49;133-8.
5. Chandrashekhar C.H., Latha K.P., Vagdevi H.M and Vaidya V.P (2017); Anthelmintic activity of the crude extracts of Ficus racemosa; IJGP; 100-3.
6. Choudhary S., Pathak AK., Khare S and Kushwah S (2011); Evaluation of antidiabetic activity of leaves and fruits of Ficus religiosa Linn; IJPLS; 2(12);1325-7.
7. Deep P., Singh A.K., Ansari T and Raghav P (2013); Pharmacological Potentials of Ficus racemosa – A review; Pharm Sci Rev Res; 22(1);29-34.
8. Ganatra S.H., Durge S.P and Patil S.U (2012); Preliminary Phytochemicals Investigation and TLC Analysis of Ficus racemosa Leaves; J Chem pharm Res; 4(5);2380-4.
9. Mandal S.C., Maity T.K., Das J., Saba B.P. and Pal M (2000); Anti-inflammatory evaluation of Ficus racemosa Linn. Leaf extract; J Ethnopharmacol; 72;87-92.
10. Mishra R. and Tiwari A.K (2013); Phytochemical and chromatographic studies in the latex of Ficus racemosa Linn; pelagia research library; 3(4);150-4.
11. Paarakh M.P (2009); Ficus racemosa Linn.-An overview; Nat Prod Rad; 8(1);84-90.
12. Patil V.V., Pimprikar R.B., Sutar N.G., Barhate A.L and Patil L.S., et al (2009); Anti- Hyperglycemic activity of Ficus racemosa Linn leaves; JPR; 2(1);54-7.
13. Poongothai A., Sreena K.P., Sreejith K. Uthiralingam M and Annapoorani S (2011); Preliminary phytochemicals screening of Ficus racemosa Linn. Bark; IJPBS; 2(2);431-4.
14. Rahuman A.A., Venkatesan P., Geetha K., Gopalakrishnan G., Bagavan A and Kamaraj C (2008); Mosquito larvicidal activity of gluanol acetate, a tetracyclic triterpenes derived from Ficus racemosa Linn; Parasitol Res; 103; 333-9.
15. Rao B.R., Anupama K., Swaroop K.R.L., Murugesan T., Pal M and Mandal S.C (2002); Evaluation of anti-pyratic potential of Ficus racemosa bark; Phytomedicine; 731-3.
16. Shah S.K., Garg G., Jhade D and Pandey H (2016); Ficus racemosa Linn: its Potentials Food Security and Rural Medicinal Management (review article); J Pharm Sci. & Res.; 8(5);317-22.
17. Sultana J., Kabir A.S., Hakim A., Abdullah M., Islam N and Reza A (2013); Ealuation of the antioxidant activity of Ficus racemosa plant extracts from north-western district of Bangladesh; J. Life Earth Sci; 8;93-9.
18. Sumi S.A., Siraj A., Hossain A., Mia S., Afrin S and Rahman M (2016); Investigation of the Key Pharmacological Activity of Ficus Racemosa and Analysis of its Major Bioactive Polyphenols by HPLC-DAD; Hindawi; 1-9.
19. Ushir Y.V., Tiwari K.J. and Kare P.T (2015); Cecidological and pharmacognostical study of Ficus racemosa leaf galls; JPP; 4(4);41-4.
20. Veerapur V.P., Prabhakar K.R., Thippeswamy B.S., Bansal P and Srinivasan K.K (2012); Antidiabetic effect of Ficus racemosa Linn. Stem bark in high – fat diet and low-dose streptozotocin-induced type 2 diabetic rats: Amechanistic study; Food chem; 132;186-193.
21. Zhang G., Song Q and Yang D (2006); Phenology of Ficus racemosa in Xishuangbanna, Southwest China Biotropica; 38(3);334-1.
22. Zulfiker A.H.M., Rahman M.M., Hossain M.K., Hamid K., Mazumder M.E.H and Rana M.S (2010); In vivo analgesic activity of ethanolic extracts of two medicinal plants- Scoparia dulcis L. and Ficus racemosa Linn; Biol Med; 2(2);42-8.
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT editor-in-chief@pharmatutor.org
Subscribe to Pharmatutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE









