{ DOWNLOAD AS PDF }
ABOUT AUTHOR:
Yesmin Begum
Department of Pharmacy,
Southeast University, Banani, Dhaka
yesumyta@gmail.com
ABSTRACT
The present research aims to investigate the antibacterial and antioxidant activities of different leaf extracts of Aquillaria malaccensis. Antibacterial tests were done by Disc diffusion method using Kanamycin as standard. The chloroform extract showed good antibacterial activity against Shigella boydii and Escherichia coli with 19 mm and 17 mm zone of inhibition respectively. All other extracts showed mild to moderate antibacterial activity against other bacteria with 7 – 16mm zone of inhibition. The antioxidant activity of the extracts was determined by using DPPH spectrophotometrically and ascorbic acid was used as standard. All three extracts showed significant antioxidant activities having IC50 value of 38.96 µg/ml, 39.63 µg/ml and 64.75 µg/ml for ethyl acetate, n –hexane and chloroform extracts respectively.
INTRODUCTION
Aquillaria malaccensisor Agarwood (Family : Thymelaeaecae) is a large evergreen tree growing over 15-30 m tall and distributes inBangladesh, Bhutan, India, Indonesia, Iran (Islamic Republic of), Malaysia, Myanmar, Philippines, Singapore, Thailand.Agarwood has been used for medicinal purposes for thousands of years, and continues to be used in Ayurvedic, Tibetan and traditional East Asian medicine. Wood is heating, alterative tonic, carminative, laxative, stomachic, diuretic, aphrodisiac and febrifuge; useful in diseases of ear and skin, hiccup, leucoderma, chronic diarrhoea, bronchitis, asthma. The fragrant resinous substance is prescribed in gout and rheumatism. The bark is used for heart disease in Khagrachari. Besides these uses, various important pharmacological activities had been evaluated from Agarwood extract. Agarwood extract showed strong anticancer activity towards human colon cancer cells[1]. Antihyperglycemic and antioxidant activity of the leaves of A. malaccensis had also been reported [2, 3]. Medicinal uses and biological properties of Agarwood manifest to the presence of many bioactive chemical constituents. Agarwood is reach in alkaloids, tannins, phenols, terpenoids, quinones and flavonoids[4]. Apart from these, many compounds as 4-phenyl-2-butanone; Jinkoh-eremol; Alpha-guaiene; Beta-agarofuran; Alpha-bulnesene; Kusunol; Selina-3,11-dien-9-one; Oxo-agarospirol; Guaia-1 (10), 11-dien-15,2-olide; 1,2,3-propanetriol, monoacetate; Phenyl-β-D-glucoside, n-hexadecanoic acid,; Glycerine; Squalene; 2-propanone,1,3 dihydroxy; 3,7,11,15-Tetramethyl-2-Hexadecen-1-ol (Phytol); Dodecyl acrylate; 2,3-Dihydro-3,5-Dihydroxy-6-methyl-4H-pyran-4-on; 1-Tetradecanol; 6-ethyl-5-hydroxy-2,3n,7-trimethoxynaphthoquinone; 9,12,15-Octadecatrienoic acid, Octaethylene glycol monodecyl ether had also been isolated from the volatile oils of Agarwood [5,6]. Nine endophytic fungal strains were isolated with three new compounds as 1,7-dihydroxy-3-methoxyanthraquinone, propyl p-methoxy phenyl ether and 6-methoxy-7-O-(p-methoxyphenyl)-coumarin having moderate antimicrobial activity [7].
Drugs and chemicals
DPPH (1, 1-diphenyl - 2-picryl hydrazyl) was obtained from Sigma Aldrich USA. Ascorbic acid was obtained from SD Fine Chem. Ltd, Biosar, India. Kanamycin was collected from Square Pharmaceuticals Ltd., Bangladesh. Vincristine sulfate was collected from Alfa Asear Ltd. USA.
Instrumentation
The antioxidant potentiality was assayed from the bleaching of purple colored methanol solution of DPPH radical by the plant extracts using UV- spectrophotometer (Model NO. 1501PC Shimadzu, Japan) at 517 nm.
Collection and identification of the plant
The fresh leaves of A. malaccensis were collected in May 2014 fromKhagracharidistrict, Bangladesh and identified by Dr. M. A. Razzaque Shah, Tissue Culture Specialist, BRAC Plant Biotechnology Laboratory, Dhaka, Bangladesh.
Plant materials extraction and fractionation
The fresh leaf was collected, sun dried for seven days and ground. The dried powder of A. malaccensis leaf (200gm) was soaked in 600ml of methanol for 7 days and filtered through a cotton plug followed by Whatman filter paper number 1. The concentrated ethanolic extract of leaf (16 gm) was fractionated by the modified Kupchan partitioning method [8] into n-hexane, chloroform and ethyl acetate. The subsequent evaporation of solvents afforded n-hexane (450 mg), chloroform (700 mg) and ethyl acetate (350 mg) from leaf extract.
Microbial strains and culture media
Antimicrobial activity was carried out against six Gram negative bacteria such as Vibrio parahemolyticus, Vibrio mimicus, Pseudomonas aeruginosa, Shigella boydii, Escherichia coli, Salmonella paratyphi and three Gram positive bacteria such asSarcina lutea, Bacillus megaterium and Bacillus subtilis. These bacteria were chosen to be studied as they are important pathogens and also due to rapidly developed antibiotic resistance. The microorganisms were collected as pure cultures from the Institute of Nutrition and Food Science (INFS), University of Dhaka, Bangladesh. For bacteria, the culture media was prepared by nutrient agar, reconstituting with distilled water according to specification (2.8% w/v).
Antibacterial screening by disc diffusion method
Antibacterial activities of various extracts of A. malaccensis were carried out in vitro by the standard disc diffusion method [9]. In this method, solutions of known concentration (500 μg/disc) of the test samples were made by dissolving measured amount of the samples (50 mg) in 1 ml of methanol. Then sterile filter paper discs (5 mm diameters) were impregnated with known test substances and dried. The dried discs were placed on plates (Petri dishes, 120 mm diameters) containing a suitable medium (nutrient agar) seeded with the test organisms. Standard disc of kanamycin (30 μg/disc) and blank discs (impregnated with solvents followed by evaporation) were used as positive and negative control. These plates were kept at low temperature (4 ºC) for 24 hours to allow maximum diffusion. The plates were then kept in an incubator (37 º C) for 24 hours to allow the growth of microorganisms. Antibacterial activity of the test samples was observed by growth inhibition of organisms forming clear, distinct zone surrounding the discs. The antibacterial activity was expressed in terms of millimeter by measuring the diameter of the zone of inhibition. The greater zone of inhibition indicates the greater activity of the test material against the test organism.
Antioxidant activity using DPPH free radical scavenging activity
The free radical scavenging activity (antioxidant capacity) of the plant extracts on the stable radical 1,1-diphenyl-2- picrylhydrazyl (DPPH) were estimated by the method of Brand-Williams et al.,(1995)[10]. During this experiment the test samples of the extracts of A. malaccensis atdifferent concentrations were mixed with 3.0 ml of DPPH methanol solution. The antioxidant potentiality was assayed from the bleaching of purple colored methanol solution of DPPH radical by the plant extracts by UV- spectrophotometer at 517 nm. Ascorbic acid was used as a positive control. Percent scavenging of the DPPH free radical was measured using the following equation-
% DPPH radical scavenging = [1 – (As/Ac)] × 100
Here, Ac = absorbance of control, As = absorbance of sample solution.
Then % inhibitions were plotted against respective concentrations used and from the graph IC50 was calculated. The lower IC50indicates higher radical scavenging activity and vice versa.
Result and Discussion
Antibacterial assay
Antibacterial activity of A. malaccensis leaves was evaluated by Disc diffusion method using Kanamycin as standard. The extractspossess anti bacterial activity with average zone of inhibition 6 -17mm against nine human pathogenic gram positive and gram negative bacteria. The chloroform extract showed highest zone of inhibition of 19mm, 17mm and 16mm againstS. boydii, E. coliand P. aeruginosa. Ethyl acetate and n – hexane extracts also showed good antibacterial activity with 15 mm zone of inhibition against V. mimicusand P. aeruginosa. These extracts showed mild to moderate activity against other pathogenic bacteria. Among the all extracts the chloroform extract correlate the medicinal uses of Agarwood with the research study.
Antioxidant activity
Antioxidant activity of Agarwood leaves was evaluated by DPPH free radical scavenging activity using ascorbic as standard. The n-hexane, chloroform and ethyl acetate leaf extracts of Agarwood possess significant antioxidant activity withIC50 value of39.63µg/ml, 64.75µg/ml and 38.96µg/ml respectively whereas IC50 value ofascorbic acid was 13.96µg/ml. As is observed in Figure 1, the extracts showed concentration dependant DPPH radical scavenging activity that verifies the previous chemical reports.
CONCLUSION
The present study reveals Agarwood as a potential source of antimicrobial agents and natural antioxidants. The chloroform extract showed the strongest antibacterial activities that pursue further characterization in different models to obtain bioactive compounds. All extracts potentiate the medicinal uses of Agarwood having strong antioxidant activity. Safe natural antioxidants are of great demand in recent years. So further studies may also be conducted active constituents.
Table 1: Antibacterial Screening of different extracts of A. malaccensis.
|
Group |
Test Organisms
|
Diameter of Zone of Inhibition (mm) |
||||
|
n-hexane 500µg/ disc |
Chloroform 500µg/ disc |
Ethyl acetate 500µg/ disc |
Kanamycin 30µg/ disc |
|
||
|
Gram positive bacteria |
B. subtilis |
11 |
14 |
7 |
33 |
|
|
S. lutea |
7 |
14 |
8 |
29 |
|
|
|
B. megaterium |
9 |
12 |
8 |
30 |
|
|
|
Gram negative bacteria |
V. parahaemplyticus |
11 |
14 |
7 |
32 |
|
|
V. mimicus |
9 |
8 |
15 |
29 |
|
|
|
S. dysenteriae |
7 |
13 |
8 |
30 |
|
|
|
E. coli |
8 |
17 |
9 |
35 |
|
|
|
P. aeruginosa |
15 |
16 |
6 |
28 |
|
|
|
S. boydii |
11 |
19 |
10 |
34 |
|
|
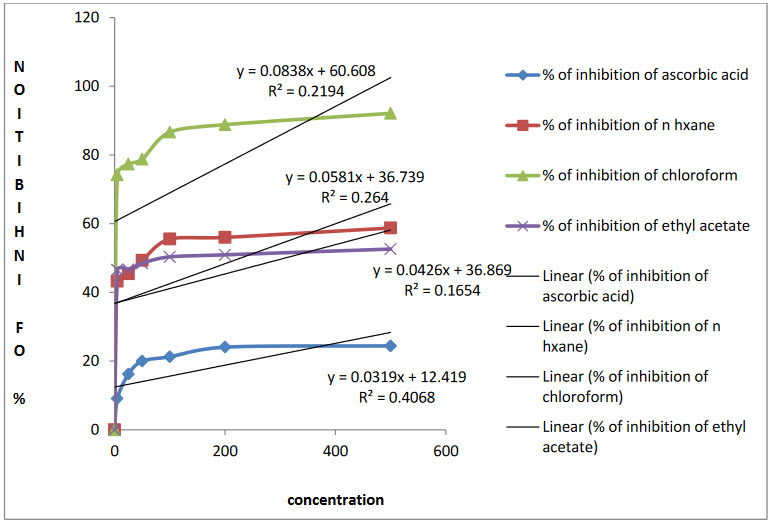
Figure 1: Determination of IC50 values for standard and n - hexane, chloroform and ethyl acetate extracts of A. malaccensis from linear correlation between of concentration versus percentage of scavenging of DPPH.
REFERENCES
1. A. H. Ibrahima, S. S. Al-Rawib, A. M. S. Abdul Majida, N. N. Ab. Rahmanc, K.M. Abo- Salahd, M. O. Ab Kadirb; Separation and fractionation of Aquilaria Malaccensis oil using supercritical fluid extraction and the cytotoxic properties of the extracted oil; Procedia Food Science; 2011; 1; 1953-1959.
2. Pranakhon, R., Pannangpetch, P. and Aromdee C., Antihyperglycemic activity of agarwood leaf extracts in STZ induced diabetic rats and glucose uptake enhancement activity in rat adipocytes; Songklanakarin J. Sci. Technol; 2011; 33(4); 405-410.
3. Huda AW N, Munira M A S, Fitrya S D, Salmah M; Antioxidant activity of Aquilaria malaccensis (Thymelaeaceae) Leaves; Pharmacog. Research. 2009; 1(5): 270-273.
4. Dash M, Patra J K, Panda P P.; Phytochemical and antimicrobial screening of extracts of Aquilaria agallocha Roxb; Afr. J. of Biotechnology, 2008; 7(20); 3531-3534.
5. Tajuddin SN, Yusoff MM; Chemical composition of volatile oils of Aquilaria malaccensis (Thymelaeaceae) from Malaysia; Nat Prod Commun; 2010; 5(12); 1965-8.
6. Khalil, A. S., Rahim, A. A., Taha, K. K., Abdallah, K. B.; Characterization of Methanolic Extracts of Agarwood Leaves; J. of Applied and Ind. Sciences; 2013; 1(3); 78-88.
7. Mohammad Shoeb, Shahanara Begum, Nilufar Nahar; Study of an endophytic fungus from Aquilaria malaccensis Lamk; Bangladesh Journal of Pharmacology; 2010; 5(1); 21-24.
8. Van Wagenen, B.C. R. Larsen, J.H.Cardellina, D.Ran dazzo, Z.C. Lidert and C.S withenbank, Ulosantoin; a potent insecticide from the sponge Ulosa ruetzleri; J Org Chem. 1993; 58(2); 335 -337.
9. Bauer, A.W., Kirby, W.M.M., Sherries and Tuck M; Antibiotic susceptibility testing by a standardized disc diffusion method; American J. Cli. Path; 1966; 45(4); 493-496.
10. Brand-Williams W, Cuvelier ME, Berset C; Use of free radical method to evaluate antioxidant activity; Lebensmittel Wiss. and Technologie; 1995; 28(1); 25-30.
REFERENCE ID: PHARMATUTOR-ART-2391
|
PharmaTutor (Print-ISSN: 2394 - 6679; e-ISSN: 2347 - 7881) Volume 4, Issue 2 Received On: 12/09/2015; Accepted On: 21/09/2015; Published On: 01/02/2016How to cite this article: Begum Y; Study on Agarwood (Aquilaria malaccensis) to evaluate Antibacterial and Antioxidant activities of n-hexane, chloroform and ethyl acetate extracts; PharmaTutor; 2016; 4(2); 47-50 |
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT editor-in-chief@pharmatutor.org
Subscribe to Pharmatutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE









