{ DOWNLOAD AS PDF }
 ABOUT AUTHOR:
ABOUT AUTHOR:
Bushra Shamim
Department of Pharmaceutics,
Faculty of Pharmacy,
Hamdard University, New Delhi, India
bushrashamim21@gmail.com
ABSTRACT
This is a review article on potential treatments for Ebola virus disease, also known as Ebola haemorrhagic fever. Ebola fever has emerged as one of the deadliest known forms of hemorrhagic fever since discovered in 1976. Transmission among humans occurs through the exchange of blood and body secretions. The Ebola virus causes an acute, serious illness which is often fatal if untreated. In this article experimental treatments like blood, immunological and drug therapies are discussed that are under development
[adsense:336x280:8701650588]
REFERENCE ID: PHARMATUTOR-ART-2302
|
PharmaTutor (ISSN: 2347 - 7881) Volume 3, Issue 1 Received On: 05/11/2014; Accepted On: 09/11/2014; Published On: 01/01/2015How to cite this article: B Shamim; Deadly but Curable: A Review on Potential Treatments for Ebola Haemorrhagic Fever; PharmaTutor; 2015; 3(1); 14-18 |
INTRODUCTION
Ebola hemorrhagic fever is an acute viral syndrome that presents with fever and an ensuing bleeding diathesis that is marked by high mortality in human and non- human primates.[2] Ebola virus disease, formerly known as Ebola haemorrhagic fever, is a severe, often fatal illness in humans.[1] Fatality rates are between 50% and 100%. Ebola virus is a class 4 pathogen belonging to the family Filoviridae[1][2], which causes fever and severe hemorrhaging and for which there is no specific treatment.[2]
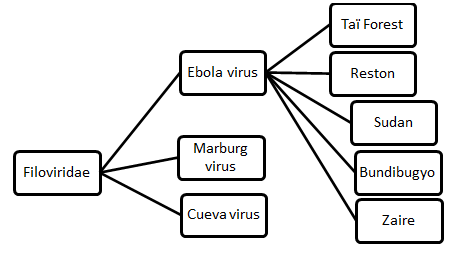
The virus family Filoviridae includes 3 genera:
Bundibugyo ebola virus, Zaire ebola virus, and Sudan ebola virus have been associated with large outbreaks in Africa. The first Ebola outbreaks occurred in remote villages in Central Africa[1][2], near tropical rainforests, but the most recent outbreak in west Africa has involved major urban as well as rural areas.[1]
There is no FDA-approved antiviral treatment [1] for Ebola hemorrhagic fever. Incubation ranges from 2 to 21 days. Patients who are able to mount an immune response to the virus will begin to recover in 7 to 10 days and start a period of prolonged convalescence. Supportive management of infected patients is the primary method of treatment, with particular attention to maintenance of hydration, circulatory volume, blood pressure, and the provision of supplemental oxygen.[2] Ebola is transmissible from person to person via direct contact with an infected patient’s blood or other body fluids. All body fluids (blood, saliva, urine, and stool) contain infectious virions and should be handled with great care.[3] Airborne transmission of Reston ebola virus is known to have occurred among primates; thus, although most cases in humans occur after direct contact with a patient or their blood or body fluids, transmission of Ebola virus via the airborne route cannot be dismissed.[3] Containment of Ebola virus greatly depends on the isolation of infected patients[2] along with the strict observance of barrier nursing techniques and by taking universal precautions for the handling of infectious material.[1][2][3]
EXPERIMENTAL TREATMENT AND VACCINES
[adsense:468x15:2204050025]
Potential treatments including blood products, immune therapies and drug therapies are currently being evaluated. Combinant human monoclonal antibody directed against the envelope GP of Ebola has been demonstrated to possess neutralizing activity. This Ebola neutralizing antibody may be useful in vaccine development or as a passive prophylactic agent.[1][3]
Supportive therapy with attention to intravascular volume, electrolytes, nutrition, and comfort care is of benefit to the patient. Intravascular volume repletion is one of the most important supportive measures.[3] Survivors can produce infectious visions for prolonged periods. Therefore, strict barrier isolation in a private room away from traffic patterns must be maintained throughout the illness.[1][3] Patient’s urine, stool, sputum, and blood, along with any objects that have come in contact with the patient or the patient’s body fluids (such as laboratory equipment), should be disinfected with a0.5% sodium hypochlorite solution. Patients who have died of Ebola virus disease should be buried promptly and with as little contact as possible.[1][2][3]
Nucleoside analogue inhibitors of the cell-encoded enzyme S-adenosyl homocysteine hydrolase (SAH) have been shown to inhibit Zaire ebola virus replication in adult BALB/cmice infected with mouse-adapted Ebola virus. Inhibition of SAH indirectly inhibits transmethylation reactions required for viral replication. Treatment response was dose-dependent. When doses of 0.7 mg/kg or more every 8 hours were begun on day 0 or 1of infection, mortality was completely prevented. Even when the drug was given on day2, 90% survived.[3]
Smith et al found that in rhesus macaques infected with a lethal dose of Ebola virus, treatment with interferon beta early after exposure led to a significant increase in survival time, though it did not reduce mortality significantly. These findings suggest that early post exposure interferon-beta therapy maybe a promising adjunct in the treatment of Ebola virus infection.[3]
Passive immunity has been attempted by using equine-derived hyper immune globulins and human-derived convalescent immune globulin preparations. In Ebola virus -infected cynomolgus macaques , use of human recombinant interferonalfa-2b in conjunction with hyper immune equine immunoglobulin G (IgG)delayed but did not prevent death. Equine IgG containing high-titer neutralizing antibodies to Ebola virus protected guinea pigs and baboons but was not effective in protecting infected rhesus monkey.[3]
In Kikwit during the 1995 outbreak, DRC, human convalescent plasma was used to treat 8 patients with proven Ebola disease, and only 1 patient died. Subsequent studies could not demonstrate survival benefit conferred by convalescent plasma products. The survival of these patient suggests that passive immunity may be of benefit in some patients.[3]
Four laboratory workers in Russia who had possible Ebola exposure were treated with a combination of a goat-derived anti-Ebola immunoglobulin plus recombinant human interferonalfa-2. One of these patients had a high-risk exposure and developed clinical evidence of Ebola virus infection. All 4 patients recovered.[3]
A recombinant human monoclonal antibody directed against the envelope glycoprotein (GP) of Ebola virus has been demonstrated to possess neutralizing activity. This Ebola virus−neutralizing antibody may be useful in vaccine development or as a passive prophylactic agent.[3] TKM-Ebola is a small interfering RNA compound, currently being tested in a Phase I clinical trial in humans. [4][5] Sarepta Therapeutics has completed a Phase I clinical trial with its phosphorodiamidate morpholino oligomers (PMO) protecting up to 80-100 percent of the non-human primates tested. [6]
ZMapp is a monoclonal antibody vaccine. The limited supply of the drug has been used to treat a small number of individuals infected with the Ebola virus. Although some individuals have recovered, the outcome is not considered statistically significant. [7] ZMapp has proved effective in a trial involving Rhesus macaque monkey.[4][8]
An antibody-based treatment for Ebola using synthesized fragments of the virus is developed by researchers in Thailand. Scientists from the WHO and NIH have offered to test the treatment against live Ebola virus, but there is still a great deal of development needed before human trials. [9]
DNA vaccines expressing either envelope GP or nucleo capsidprotein (NP) genes of Ebola virus have been demonstrated to induce protection in adult mice exposed to the virus. These vaccines were administered by coating gold beads with DNA expressing the genes for either GP or NP, and they were delivered by skin particle bombardment using a Powder Ject-XR genegun. Both vaccines induced measurable antibody responses detected by enzyme-linked immunosorbent assay(ELISA)and induced cytotoxic T-cell immunity.[3] Agents that may reduce mortality without directly effecting viral replication include activated proteinC and a recombinant nematode anticoagulant protein (NAP) that inhibits activated factor VII-tissue factor complex. NAP resulted in attenuation of the coagulopathy associated with decreased fibrinolysis and fibrin deposition with a resultant decrease in the severity of the systemic inflammatory response syndrome.[3]
In a rhesus macaque model of Ebola hemorrhagic fever, which carries a mortality approaching 100%, Geisbert et al administered recombinant nematode anticoagulant protein, a potent inhibitor of TF-initiated coagulation. One third of the monkey given the nematode anticoagulant protein survived a lethal dose of Ebola virus, whereas16 of the17 (94%) control animals died. This approach targeted the hemorrhagic disease component of the infection rather than the virus itself.[3]
The WHO has stated that transfusion of whole blood or purified serum from Ebola Survivors is the therapy with the greatest potential to be implemented immediately, although there is little information as to its efficacy.[10] September 2014, WHO issued an interim guideline for this therapy. [11] The blood serum from those who have survived an infection is currently being studied to see if it is an effective treatment. [12] Seven of eight people with Ebola survived after receiving a transfusion of blood donated by individuals who had previously survived the infection in 1999 outbreak in the Democratic Republic of the Congo. [13][14] This treatment, however, was started late in the disease meaning they may have already been recovering on their own and the rest of their care was better than usual.[14] Thus this potential treatment remains controversial. [15]
Intravenous antibodies appear to be protective in non-human primates who have been exposed to large doses of Ebola. [16] The World Health Organization has approved the use of convalescent serum and whole blood products to treat people with Ebola. [17]
Work continues on a vaccine for Ebola virus infection in primates. Other promising treatments depends on antisense technology. Both small interfering RNAs (siRNAs) and phosphorodiamidate morpholino oligomers (PMOs) targeting Ebola virus (EBOV) RNA polymerase L protein may prevent disease in nonhuman primates. [18][19]
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT editor-in-chief@pharmatutor.org
Subscribe to Pharmatutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE
PREVENTION AND CONTROL
WILDLIFE-TO-HUMAN TRANSMISSION
· Reducing the risk of wildlife-to-human transmission from contact with infected fruit bats or monkey/apes and the consumption of their raw meat. Animals should be handled with gloves and other appropriate protective clothing. Animal products (blood and meat) should be thoroughly cooked before consumption.[1]
HUMAN-TO-HUMAN TRANSMISSION
· Reducing the risk of human-to-human transmission from direct or close contact with people with Ebola symptoms, particularly with their bodily fluids. Gloves and appropriate personal protective equipment should be worn when taking care of ill patients at home. Regular hand washing is required after visiting patients in hospital, as well as after taking care of patients at home.[1]
OUTBREAK CONTAINMENT MEASURES
· Outbreak containment measures including prompt and safe burial of the dead, identifying people who may have been in contact with someone infected with Ebola, monitoring the health of contacts for 21 days, the importance of separating the healthy from the sick to prevent further spread, the importance of good hygiene and maintaining a clean environment.[1]
PREVENTION IN HEALTHCARE PERSONNEL GUIDANCE
· Prevention in healthcare personnel guidance from the CDC recommends that healthcare personnel who care for patients infected with Ebola virus (ie, physicians, nurses, other clinicians) wear personal protective equipment (PPE) that does not expose any skin recently (2014). This includes a surgical hood that covers the head and neck and a single-use full face shield, in addition to either a N95 respirator or powered air-purifying respirator instead of a mask. [1][3]
CONCLUSION
For controlling outbreaks community engagement is important. Control of outbreak depends on applying a package of interventions that is case management, surveillance and contact tracing, a good laboratory service, safe burials and social mobilisation. A response team is needed with integrated, prepared to execute selected functions, and equipped. An effective way to reduce human transmission raising awareness of risk factors for Ebola infection and protective measures that individuals can take. Effective containment of Ebola fever greatly depends on the isolation of the infected patients, and the careful adherence to barrier nursing techniques and universal precautions for the handling of infectious materials. Survival of infection is dependent on the ability of an individual to mount an effective immune response. The development of an effective Ebola virus vaccine is what future waits.
REFERENCES
1. Ebola virus disease., September 2014: https://www.who.int/mediacentre/factsheets/fs103/en/
2. Adrian M. C., Adeline M. N., Anthony S., Cam L. W., Heather S.; A Current Review of Ebola Virus: Pathogenesis, Clinical Presentation, and Diagnostic Assessment; Biol Res Nurs; 2003; (4):268
3. Ebola virus disease., October 2014: https://emedicine.medscape.com/article/216288-treatment#
4. Pollack., Andrew; Second drug is allowed for treatment of Ebola; The New York Times; 7 August 2014; Retrieved 8 August 2014.
5. Helen Branswell., Nancy Writebol; U.S. missionary, didn't get TKM-Ebola drug., Tekmira says, CBC News; Canadian Press; 3 August 2014.
6. Heald A.E., Iversen P.L., Saoud J.B., Sazani .P., Charleston J.S., Axtelle T., Wong M., Smith WB., Vutikullird A., Kaye E; Safety and pharmacokinetic profiles of phosphorodiamidate morpholino oligomers with activity against Ebola virus and Marburg virus: results of two single ascending dose studies; Antimicrobial Agents and Chemotherapy; 25 August 2014.
7. Kroll., David; How will we know if the Ebola drugs worked?; Forbes; 26 August 2014; Retrieved 1 October 2014.
8. Nathan Seppa; ZMapp drug fully protects monkey against Ebola; Science News- Society for Science & the Public; 29 August 2014; Retrieved 1 October 2014.
9. Ron Corben; Thai researchers claim breakthrough in Ebola treatment; Voice of America News; 3 October 2014
10. Blood transfusion named as priority treatment for Ebola; Nature; Retrieved 11 September 2014.
11. Use of convalescent whole blood or plasma collected from patients recovered from Ebola virus disease Empirical treatment during outbreaks; WHO; Retrieved 4 October
12. Statement on the WHO Consultation on potential Ebola therapies and vaccines; WHO; 5 September 2014; Retrieved 1 October 2014.
13. Mupapa K., Massamba M., Kibadi K., Kuvula K., Bwaka A., Kipasa M., Colebunders R., Muyembe-Tamfum J.J;Treatment of Ebola hemorrhagic fever with blood transfusions from convalescent patients: International Scientific and Technical Committee; J. Infect. Dis; 1999; 179 Suppl 1: S18–23.
14. Clark DV., Jahrling PB;, Lawler JV: Clinical management of filovirus-infected patients;; Viruses;, 2012; 4 (9) : 1668–86
15. Feldmann H., Geisbert T.W; Ebola haemorrhagic fever; Lancet; 5 March 2011: 849–62.
16. Saphire E.O; An update on the use of antibodies against the filoviruses; Immunotherapy; 2013; 5 (11) : 1221–33.
17. Gulland A; First Ebola treatment is approved by WHO; BMJ 349 ; Sep 08; 7 : 5539–5539.
18. Geisbert T.W., Lee A.C., Robbins M., Geisbert J.B., Honko A.N., Sood V., Johnson J.C., de Jong S., Tavakoli I., Judge A., Hensley L.E., Maclachlan I; Postexposure protection of non-human primates against a lethal Ebola virus challenge with RNA interference: A proof-of-concept study; The Lancet ; 29 May 2010 ; 375. 9729 : 1896–1905.
19. Warren TK., Warfield KL., Wells J., Swenson DL., Donner KS., Van Tongeren SA., Garza NL., Dong L., Mourich DV., Crumley S., Nichols DK., Iversen PL., Bavari S; Advanced antisense therapies for postexposure protection againstlethal filovirus infections; Nature Medicine; September 2010 ; 16(9) : 991–994.
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT editor-in-chief@pharmatutor.org
Subscribe to Pharmatutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE









