{ DOWNLOAD AS PDF }
About Authors:
Fiza Farheen*, Sudhir Bharadwaj
Department of Pharmaceutics, Shri Ram College of Pharmacy banmore,
Morena (M.P.) india
fizafarheen31@gmail.com
Abstract
Osmotic devices are most promising strategy based systems for controlled drug delivery. These are among the most reliable controlled drug delivery systems and could be employed as oral drug delivery systems or implant able devices. Osmotic pumps offer many advantages over other controlled drug delivery systems, i.e. they are easy to formulate and simple in operation, improve patient compliance with reduced dosing frequency and more consistent and prolonged therapeutic effect with uniform blood concentration. Drugs can be delivered in a controlled pattern over a long period of time by the process of osmosis. Osmotic pumps consist of an inner core containing drug and osmogens, coated with a semi permeable membrane. As the core absorbs water, it expands in volume, which pushes the drug solution out through the delivery ports. This article highlights the principle of osmosis, materials used for fabrication of pumps, types of pumps, advantages, disadvantages, and marketed products of this system.
INTRODUCTION
During the past three decades significant advances have been made in the area of novel drug delivery. This was in part due to the evolving discipline of biopharmaceutics, pharmacokinetics and pharmacodynamics. In a typical therapeutic regimen the drug dose and dosing interval are optimized to maintain drug concentration within the therapeutic window, thus ensuring efficacy while minimizing toxic effects. Survey indicated that dosing more than one or twice daily, greatly reduces patient compliance. So in recent year considerable attention has been focused on the development of novel drug delivery system and the main reason for this paradigm shift is relatively low development cost and time required for introducing a novel drug delivery system as compared to a new chemical entity.[1]
A number of design options are available to control or modulate the drug release from a dosage form. Majority of per oral dosage form fall in the category of matrix, reservoir or osmotic system. In matrix system, the drug is embedded in polymer matrix and the release takes place by partitioning of drug into the polymer matrix and the release medium. In contrast, reservoir systems have a drug core surrounded\coated by the rate controlling membrane. Osmotic systems utilize the principle of osmotic pressure for the delivery of drugs. Drug release from these systems is independent of pH and other physiological parameter to a large extent and it is possible to modulate the release characteristic by optimizing the properties of drug and system.[2] The oral osmotic pumps have certainly came a long way and the available products on this technology and number of patent granted in the last few years makes it presence felt in the market.[3] They are also known as gastro intestinal therapeutic system. Alza corporation of the USA was first to develop an oral osmotic pump and today also they are the leaders in this field with a technology named OROS. Osmotic drug delivery has come long way since Australian pharmacologist Rose and Nelson developed an implantable osmotic pump in 1955. Next quantum leap in osmotic dosage form came in 1972 when Theuwes invented elementary osmotic pump. After that many of have been invented which enable controlled delivery of almost all drugs.[4]
The bioavailability of drug from these formulations may vary significantly, depending on factors such as physico-chemical properties of the drug, presence of excipient, various physiological factors such as the presence or absence of food, pH of the GI tract, GI motility,etc.[5] To overcome this limitation of oral route is replied by parenteral route. This route offers the advantage of reduced dose, targeting of site and avoiding GI stability, hepatic by-pass of drug molecule.In the recent years, pharmaceutical research has led to the development of several novel drug delivery systems. The role of drug development is to take a therapeutically effective molecule with sub-optimal physicochemical and/or physiological properties and develop an optimized product that will still be therapeutically effective but with additional benefits such as.[6]
· Greater effectiveness in the treatment of chronic conditions,
· Sustained and consistent blood levels within the therapeutic window,
· Enhanced bioavailability,
· Reduced interpatient variability,
· Customized delivery profiles,
· Decreased dosing frequency,
· Improved patient compliance due to simplified dosing schedule,
· Reduced side effects,
OSMOSIS
Osmosis refers to the process of movement of solvent molecules from lowe concentration to higher concentration across a semi permeable membrane. Osmosis is the phenomenon that makes controlled drug delivery a reality. Osmotic pressure created due to imbibitions of fluid from external environment into the dosage form regulates the delivery of drug from osmotic device. Rate of drug delivery from osmotic pump is directly proportional to the osmotic pressure developed due to imbibitions of fluids by osmogen. Osmotic pressure is a colligative property of a solution in which the magnitude of osmotic pressure of the solution is independent on the number of discrete entities of solute present in the solution.(fig.1) Hence the release rate of drugs from osmotic dispensing devices is dependent on the solubility and molecular weight and activity coefficient of the solute (osmogent).[7,8]
PRINCIPLES OF OSMOSIS
The solvent membrane control delivery of agent from the osmotic system across the semi permeable membrane, which in turn drive the agent out. Water influx of osmotic pump can be describe as,
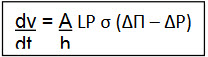
Where dv = water influx
dt
A = membrane area
h = membrane thickness
P = mechanical permeability
Δ? = osmotic pressure
ΔP = hydrostatic pressure difference between inside and outside the system
σ = describes the lickages of solute through the membrane.

The general expression for the solute delivery rate, dM / dt obtained by pumping through the orifice of the reservoir is given by,
Where C is concentration of solute if dispersed fluid.[9]
OSMOTICALLY CONTROLLED DRUG DELIVERY SYSTEMS
Osmotic pressure is used as driving force for these systems to release the drug in controlled manner. Osmotic drug delivery technique is the most interesting and widely acceptable among all other technologies used for the same. Intensive research has been carried out on osmotic systems and several patents are also published. Development of osmotic drug delivery systems was pioneered by Alza and it holds major number of the patents analyzed and also markets several products based on osmotic principle. These systems can be used for both route of administration i.e. oral and parenterals. Oral osmotic systems are known as gastro-intestinal therapeutic systems (GITS). Parenteral osmotic drug delivery includes implantable pumps.
ADVANTAGE OF OSMOTICALLY CONTROLLED DRUG DELIVERY SYSTEMS
· They typically give a zero order release profile after an initial lag.
· Deliveries may be delayed or pulsed if desired.
· Drug release is independent of gastric pH and hydrodynamic condition.
· They are well characterized and understood.
· The release mechanisms are not dependent on drug.
· A high degree of in-vitro and in-vivo correlation (ivivc) is obtained in osmotic systems.
· The rationale for this approach is that the presence of water in git is relatively constant, at least in terms of the amount required for activation and controlling osmotically base technologies.[10,11]
DISADVANTAGE OF OSMOTICALLY CONTROLLED DRUG DELIVERY
SYSTEM
* Expensive.
* Chances of toxicity due to dose duping.
* Rapid development of tolerance.
* Hypersensitivity reaction may occur.
* Integrity & consistency are difficult.
* Release of drug depends on
- Size of hole
- Thickness of membrane
- Surface area
- Composition of membrane[12]
CLASSIFICATION OF OSMOTIC DRUG DELIVERY SYSTEM
IMPLANTABLE
i. The Rose and Nelson Pump
ii. Higuchi Leeper Pump
iii. Higuchi Theuwes pump
iv. Implantable Mini osmotic pump
ORAL OSMOTIC PUMP
i. Single chamber osmotic pump:
Elementary osmotic pump
ii. Multi chamber osmotic pump:
Push pull osmotic pump
Osmotic pump with non-expanding second chamber
SPECIFIC TYPES
i. Controlled porosity osmotic pump
ii. Osmotic bursting osmotic pump
iii. Liquid OROS
iv. Delayed Delivery osmotic system
v. OROS-CT (colon targeting)
vi. Sandwiched oral therapeutic system
vii. Osmotic pump for insoluble drugs
viii. Monolithic osmotic system and OSMAT [13]
IMPLANTABLE
i. The Rose and Nelson Pump
In, 1955, two Australian physiologists reported the first osmotic pump. They were interested in delivery of drug to the gut of sheep and cattle. The pump consisted of three chambers a drug chamber with an orifice, a salt chamber with elastic diaphragm containing excess solid salt, and a water chamber. A semipermiable membrane separates the drug and water chamber. (fig.2) The difference in osmotic pressure across the membrane moves water from the water chamber in to the salt chamber. The volume of chamber increases because of this water flow, which distends the latex diaphragm separating the salt and drug chambers, thereby pumping drug out of the device.[14]
ii. Higuchi Leeper Pump
A number of variations of rose and nelson pump have been proposed by higuchi and leeper. In U.S patent these designs have been described. Alza corporation represented the first series of rose nelson pump . Which is illustrated in (fig.3) there is no water chamber in higuchi leeper pump and the device activate after penetration of water inside the device from the surrounding environment. A new patented series of modified higuchi leeper pump has issued in 1991. Higuchi leeper pump is widely used for veterinary use . this type of pump is either swallowed or implanted in the body of animal for delivery of antibiotic or growth hormone. higuchi leeper pump consist of rigid housing and semipermeable membrane. A layer of low melting waxy solid. Such as microcrystalline paraffin wax is used in place of elastic diaphragm to separate the drug and osmotic chamber. Recent modification in higuchi-leeper pump accommodated pulsatile drug delivery.[15]
iii. Higuchi -Theeuwes pump
In the early 1970s, Higuchi and Theeuwes developed another, even simpler variant of the Rose-Nelson pump. As with the Higuchi- Leeper pump, water to activate the osmotic action of the pump is obtained from the surrounding environment. In the Higuchi-Theeuwes device, however, the rigid housing is dispensed with and the membrane acts as the outer casing of the pump. This membrane is quite sturdy and is strong enough to withstand the pumping pressure developed inside the device. The device is loaded with the desired drug prior to use. When the device is placed in an aqueous environment, release of the drug follows a time course set by the salt used in the salt chamber and the permeability of the outer membrane casing. (fig.4) Most of the Higuchi-Theeuwes pumps use a dispersion of solid salt in a suitable carrier for the salt chamber of the device.[16]
iv. Implantable Mini osmotic pump
This is most advanced version in the category of implantable pumps developed by Alza corporation. It is composed of three concentric layers – the drug reservoir, the osmotic sleeve and the rate controlling semi permeable membrane. The addition component called flow moderator is inserted into the body of the osmotic pump after filling. The inner most compartment is drug reservoir which is surrounded by a osmotic sleeve, a cylinder containing of osmotic agent. (fig.5) The osmotic sleeve is covered by a semi permeable membrane when the system is placed in aqueous environment water enters the sleeve through the flow moderator. These pump are available with variety of delivery rates between 0.25 to 10 ml per hour and delivery duration between 1 day and 4 weeks.[17]
ORAL OSMOTIC PUMP
i. Single chamber osmotic pump:
* Elementary osmotic pump (EOP) :
Elementary osmotic pump was invented by Theeuwes in 1974 and it essentially contains an active agent having a suitable osmotic pressure, it is fabricated as a tablet coated with semi permeable membrane, usually cellulose acetate.[18,19] A small orifice is drilled through the membrane coating. When this coated tablet is exposed to an aqueous environment, the osmotic pressure of the soluble drug inside the tablet draws water through the semi permeable coating and a saturated aqueous solution of drug is formed inside the device. (fig.6) The membrane is non-extensible and the increase in volume due to inhibition of water raises the hydrostatic pressure inside the tablet, eventually leading to flow of saturated solution of active agent out of the device through a small orifice.[20]
The pump initially releases the drug at a rate given by equation;
dMt/dt = (dV/dt). Cs
Where,
dV/dt depicts the water flow into the tablet
Cs is the solubility of the agent inside the tablet.
iii. Multi chamber osmotic pump:
* Push pull osmotic pump :
Push pull osmotic pump is a modified EOP. Through, which it is possible to deliver both poorly water-soluble and highly water soluble drugs at a constant rate. This system resembles a standard bilayer coated tablet. One layer (depict as the uppe layer) contains drug in a formulation of polymeric, osmotic agent and other tablet excipients. This polymeric osmotic agent has the ability to form a suspension of drug in situ. When this tablet later imbibes water, the other layer contains osmotic and colorings agents, polymer and tablet excipients. This layer are formed and bonded together by tablet compression to form a single bilayer core. The tablet core is then coated with semipermeable membrane. After the coating has been applied, a small hole is drilled through the membrane by a laser or mechanical drill on the drug layer side of the tablet. (fig.7) When the system is placed in aqueous environment water is attracted into the tablet by an osmotic agent in both the layers. The osmotic attraction in the drug layer pulls water into the compartment to form in situ a suspension of drug. The osmotic agent in the non-drug layer simultaneously attract water into that compartment, causing it to expand volumetrically and the expansion of non-drug layer pushes the drug suspension out of delivery orifice.[21,22]
* Osmotic pump with non-expanding second chamber:
The second category of multi-chamber devices comprises system containing a non-expanding second chamber. This group can be divided into two sub groups, depending on the function of second chamber. In one category of these devices, the second chamber is used to dilute the drug solution leaving the devices. This is useful because in some cases if the drug leaves the oral osmotic devices a saturated solution, irritation of GI tract is a risk. This type of devices consist of two rigid chamber, the first chamber contains a biologically inert osmotic agent, such as sugar or a simple salt like sodium chloride, the second chamber contains the drug. (fig.8) In use water is drawn into both the chamber through the surrounding semi permeable membrane. The solution of osmotic agent formed in the first chamber then passes through the connecting hole to the drug chamber where it mixes with the drug solution before exiting through the micro porous membrane that form a part of wall surrounding the chamber. The device could be used to deliver relatively in soluble drugs.[23]
SPECIFIC TYPES
i. Controlled porosity osmotic pump (CPOP)
It is laser or micro driven orifice. When Controlled Porosity Osmotic Pump is placed in aqueous environment the water soluble component of coating dissolves and forms micropores in membrane and water diffuses inside the core through microporous membrane, setting up an osmotic gradint and thereby controlling the release of drug. Some of the pores forming additives that can be used are NaCl, KCl, and Urea. The rate of release from Controlled Porosity Osmotic Pump has been reported to be dependent on the coating thickness, level of soluble component in coating, solubility of drug in tablet core and osmotic pump diffuses across the membrane.(fig.9)
The rate of release from controlled porosity osmotic pump is dependent on
· Level of soluble component in coating
· Coating thickness
· Osmotic pressure across the membrane
· Solubility of drug in tablet core
Drug release from the whole surface of device rather than from a single hole which may reduce stomach irritation problem. Hole is produce by the coating procedure hence complicated laser drilling is not required. Citric acid is use as pore forming agent in Chitosan based colon specific pumps.[24]
ii. Osmotic bursting osmotic pump
In this system delivery orifice is absent and size may be smaller. When it is placed in an aqueous environment, water is imbibed and hydraulic pressure is built up inside until the wall rupture and the content are released to the environment.[25]
iii. Liquid OROS
Liquid OROS are designed to deliver drugs as liquid formulations and combine the benefits of extended release with high bioavailability.
They are of three types:
· L OROS hard cap,
· L OROS soft cap,
· Delayed liquid bolus delivery system.
Each of these systems includes a liquid drug layer, an osmotic engine or push layer and a semi permeable membrane coating. When the system is in contact with the aqueous environment water permeates across the rate controlling membrane and activate the osmotic layer. The expansion of the osmotic layer results in the development of hydrostatic pressure inside the system, thereby forcing the liquid formulation to be delivered from the delivery orifice. (fig.10) Whereas L OROS hardcap or softcap systems are designed to provide continuous drug delivery, the L OROS delayed liquid bolus drug delivery system is designed to deliver a pulse of liquid drug. The delayed liquid bolus delivery system comprises three layers: a placebo delay layer, a liquid drug layer and an osmotic engine, all surrounded by rate controlling semi permeable membrane. The delivery orifice is drilled on the placebo layer end of the capsule shaped device. When the osmotic engine is expands, the placebo is released first, delaying release of the drug layer. Drug release can be delayed from I to 10 hour, depending on the permeability of the rate controlling membrane and thickness of the placebo layer.[26]
iv. Delayed Delivery osmotic system
This device consists of two chambers, the first contains the drug and an exit port, and the second contains an osmotic engine. A layer of wax like material separates the two section. To assemble the delivery device, the desired active agent is placed into one of the sections by manual or automated fill mechanism. The bilayer tablet with the osmotic engine is placed into a completed cap part of the capsule with the convex osmotic layer pointed in to the closed end of the cap and the barrier into the closed end of the cap and the barrier layer exposed towards the cap opening. The open end of the filled vessel is fitted inside the open end of the cap, and the two pieces are compressed together until the cap, osmotic bilayer tablet and vessel fit together tightly. (fig.11) As fluid is imbibed the housing of the dispensing device, the osmotic engine expand and exerts pressure on the slidable connected first and second wall sections. During the delay period the volume of reservoir containing the active agent is kept constant, therefore a negligible pressure gradient exists between the environment of use and interior of the reservoir. As a result, the net flow of environmental fluid driven by the pressure enter the reservoir is minimal and consequently no agent is delivered for the period.[27]
v. OROS-CT (colon targeting)
OROS-CT is used as a once or twice a day formulation for targeted delivery of drugs to the colon. The OROS-CT can be a single osmotic agent or it can be comprised of as many as five to six push pull osmotic unit filled in a hard gelatin capsule. After coming in contact with the gastric fluids, gelatin capsule dissolved and the enteric coating prevents entry of fluids from stomach to the system as the system enters into the small intestine the enteric coating dissolves and water is imbibed into the core thereby causing the push compartment to swell. At the same time flowable gel is formed in the drug compartment, which is pushed out of the orifice at a rate, which is precisely controlled, by the rate of water transport across the semi permeable membrane.[28]
vi. Sandwiched oral therapeutic system
It is composed of polymeric push layer sandwiched between two drug layers with two delivery orifices. When placed in the aqueous environment the middle push layer containing the swelling agent’s swells and the drug is released from the two orifices situated on opposite sides of the tablet and thus SOTS can be suitable for drugs prone to cause local irritation of the gastric mucosa.[29]
vii. Osmotic pump for insoluble drugs
The device concerns an osmotic agent for dispensing beneficial active agent that has poor solubility in water. The core of the system comprises a beneficial amount of a substantially water- insoluble active agent, which is lipid soluble or lipid- wettable; a sufficient amount of water insoluble lipid carrier, which is liquid at the temperature of use to dissolve or suspend the drug and agent to ensure the release of the lipid carrier of the drug from the pump. The water insoluble wall is micro porous and is wetted by lipid carrier. The device is prepared by first dissolving the drug of interest in the lipid vehicle. The osmogent (Sodium chloride) is dispersed in the melted lipid and then quenched-cool to form a lump that are broken and made into tablet. The micro porous is coated at a moderate flow of unheated ambient air.[30]
viii. Monolithic osmotic system and OSMAT
It constitutes a simple dispersion of water-soluble agent in polymer matrix. When the system comes in contact in with the aqueous environment. Water imbibtion by the active agents takes place rupturing the polymer matrix capsule surrounding the drug. Thus liberating it to the outside environment. Initially this process occurs at the outer environment of the polymeric matrix, but gradually proceeds towards the interior of then matrix in a serial fashion. However this system fails if more then 20 –30 volumes per liter of the active agents are incorporated in to the device as above this level, significant contribution from the simple leaching of the substance take place.[31]
Osmat It is a novel osmotically driven matrix system, which utilizes the hydrophilic polymers to swell, and gel in aqueous medium forming a semipermiable membrane in-situ releases from such a matrix system containing an osmogen could, therefore be modulated by the osmotic phenomenon. Osmat thus judiciously combines both matrix osmotic characteristics resulting in a quantum improvement in drug delivery from swellable matrix system. Osmat produces controlled drug release with adequate delivery rates in an agitation in dependent manner. Thus osmat represents simple, versatile, and easy to fabricate osmotically driven controlled drug delivery system based upon low cost technology.[32]
BASIC COMPONENT OF OSMOTICALLY DRUG DELIVERY SYSTEM
· Drug
· Osmotic agent
· Semi permeable membrane
· Pore forming agents
· Plasticizers
Drugs:
Characteristics of drug candidate for osmotically controlled drug delivery
· Short biological half-life (2-6h)
· Highly potent drug
· Required for prolonged treatment e.g. Nifedipine, Glipizide, Virapamil, Famotidine.
Osmotic agent:
Osmogent are essential ingredient of osmotic pump, usually is ionic compounds consisting of either inorganic salts or hydrophilic polymers and carbohydrates. Generally combination of osmogent is used to achieve desired osmotic pressure within the device. Some of the osmotic agents that can be used for such systems are classified below. Different type of osmogents can be used for such systems arecategorized as.
· Inorganic water-soluble osmogents:Magnesium sulphate, Sodium chloride, Sodium sulphate, potassium chloride, sodium bicarbonate.
· Organic polymer osmogents:sodium carboxy methyl cellulose, hydroxy propyl methl cellulose, Hydroxyethylmethylcellulose, Methylcellulose, Polethylene oxide, polyvinyl pyrollidine.[33,34]
Semi permeable membrane:
The semi permeable membrane should be a stable both to the outer inner environment of the device. The membrane must be sufficiently rigid so as to retain its dimensional integrity during the operational lifetime of the device. The membrane should also be relatively impermeable to the contents of dispenser so that osmogent is not lost by diffusion across the membrane finally, the membrane must be biocompatible.
Ideal Property of Semi Permeable Membrane:
The Semi Permeable Membrane must meet some performance criteria:
· The material must possess sufficient wet strength (-105) and wet modulus so as to retain its dimensional integrity during the operational lifetime of the device.
· The membrane exhibit sufficient water permeability so as to retain water flux rate in the desired range. The water vapor transmission rates can be used to estimate water flux rates
· The membrane should also be biocompatible
· Rigid and non-swelling
·Should be sufficient thick to withstand the pressure within the device.[35]
Pore forming agents:
These agents are particularly used in the development of pump for poorly water soluble drugs and in controlled porosity tablets. These agents cause the formation of micro porous membrane. The micro porous wall may be formed by the leaching of water soluble substrate from membrane leaving a micro porous structure. The pore former can be organic or inorganic and solid or liquid in nature. Some examples of pore former are given below. Alkaline earth metal salts Sodium chloride, potassium chloride, sodium bromide, potassium phosphate. Carbohydrate such as sucrose, lactose, glucose, mannitol, fructose etc is used as pore forming agent.[36]
Plasticizers:
Different types and amount of plasticizers used in coating membrane. They can change visco-elastic behavior of polymers and these changes may affect the permeability of the polymeric films. Some of the plasticizers used are as below:
· Polyethylene glycols
· Ethylene glycol monoacetate; and diacetate- for low permeability
· Tri ethyl citrate
· Diethyl tartarate or Diacetin- for more permeable films.[37]
FACTORS AFFECTING DRUG RELEASE RATE
Factors affecting the release rate of medicament from osmotic drug delivery system are,
· Solubility
· Osmotic pressure
· Delivery orifice
· Membrane type
Solubility:
Osmotic pumps are suitable for delivery of drugs having intermediate water solubility. It has been reported that in case of high water soluble drugs, meaningful release rates may not be obtained using elementary osmotic pump (EOP) or controlled-porosity osmotic pump (CPOP). This is because the kinetics of osmotic drug release is directly related to solubility of drug within the core. APIs for osmotic delivery should have water solubility in the desired range to get optimize drug release. However, by modulating the solubility of these drugs within the core, effective release patterns may be obtained for the drugs, which might otherwise appear to be poor candidate for osmotic delivery.[38]
Osmotic Pressure:
Drug selected as candidate for formulation as osmotic system, should possess osmotic pressure. The release rate of drug from osmotic system is directly proportional to the osmotic pressure of the core formulation. If the drug does not possess sufficient osmotic pressure, an osmogent like sodium control the release of drug from the osmotic system.
In order to achieve optimized and constant Osmotic Pressure in compartment Osmotic agent must be added to formulation.(table no.1)
So varying the osmogen vary osmotic pressure and hence drug release. Osmogen are classified as inorganic and organic osmogens.(table no.2)
Delivery orifice:
· To achieve an optimal zero order delivery profile, the cross sectional area of the orifice must be smaller than a maximum size to minimize drug delivery by diffusion through the orifice
· Furthermore, the area must be sufficiently large, above a minimum size to minimize hydrostatic pressure build up in the system
· The typical orifice size in osmotic pumps ranges from 600µ to 1 mm.
Methods to create a delivery orifice:
· Mechanical drill
· Laser drilling : CO2 laser beam
· Use of modified punches
· Use of pore formers : used in controlled porosity osmotic pump
Membrane type:
Drug release from osmotic system is largly independent of pH and agitational intensity of GIT.
Example are Cellulose Ester, Cellulose Triacetate, Cellulose Propionate, Cellulose Acetate Butyrate, Ester, Ethyl Cellulose and Eudragits.
Among above Cellulose Acetate Butyrate is most commanly used since of its,
· High water permeability,
· Permeability can be adjusted by varying the degree of acetylation of polymer and also by increasing plastisizer concentration,
· Flux enhancer and,
· Superior drying property so advantageneous to thermolabile drugs.[39]
MARKETED PRODUCTS
List of Osmotic drug delivery systems in market with their dosage and use (table .3).[40]
CONCLUSION
In recent years, controlled drug delivery system (CDDS) has been recognized as an attractive niche for the pharmaceutical and health industry. Among various CDDS, osmotic pumps have matured from their use with laboratory animals to the most reliable controlled release systems for human. Osmotically controlled drug delivery system use osmotic pressure for controlled delivery of active agents. Drug delivery from these systems, to a large extent, is independent tract. Because of their unique advantages over other types of dosage forms, osmotic pumps from a class of their own among the various drug delivery technologies, and a variety of products based on this technology are available on the market.
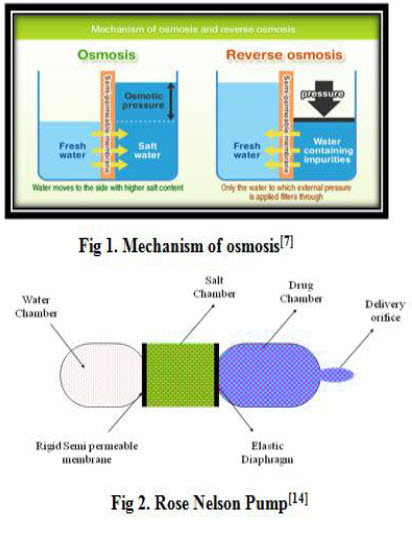
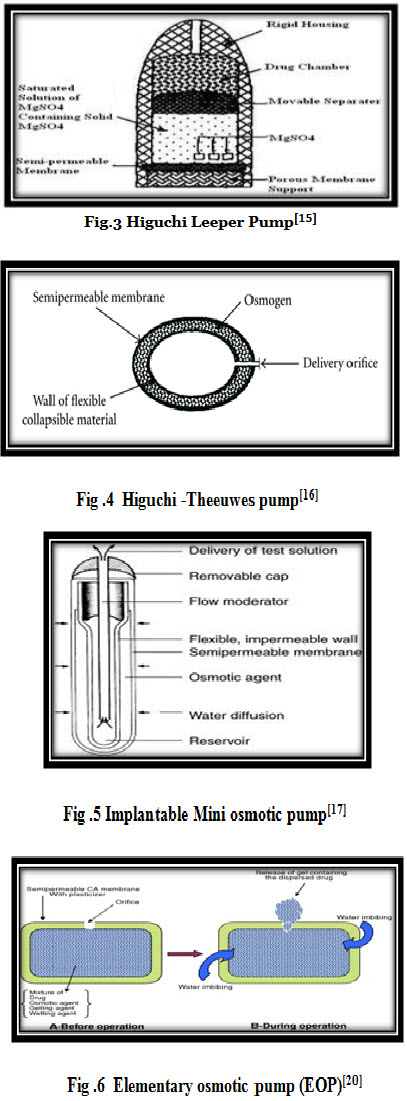
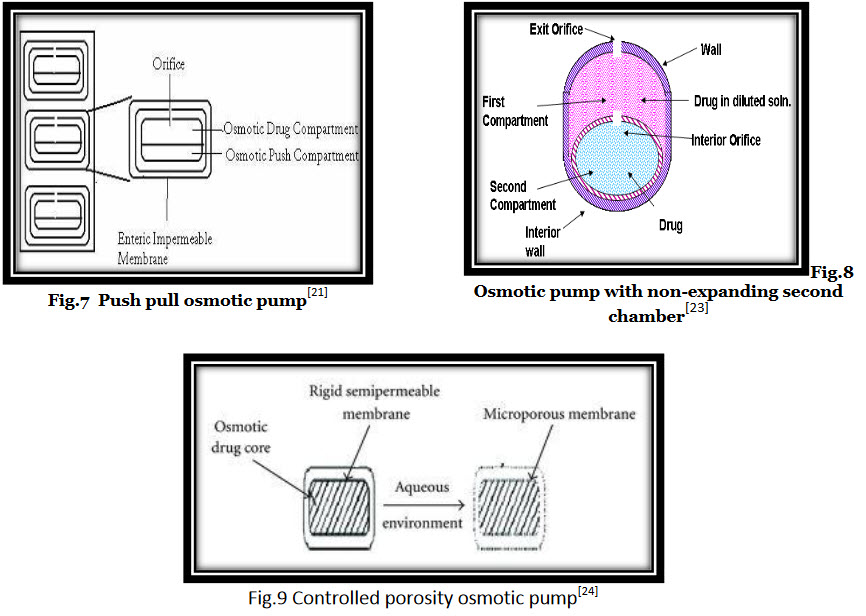
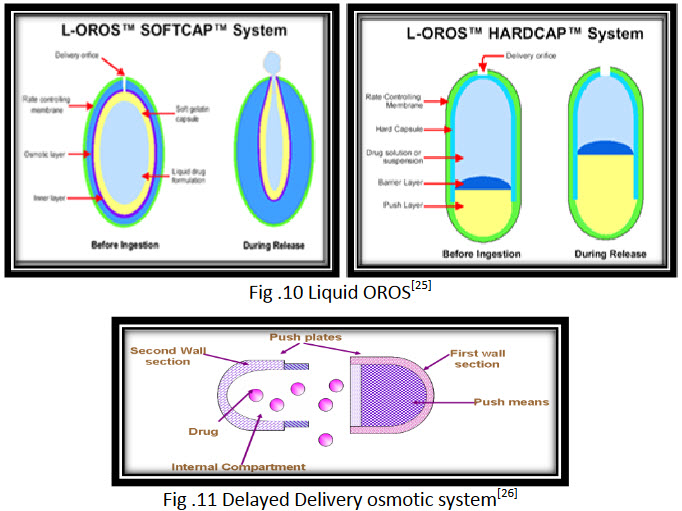
Table no. 1
|
S.NO. |
OSMOGEN |
OSMOTIC PRESSURE |
|
1. |
Nacl |
356 |
|
2. |
Fructose |
355 |
|
3. |
Kcl |
345 |
|
4. |
Sucrose |
150 |
|
5. |
Xylitol |
104 |
|
6. |
Sorbitol |
84 |
|
7. |
Dextrose |
82 |
|
8. |
Citric acid |
69 |
|
9. |
Tartaric acid |
67 |
|
10. |
Mannitol |
38 |
Table no. 2
|
S.NO. |
COMBINED OSMOGEN |
OSMOTIC PRESSURE |
|
1. |
Lactose - Fructose |
500 |
|
2. |
Dextrose – Fructose |
450 |
|
3. |
Sucrose – Fructose |
430 |
|
4. |
Mannose – Fructose |
415 |
|
5. |
Lactose – sucrose |
250 |
|
6. |
Mannital – sucrose |
170 |
|
7. |
Mannose – Lactose |
225 |
|
8. |
Lactose - Dextrose |
225 |
|
9. |
Dextrose – Sucrose |
190 |
|
10. |
Mannitol – Lactose |
130 |
Table no. 3
|
Sr.No. |
Trade Name |
Active ingredient |
Design system |
Dose |
Use |
|
1. |
Alpress LP |
Prazosin
|
Push –Pull |
2.5 – 5 mg |
For the treatment of Hypertension. |
|
2. |
Acutrim |
Phenylpropanolamine |
Elementary pump |
75 mg |
allergies, hay fever, sinus irritation, and the common cold. |
|
3. |
Cardura XL |
Doxazosin |
Push –Pull |
4, 8 mg |
For the treatment of Hypertension. |
|
4. |
Covera HS |
Verapamil |
Push -Pull with time delay |
180, 240 mg |
For the management of Hypertension and angina. |
|
5. |
Efidac 24 |
Chlorpheniramine Maleate |
Elementary Pump |
4 mg IR, 12 mg CR |
Chlorpheniramine is an antihistamine. Chlorpheniramine is used to treat sneezing; runny nose; itching, watery eyes; hives; rashes; itching; and other symptoms of allergies and the common cold |
REFERENCES
1.Verma. R K, Garg S; Current status of drug delivery technologies and future directions. Pharm Technol. 2001; 25:1-14.
2.Theeuwes F, Swanson D R, Guitttard G, Ayer A, Khanna S; Osmotic delivery systems for the β-adrenoceptor antagonists Metoprolol and Oxprenolol: Design and evaluation of systems for once-daily administration. Br. J Clin Pharmacology. 1985; 19, 69-76.
3.Santus G, Baker R W; Osmotic drug delivery: A review of the patent literature. J Control Release, 1995, 35, 1-21.
4.Verma R K, Mishra B, Garg S; Osmotically controlled oral drug delivery. Drug Dev Ind Pharm. 2000; 26(7):695-708.
5. Prescott L F; The need for improved drug delivery in clinical practice, In: Novel Drug Delivery and Its Therapeutic application, John Wiley and Sons, West Susset, UK, 1989, 1-11.
6.Dr Bhatt P P; Osmotic drug delivery systems for poorly water soluble drugs Pharmaventures Ltd., Oxford, UK, 2004; 26-29
7.Pfefer, W E P; Osmotishe Umtersuchen, Leipzig. 1877; 232
8.Li X and Jasti B R; Osmotic controlled drug delivery systems, In: Design of controlled release of drug delivery systems, McGraw Hill, 2006; 203-229.
9. Rastogi S K, Vaya N, Mishra B; Osmotic pump: A novel concept in rate controlled oral drug delivery. Eastern pharmacist. 1995; (38):79-82.
10.Fix J, In: Encyclopedia of controlled drug delivery, Edmathiowitz, vol-2, John Wiley and sons, Inc 700.
11. Kaushal A M and Garg S; An update on osmotic drug delivery patents, Pharm Tech. Aug 2003; 38-44.
12.Lachman L., Liberman H. A., Kanig J. L., The theory and practise of industrial pharmacy. 2nd Edition 1991, Varghese publishing house, P=455
13.Stuti G, Ravindra PS, Rohitashva S. International Journal of Comprehensive Pharmacy 2011; 6: 1.
14.NK Jain. Advances in controlled and novel drug delivery, CBS Publisher & distributer, first adition. Page No.20
15.Theuwes F., Swanson D. R., Guitttard G. and Ayer A. 1985. Br. J.Clin. Pharmacology, 1985, 19, 69-76.
16.Parashar Bharat, Maurya Brajesh, Yadav Virendra, Sharma Love. “A review on osmotically regulated devices.” The pharma innovation.2012; 1(4):48-56.
17.Gadwal P, Rudrawal P. International Journal of Pharmacy & Life Sciences 2010; 1(6): 302-312.
18.Ouyang D, Nie S, Li W, Guo H, Liu H, Pan W. J Pharm Pharmacol 2005; (57): 817-820.
19.Tanmoy Ghosh, Amitava Ghosh. Journal of Applied Pharmaceutical Science 2011; 1 (2): 38-49
20.F Theeuwes. Journal of Pharmaceutical Sciences 1975; (64): 1987– 1991.
21.Mane Suraj S., Kamble Meghana S., Chaudhari Pravin D., Bhosale Ashok V., “A review on oral osmotically controlled drug delivery system.” International J. Of universal pharmacy and life sciences (IJUPLS).2012; 2(2):19-36
22.Parma NS, Vyas SK, NK Jain. In: Advanced in controlled and novel drug delivery. CBS publisher, 28-29.
23.Dong L, Shafi K, Wan J, Wong; In: Proceeding of the international symposium on controlled release of bioactive material, paris (july) 2003.
24.Cortese R and Theeuwes F; Osmotic device with hydro gel driving member. US Patent 4327725; 1982.
25.Javad Shokri, Parinaz Ahmadi, Parisa Rashidi, Mahbobeh Shahsavari, Ali Rajabi-Siahboomi, Ali Nokhodchi. European Journal of Pharmaceutics and Biopharmaceutics 2008; 68: 289–297.
26.Gaylen Zentner M, Gerald S Rork and Kenneth J Himmerstein; The Controlled Porosity Osmotic Pump. J Control Rel. 1985: 1: 269-282.
27.Kaushal, Aditya and Garg Sanjay. Pharma Technol 2003; 27: 39.
28.Theeuwes F, Wong P S L, Burkoth T L and Fox D A; Osmotic systems for colon-targeted drug delivery. In: Colonic drug absorption and metabolism. Marcel Dekker, NY, 1993; 137-158.
29.Longxiao Liu, Jeong Ku, Gilson Khang, Bong Lee, John M Rhee, Hai Bang Lee. Journal of Controlled Release 2000; 68: 145–156.
30.Patel Harnish, Patel Upendra, Kadikar Hiren, Bhimani Bhavin, Daslaniya Dhiren, Patel Ghanshyam. “A review on osmotic drug delivery system.” International research J. Of pharmacy (IRJP).2012; 3(4):88-94.
31.Wong P S I, Barclay B, Deters J C and Theeuwes F; Osmotic device with dual thermodynamic activity. US Patent 4612008; 1986.
32.Ghosh Tanmoy, Ghosh Amitava. “A review on drug delivery through osmotic systems.” J. Of applied pharmaceutical science (JAPS).2011; 01(02):38-49.
33. Sudeesh E: Formulation development and optimization of controlled porosity osmotic pump tablets of diclofenac sodium. International Journal of Pharmacy and Pharmaceutical Sciences 2011; 3(1): 80-87.
34.Prakash RB, Geetha M, Purushothama N, Utpal S: Optimization and Development of Swellable Controlled Porosity Osmotic Pump Tablet for Theophylline. Tropical Journal of Pharmaceutical Research 2009; 8(3) 247-255.
35.Nikam PH, Kareparamban, Jadhav AP, Kadam VJ., “A review on osmotic pump: A Reliable drug delivery system. Research J. Of pharmaceutical, Biological Chemical Sciences (RJPBCS).2012; 3(3):478- 493.
36.Mahalaxmi R, Phanidhar S, Ravikumar, Atin K, Pritam K, Narkhede: Enhancement of Dissolution of Glipizide from Controlled Porosity Osmotic Pump Using a Wicking Agent and a Solubilizing Agent International Journal of PharmTech Research 2009;13: 705
37.GM Zentner, GS Rork, KJ Himmelstein. US patent 4, 1990,968,507
38.Gaebler F; Laser drilling enables advanced drug delivery systems, Coherent article for Pharmaceutical Manufacturing, Jan-2007, 1-7. 39.Speers M, Bonnano C. Economic aspects of controlled drug delivery. In: Mathiowitz E, ed. Encyclopedia of Controlled Drug Delivery. New York, NY: Wiley; 1999:341Y347
40.Eckenhoff, Yum SI: The osmotic pump: novel research tool for optimizing drug regimen. Biomaterials, 1981; 2:89-97
REFERENCE ID: PHARMATUTOR-ART-2166
|
PharmaTutor (ISSN: 2347 - 7881) Volume 2, Issue 5 Received On: 17/03/2014; Accepted On: 26/03/2014; Published On: 01/05/2014 How to cite this article: F Farheen, S Bharadwaj; A Review on Osmotically Regulated Systems; PharmaTutor; 2014; 2(5); 51-64 |
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT articles@pharmatutor.org
Subscribe to Pharmatutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE









