{ DOWNLOAD AS PDF }
About Authors:
*Dilipkumar.J.P, Agliandeshwari.D
Rajiv Gandhi University of Health Sciences
Bangalore, India
*dilipkumar9447@gmail.com
INTRODUCTION
Natural products, especially those derived from plants, have been used to help mankind sustain its health since the dawn of medicine. Over the past century, the phytochemicals in plants have been a pivotal pipeline for pharmaceutical discovery. The importance of the active ingredients of plants in agriculture and medicine has stimulated significant scientific interest in the biological activities of these substances1. Despite these studies, a restricted range of plant species has experienced detailed scientific inspection, and our knowledge is comparatively insufficient concerning their potential role in nature. Hence, the attainment of a reasonable perception of natural products necessitates comprehensive investigations on the biological activities of these plants and their key phytochemicals2. In a pharmaceutical landscape, plants with a long history of use in ethno medicine are a rich source of active phytoconstituents that provide medicinal or health benefits against various ailments and diseases. One such plant with extensive traditional use is Annona muricata. In this review, we describe the botany, distribution and ethnomedicinal uses of this plant, and we summarize the phytochemistry, biological activities and possible mechanisms of A. muricata bioactivities.
[adsense:336x280:8701650588]
Reference Id: PHARMATUTOR-ART-2528
|
PharmaTutor (Print-ISSN: 2394 - 6679; e-ISSN: 2347 - 7881) Volume 5, Issue 10 Received On: 14/06/2017; Accepted On: 15/06/2017; Published On: 01/10/2017 How to cite this article: Dilipkumar JP, Agliandeshwari D; Preparation & evaluation of Annona Muricata extract against cancer cells with modified release; PharmaTutor; 2017; 5(10); 63-106 |
Botanical Description and Distribution
A. muricata L., commonly known as soursop, graviola, guanabana, paw-paw and sirsak, is a member of the Annonaceae family comprising approximately 130 genera and 2300 species3,4. A. muricata is native to the warmest tropical areas in South and North America and is now widely distributed throughout tropical and subtropical parts of the world, including India, Malaysia and Nigeria5. A. muricata is an evergreen, terrestrial, erect tree reaching 5–8 m in height and features an open, roundish canopy with large, glossy, dark green leaves. The edible fruits of the tree are large, heart-shaped and green in color, and the diameter varies between 15 and 20 cm (Figure 1)6.

Figure 1.1: (A) Annona muricata L.; the appearance of the (B) leaves; (C) flowers and (D) fruits.
Ethnomedicinal Uses
All portions of the A. muricata tree, similar to other Annona species, including A. squamosa and A. reticulata are extensively used as traditional medicines against an array of human ailments and diseases, especially cancer and parasitic infections. The fruit is used as natural medicine for arthritic pain, neuralgia, arthritis, diarrhea, dysentery, fever, malaria, parasites, rheumatism, skin rushes and worms, and it is also eaten to elevate a mother’s milk after childbirth. The leaves are employed to treat cystitis, diabetes, headaches and insomnia. Moreover, internal administration of the leaf’s decoction is believed to exhibit anti-rheumatic and neuralgic effects, whereas the cooked leaves are topically used to treat abscesses and rheumatism3,5,7. The crushed seeds are believed to have anthelmintic activities against external and internal worms and parasites. In tropical Africa, the plant is used as an astringent, insecticide and piscicide agent and to treat coughs, pain and skin diseases. In India, the fruit and flower are employed as remedies against catarrh, while the root-bark and leaves are believed to have antiphlogistic and anthelmintic activities8,9. In Malaysia, the crushed leaf mixture of A. muricata together with A. squamosa and Hibiscus rosa-sinensis is used as a juice on the head to protect against fainting10. In South America and tropical Africa, including Nigeria, leaves of A. muricata are deployed as an ethnomedicine against tumors and cancer8. In addition, the anti-inflammatory, hypoglycemic, sedative, smooth muscle relaxant, hypotensive and antispasmodic effects are also attributed to the leaves, barks and roots of A. muricata3,5. In addition to ethnomedicinal uses, the fruits are widely employed for the preparation of beverages, candy, ice creams, shakes and syrups11,12.
Phytochemistry
Extensive phytochemical evaluations on different parts of the A. muricata plant have shown the presence of various phytoconstituents and compounds, including alkaloids (ALKs)4,13, megastigmanes (MGs)14 flavonol triglycosides (FTGs)15, phenolics (PLs)16, cyclopeptides (CPs) and essential oils (Table 1, Figure 2)17,18. However, Annona species, including A. muricata, have been shown to be a generally rich source of annonaceous acetogenin compounds (AGEs)19. The presence of different major minerals such as K, Ca, Na, Cu, Fe and Mg suggest that regular consumption of the A. muricata fruit can help provide essential nutrients and elements to the human body20.
Table No. 1.1: Chemical compounds isolated from Annona muricata. ALK: alkaloid; AGE: annonaceous acetogenin; MG: megastigmane; FTG: flavonol triglycoside; PL: phenolic; CP: cyclopeptide.
|
Plant Part |
Compound |
Class |
Biological Activity |
References |
|---|---|---|---|---|
|
Fruits |
annonaine |
ALK |
anti-depressive |
[21,22] |
|
Fruits |
nornuciferine |
ALK |
anti-depressive |
[21,22] |
|
Fruits |
asimilobine |
ALK |
anti-depressive |
[21,22] |
|
Fruits |
epomusenin-A |
AGE |
- |
[23] |
|
Fruits |
epomusenin-B |
AGE |
- |
[23] |
|
Fruits |
epomurinin-A |
AGE |
- |
[23] |
|
Fruits |
epomurinin-B |
AGE |
- |
[23] |
|
Fruits |
cis-annoreticuin |
AGE |
- |
[24] |
|
Fruits |
muricin J |
AGE |
toxicity against prostate PC-3 cancer cells |
[25] |
|
Fruits |
muricin K |
AGE |
toxicity against prostate PC-3 cancer cells |
[25] |
|
Fruits |
muricin L |
AGE |
toxicity against prostate PC-3 cancer cells |
[25] |
|
Fruits |
cinnamic acid derivative |
PL |
- |
[16] |
|
Fruits |
coumaric acid hexose |
PL |
- |
[16] |
|
Fruits |
5-caffeoylquinic acid |
PL |
- |
[16] |
|
Fruits |
dihydrokaempferol-hexoside |
PL |
- |
[16] |
|
Fruits |
p-coumaric acid |
PL |
- |
[16] |
|
Fruits |
caffeic acid derivative |
PL |
- |
[16] |
|
Fruits |
dicaffeoylquinic acid |
PL |
- |
[16] |
|
Fruits |
feruloylglycoside |
PL |
- |
[16] |
|
Fruits |
4-feruloyl-5-caffeoylquinic acid |
PL |
- |
[16] |
|
Fruits |
p-coumaric acid methyl ester |
PL |
- |
[16] |
|
Leaves, Pericarp |
annomuricin A |
AGE |
toxicity against brine shrimp, lung A549, breast MCF-7 and colon HT-29 cancer cells |
[12,26] |
|
Leaves |
annomuricin B |
AGE |
toxicity against brine shrimp, lung A549, breast MCF-7 and colon HT-29 cancer cells |
[12] |
|
Leaves |
annomuricin C |
AGE |
toxicity against brine shrimp, lung A549, breast MCF-7 and colon HT-29 cancer cells |
[27] |
|
Leaves |
annomuricin E |
AGE |
toxicity against pancreatic MIA PaCa-2 and colon HT-29 cancer cells |
[28] |
|
Leaves |
annomutacin |
AGE |
toxicity against lung A549 cancer cells |
[29] |
|
Leaves |
(2,4-cis)-10R-annonacin-A-one |
AGE |
toxicity against lung A549 cancer cells |
[29] |
|
Leaves |
(2,4-trans)-10R-annonacin-A-one |
AGE |
toxicity against lung A549 cancer cells |
[29] |
|
Leaves |
annohexocin |
AGE |
toxicity against brine shrimp and different cancer cells |
[30] |
|
Leaves |
muricapentocin |
AGE |
toxicity against pancreatic MIA PaCa-2 and colon HT-29 cancer cells |
[28] |
|
Leaves |
(2,4-cis)-isoannonacin |
AGE |
- |
[31] |
|
Leaves, Seeds |
(2,4-trans)-isoannonacin |
AGE |
- |
[31,32] |
|
Leaves |
muricatocin A |
AGE |
toxicity against lung A549 cancer cells |
[31] |
|
Leaves |
muricatocin B |
AGE |
toxicity against lung A549 cancer cells |
[31] |
|
Leaves |
muricatocin C |
AGE |
toxicity against brine shrimp, lung A549, breast MCF-7 and colon HT-29 cancer cells |
[27] |
|
Leaves, Seeds |
gigantetronenin |
AGE |
- |
[27,32] |
|
Leaves, Seeds, Pericarp |
annonacin A |
AGE |
- |
[26,31,33] |
|
Leaves |
annopentocin A |
AGE |
toxicity against pancreatic MIA PaCa-2 cancer cells |
[34] |
|
Leaves |
annopentocin B |
AGE |
toxicity against lung A549 cancer cells |
[34] |
|
Leaves |
annopentocin C |
AGE |
toxicity against lung A549 cancer cells |
[34] |
|
Leaves |
cis-annomuricin-D-one |
AGE |
toxicity against lung A549, colon HT-29 and pancreatic MIA PaCa-2 cancer cells |
[34] |
|
Leaves |
trans-annomuricin-D-one |
AGE |
toxicity against lung A549, colon HT-29 and pancreatic MIA PaCa-2 cancer cells |
[34] |
|
Leaves |
murihexocin A |
AGE |
toxicity against different cancer cells |
[35] |
|
Leaves |
murihexocin B |
AGE |
toxicity against different cancer cells |
[35] |
|
Leaves |
murihexocin C |
AGE |
toxicity against different cancer cells |
[36] |
|
Leaves |
muricoreacin |
AGE |
toxicity against different cancer cells |
[36] |
|
Leaves |
cis-corossolone |
AGE |
toxicity against human hepatoma cells |
[37] |
|
Leaves |
annocatalin |
AGE |
toxicity against human hepatoma cells |
[37] |
|
Leaves |
annocatacin B |
AGE |
toxicity against human hepatoma cells |
[38] |
|
Leaves |
anonaine |
ALK |
neurotoxic |
[39,40] |
|
Leaves |
isolaureline |
ALK |
- |
[39] |
|
Leaves |
xylopine |
ALK |
- |
[39] |
|
Leaves |
Quercetin 3-O-α-rhamnosyl-(1→6)-β-sophoroside |
FTG |
- |
[15] |
|
Leaves |
gallic acid |
FTG |
- |
[15] |
|
Leaves |
epicatechine |
FTG |
- |
[15] |
|
Leaves |
quercetin 3-O-rutinosid |
FTG |
- |
[15] |
|
Leaves |
quercetin 3-O-neohispredoside |
FTG |
- |
[15] |
|
Leaves |
quercetin 3-O-robinoside |
FTG |
- |
[15] |
|
Leaves |
catechine |
FTG |
- |
[15] |
|
Leaves |
chlorogenic acid |
FTG |
- |
[15] |
|
Leaves |
argentinine (1-N,N-dimethylethanyl-4,6-dimethoxy-3,8-dihydroxy-phenanthrene) |
FTG |
- |
[15] |
|
Leaves |
kaempferol 3-O-rutinoside |
FTG |
- |
[15] |
|
Leaves |
quercetin 3-O-glucoside |
FTG |
- |
[15] |
|
Leaves |
quercetin |
FTG |
- |
[15] |
|
Leaves |
kaempferol |
FTG |
- |
[15] |
|
Leaves |
annonamine |
ALK |
- |
[40] |
|
Leaves |
(S)-norcorydine |
ALK |
- |
[40] |
|
Leaves |
(R)-4′-O-methylcoclaurine |
ALK |
- |
[40] |
|
Leaves |
(R)-O,O-dimethylcoclaurine |
ALK |
- |
[40] |
|
Leaves |
annoionol A |
MG |
- |
[14] |
|
Leaves |
annoionol B |
MG |
- |
[14] |
|
Leaves |
annoionol C |
MG |
- |
[14] |
|
Leaves |
annoionoside |
MG |
- |
[14] |
|
Leaves |
vomifoliol |
MG |
- |
[14] |
|
Leaves |
roseoside |
MG |
- |
[14] |
|
Leaves |
turpinionoside A |
MG |
- |
[14] |
|
Leaves |
citroside A |
MG |
- |
[14] |
|
Leaves |
blumenol C |
MG |
- |
[14] |
|
Leaves |
(+)-epiloliolide |
MG |
- |
[14] |
|
Leaves |
loliolide |
MG |
- |
[14] |
|
Leaves |
(1S,2S,4R)-trans-2-hydroxy-1,8-cineole β-d-glucopyranoside |
MG |
- |
[14] |
|
Leaves |
(Z)-3-hexenyl β-d-glucopyranoside |
MG |
- |
[14] |
|
Leaves |
rutin |
MG |
- |
[14] |
|
Leaves |
kaempferol 3-O-rutinoside |
MG |
- |
[14] |
|
Leaves |
kaempferol 3-O-robinobioside |
MG |
- |
[14] |
|
Leaves |
kaempferol 3-O-β-d-(2′′-O-β-d-glucopyranosyl,6′′-O-α-l-rhamnopyranosyl)glucopyranoside |
MG |
- |
[14] |
|
Roots |
montecristin |
AGE |
- |
[41] |
|
Roots |
cohibin A |
AGE |
- |
[42] |
|
Roots |
cohibin B |
AGE |
- |
[42] |
|
Roots |
cis-solamin |
AGE |
- |
[43] |
|
Roots |
cis-panatellin |
AGE |
- |
[43] |
|
Roots |
cis-uvariamicin IV |
AGE |
- |
[43] |
|
Roots |
cis-uvariamicin I |
AGE |
- |
[43] |
|
Roots |
cis-reticulatacin |
AGE |
- |
[43] |
|
Roots |
cis-reticulatacin-10-one |
AGE |
- |
[43] |
|
Roots |
chatenaytrienin 1 |
AGE |
- |
[44] |
|
Roots |
chatenaytrienin 2 |
AGE |
- |
[44] |
|
Roots |
chatenaytrienin 3 |
AGE |
- |
[44] |
|
Roots |
muridienin 3 |
AGE |
- |
[44] |
|
Roots |
muridienin 4 |
AGE |
- |
[44] |
|
Roots |
muricadienin |
AGE |
- |
[44] |
|
Roots |
coronin |
AGE |
- |
[45] |
|
Roots, Fruits |
sabadelin |
AGE |
- |
[24,46] |
|
Seeds |
murisolin |
AGE |
- |
[47] |
|
Seeds |
muricatacin |
AGE |
toxicity against lung A549, breast MCF7, colon HT-29 cancer cells |
[48] |
|
Seeds, Leaves, Pericarp |
annonacin |
AGE |
neurotoxic, molluscicidal, inhibitor of mitochondrial complex I |
[12,26,48,49,50,51] |
|
Seeds, Leaves |
corossolone |
AGE |
toxicity against oral KB cancer cells and brine shrimp larva, antileishmanial |
[37,52,53,54] |
|
Seeds |
corossolin |
AGE |
toxicity against oral KB cancer cells and brine shrimp larva |
[52] |
|
Seeds, Roots, Leaves |
solamin |
AGE |
toxicity against oral KB cancer and normal kidney VERO cells |
[37,43,55] |
|
Seeds |
corepoxylone |
AGE |
- |
[56] |
|
Seeds, Leaves |
annonacin-10-one |
AGE |
- |
[12,57] |
|
Seeds |
isoannonacin |
AGE |
molluscicidal, anticancer |
[49,57] |
|
Seeds |
isoannonacin-10-one |
AGE |
- |
[57] |
|
Seeds, Leaves |
goniothalamicin |
AGE |
molluscicidal |
[12,49,57] |
|
Seeds |
gigantetrocin |
AGE |
- |
[57] |
|
Seeds, Leaves |
gigantetrocin A |
AGE |
toxicity against colon HT-29 cancer cells |
[12,32,58] |
|
Seeds |
gigantetrocin B |
AGE |
toxicity against colon HT-29 cancer cells |
[12,32,58] |
|
Seeds, Leaves |
muricatetrocin A |
AGE |
toxicity against colon HT-29 cancer cells |
[58] |
|
Seeds, Leaves |
muricatetrocin B |
AGE |
toxicity against colon HT-29 cancer cells |
[58] |
|
Seeds, Leaves |
epomuricenin A |
AGE |
- |
[23,59] |
|
Seeds, Leaves |
epomuricenin B |
AGE |
- |
[23,59] |
|
Seeds |
annomuricatin A |
CP |
- |
[60,61] |
|
Seeds |
annocatacin A |
AGE |
toxicity against human hepatoma cells |
[38] |
|
Seeds |
annomuricatin C |
CP |
- |
[62] |
|
Seeds |
cis-annonacin |
AGE |
crown gall tumor inhibition, toxicity against brine shrimp, lung A549, breast MCF-7 and colon HT-29 cancer cells |
[63] |
|
Seeds |
cis-annonacin-10-one |
AGE |
crown gall tumor inhibition, toxicity against brine shrimp, lung A549, breast MCF-7 and colon HT-29 cancer cells |
[63] |
|
Seeds |
cis-goniothalamicin |
AGE |
crown gall tumor inhibition, toxicity against brine shrimp, lung A549, breast MCF-7 and colon HT-29 cancer cells |
[63] |
|
Seeds |
arianacin |
AGE |
crown gall tumor inhibition, toxicity against brine shrimp, lung A549, breast MCF-7 and colon HT-29 cancer cells |
[63] |
|
Seeds |
javoricin |
AGE |
crown gall tumor inhibition, toxicity against brine shrimp, A549, breast MCF-7 and colon HT-29 cancer cells |
[63] |
|
Seeds |
murihexol |
AGE |
- |
[33] |
|
Seeds |
donhexocin |
AGE |
- |
[33] |
|
Seeds |
cohibin C |
AGE |
- |
[64] |
|
Seeds |
cohibin D |
AGE |
- |
[64] |
|
Seeds |
muricatenol |
AGE |
- |
[32,65] |
|
Seeds |
2,4-cis-gigantetrocinone |
AGE |
- |
[32] |
|
Seeds |
2,4-trans-gigantetrocinone |
AGE |
- |
[32] |
|
Seeds |
2,4-trans-isoannonacin-10-one |
AGE |
- |
[32] |
|
Seeds |
annomontacin |
AGE |
- |
[32] |
|
Seeds |
longifolicin |
AGE |
toxicity against human hepatoma cells |
[66] |
|
Seeds |
muricin A |
AGE |
toxicity against human hepatoma cells |
[66] |
|
Seeds |
muricin B |
AGE |
toxicity against human hepatoma cells |
[66] |
|
Seeds |
muricin C |
AGE |
toxicity against human hepatoma cells |
[66] |
|
Seeds |
muricin D |
AGE |
toxicity against human hepatoma cells |
[66] |
|
Seeds |
muricin E |
AGE |
toxicity against human hepatoma cells |
[66] |
|
Seeds |
muricin F |
AGE |
toxicity against human hepatoma cells |
[66] |
|
Seeds |
muricin G |
AGE |
toxicity against human hepatoma cells |
[66] |
|
Seeds |
muricin H |
AGE |
toxicity against human hepatoma cells |
[37] |
|
Seeds |
muricin I |
AGE |
toxicity against human hepatoma cells |
[37] |
|
Seeds |
cis-annomontacin |
AGE |
toxicity against human hepatoma cells |
[37] |
|
Seeds, Leaves |
annonacinone |
AGE |
- |
[37] |
|
Seeds |
xylomaticin |
AGE |
- |
[37] |
|
Seeds |
N-fatty acyl tryptamines |
ALK |
- |
[32] |
|
Seeds |
annoreticuin-9-one |
AGE |
- |
[24] |
|
Stem barks |
epoxymurin A |
AGE |
- |
[67] |
|
Stem barks |
epoxymurin B |
AGE |
- |
[67] |
|
Leaves, Roots, Stems, Barks |
reticuline |
ALK |
- |
[68] |
|
Leaves, Roots, Stems, Barks |
coclaurine |
ALK |
- |
[68] |
|
Leaves, Roots, Stems, Barks |
coreximine |
ALK |
- |
[68] |
|
Leaves, Roots, Stems, Barks |
atherosperminine |
ALK |
- |
[68] |
|
Leaves, Roots, Stems, Barks |
stepharine |
ALK |
- |
[68] |
|
Leaves, Roots, Stems, Barks |
anomurine |
ALK |
- |
[68] |
|
Leaves, Roots, Stems, Barks |
anomuricine |
ALK |
- |
[68] |
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT editor-in-chief@pharmatutor.org
Subscribe to Pharmatutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE
Essential Oil
GC and GC-MS analyses on the leaf oil of A. muricata collected from Cameroon showed the presence of mostly sesquiterpenes, with the major compound present being β-caryophyllene69, A. muricata collected from Vietnam identified significant volatile oil constituents of β-pinene (20.6%), germacrene D (18.1%), ρ-mentha-2,4(8)-diene (9.8%), α-pinene (9.4%) and β-elemene (9.1%) from the leaf oil70, compounds of δ-cadinene, epi-α-cadinol and α-cadinol were reported to be found in the leaf oil extracts18, high concentrations of mono- and sesquiterpenes, including β-caryophyllene, 1,8-cineole and linalool, were also isolated from the fruit pulp71.
Annonaceous Acetogenins
AGEs are a unique class of C-35/C37 secondary metabolites derived from long chain (C-32/C34) fatty acids in the polyketide pathway. They are usually characterized by a combination of fatty acids with a 2-propanol unit at C-2 that forms a methyl-substituted α,β-unsaturated γ-lactone72. Since the discovery of uvaricin from Uvaria accuminata in 1982, more than 500 AGEs have been identified from different parts of the plants in the Annonaceae family73,74. Due to the special structures and extensive biological activities, AGEs have attracted significant scientific interest in recent years. Various biological activities have been reported for AGEs, including antimalarial, antiparasitic and pesticidal activities72,75. However, the biological activities of AGEs are primarily characterized with toxicity against cancer cells and inhibitory effects against the mitochondrial complex I (mitochondrial NADH: ubiquinone oxidoreductase)76,77. Phytochemical investigations and biological studies on different parts of the A. muricata plant resulted in the identification of a wide array of AGE compounds, as summarized in Table 1.
Biological Activities
Anti-Arthritic Activity
A. muricata is among the ethnomedicines employed to treat arthritic pain. An in vivo study on different doses (3, 10, 30 and 100 mg/kg) of ethanolic extract from A. muricata leaves has investigated the anti-arthritic activity in complete Freund’s adjuvant (CFA)-induced arthritis in rats. According to the results, oral administration of the extract reduced the edema in a dose-dependent manner after two weeks of injection. Because the extract at higher doses significantly suppressed TNF-α and IL-1β expression in local tissue, the anti-arthritic activity of A. muricata leaves contributed to the suppression of pro-inflammatory cytokines78. Hence, the anti-arthritic potential of A. muricata was substantiated by the findings of this in vivo study.
Anticancer Activity
Plenty of studies report the significant antiproliferative effects of different extracts of the plant and isolated AGEs towards various cancer cell lines26,79,80,81,82; however, few of these studies have illustrated the underlying mechanism of action (Table 2). Recent in vitro studies were performed by our research group to determine the mechanism of action of ethyl acetate extract of A. muricata leaves against colon cancer cells (HT-29 and HCT-116) and lung cancer cells (A549). The leaf extract was able to induce apoptosis in colon and lung cancer cells through the mitochondrial-mediated pathway. This antiproliferative effect was associated with cell cycle arrest in the G1 phase83,84. In addition, the migration and invasion of colon cancer cells were significantly inhibited by the leaf extract. The activation of caspase 3 by the ethanolic extract of the leaves also demonstrated an apoptosis-inducing effect in myelogenous leukemic K562 cells, which was confirmed with a TUNEL assay85.
Table No. 1.2: Anticancer studies on A. muricata.
|
Plant Part |
Subject of Study |
Effect |
Reference |
|---|---|---|---|
|
ethyl acetate extract of the leaves |
lung A549 cancer cells |
mitochondrial-mediated apoptosis, cell cycle arrest at G1 phase |
[83] |
|
ethyl acetate extract of the leaves |
colon HT-29 and HCT-116 cancer cells |
mitochondrial-mediated apoptosis, cell cycle arrest at G1 phase, suppression of migration and invasion |
[84] |
|
water extract of the leaves |
rat’s prostate |
reduction of prostate size |
[86] |
|
ethanolic extract of the leaves |
breast tissues of mice |
prevention of DMBA-induced DNA damage |
[87] |
|
ethanolic extract of the leaves |
DMBA/croton oil induced mice skin papillomagenesis |
suppression of tumor initiation and promotion |
[88] |
|
ethanolic extract of the leaves |
DMH induced colon cancer |
reduction of ACF formation |
[89] |
|
ethanolic extract of the leaves |
K562 chronic myeloid leukemia cells |
induction of apoptosis |
[85] |
|
leaves boiled in water |
metastatic breast cancer |
stabilization of disease |
[90] |
|
ethyl acetate of the leaves |
azoxymethane induced colon cancer |
reduction of ACF formation |
[91] |
|
ethyl acetate of the leaves |
colon HT-29 cancer cells |
bioassay-guided isolation of annomuricin E and its apoptosis inducing effect |
[91] |
Recent in vitro and in vivo studies were performed on the water extract of the A. muricata leaves against the benign prostatic hyperplasia (BPH-1) cell line and rats’ prostates. The results showed a suppressive effect on BPH-1 cells with an IC50 value of 1.36 mg/mL after 72 h associated with an up-regulation of Bax and a down-regulation of Bcl-2 at the mRNA level. After two months of treatment with the extract (30 and 300 mg/mL doses), the size of the rats’ prostates were decreased, which was suggested to occur through apoptosis induction86. This promising antitumor effect also reported in an in vivo study on 7,12-dimethylbenzene anthracene (DMBA)-induced cell proliferation in the breast tissues of mice. The protective effect against DNA damage induced by DMBA showed that oral administration of the A. muricata leaves may have protective effects towards the development of breast carcinogenesis87. The leaves, even at the low dose of 30 mg/kg suppressed the initiation and promotion stage of skin papillomagenesis in mice that was induced by DMBA and croton oil, respectively88.
Moghadamtousi and colleagues [91] also examined the in vivo chemopreventive potential of the ethyl acetate extract of the A. muricata leaves against azoxymethane-induced colonic aberrant crypt foci (ACF) in rats. The oral administration of the extract at two doses (250 and 500 mg/kg) for 60 days significantly reduced ACF formation in rats, as assessed by methylene blue staining of colorectal specimens. The immunohistochemistry analysis showed that this activity was accompanied by the up-regulation of Bax and the down-regulation of Bcl-2. This significant reduction in ACF formation was also reported for the ethanolic extract of the leaves against 1,2-dimethyl hydrazine (DMH)-induced colon cancer89. Our study was followed by an in vitro bioassay-guided investigation against HT-29 cells, which led to the isolation of annomuricin E. This AGE showed mitochondrial-dependent apoptosis activity in colon cancer cells with an IC50 value of 1.62 ± 0.24 µg/mL after 48 h91.
Anticancer studies on A. muricata were not only limited to in vitro and in vivo investigations. A case study of a 66-year old woman with a metastatic breast cancer reported that consumption of the leaves boiled in water and Xeloda resulted in stabilization of the disease90. These substantial anticancer and antitumor activities mentioned for A. muricata leaves led to tablet formulations of the ethyl acetate-soluble fraction of the leaves, which contains AGEs that can be used as a cancer adjuvant therapy92.
Anticonvulsant Activity
In African countries, the decoction of the A. muricata leaves is traditionally used to control fever and convulsive seizures93. To substantiate the anticonvulsant activity of the leaves in ethnomedicine, Gouemo and colleagues93 investigated the effect of the ethanolic extract of the leaves against pentylenetetrazol-induced tonic-clonic seizures in mice. The result showed that the plant extract at 100 and 300 mg/kg doses significantly decreased the incidence and the mortality rate of tonic seizures.
Antidiabetic and Hypolipidemic Activity
Adeyemi and colleagues94 reported that daily intraperitoneal injection of streptozotocin-induced diabetic Wistar rats with the methanol extract of A. muricata leaves (100 mg/kg) for two weeks significantly reduced their blood glucose concentration from 21.64 to 4.22 mmol/L94. In addition, the extract at the same dose significantly decreased the serum total cholesterol, low-density lipoprotein, triglyceride and very low-density lipoprotein cholesterol95.
Anti-Inflammatory and Anti-Nociceptive Activities
Oral treatment in rats with A. muricata ethanolic leaf extracts (10, 30, 100 and 300 mg/kg) significantly reduced carrageenan-induced edema in rat paws by 79% in a dose-dependent manner, exhibiting its anti-inflammatory activities96. Oral administration in mice with the same extract showed significant suppression of abdominal contortions induced with acetic acid (0.6% v/v), exhibiting a powerful anti-nociceptive activity96,97.
Antioxidant Activity
Immoderate generation of intracellular reactive oxygen species (ROS) is a precursor of oxidative stress which subsequently catalyzes metabolic deficiency and cellular death through biochemical and physiological lesions98. The identification of antioxidants from natural products has become a matter of great interest in recent studies for their noteworthy role in nullifying the destructive effects of ROS99,100.
Antihypertensive Activity
To evaluate the antihypertensive properties of A. muricata leaves, aqueous leaf extract (9.17–48.5 mg/kg) was administered to normotensive Sprague–Dawley rats. The results demonstrated that treatments of rats with the leaf extract significantly decreased blood pressure in a dose-dependent manner without affecting heart rates. This effect was suggested to be induced through peripheral mechanisms involving the antagonism of Ca2+101.
Antiparasitic Activity
As a natural agent, A. muricata has been subjected to various pathogenic parasites to determine its cytotoxic effects (Table 3). The ethyl acetate leaf extract of A. muricata was assayed against three Leishmania species (PH8, M2903 and PP75) and Trypanosoma cruzi. Promising activity was reported with IC50 values lower than 25 µg/mL102.
Table No. 1.3: Antiparasitic studies on A. muricata.
|
Plant Part |
Subject of Study |
Result |
Reference |
|---|---|---|---|
|
ethyl acetate extract of the leaves |
Leishmania species (PH8, M2903, PP75), T. cruzi |
IC50 values lower than 25 µg/mL |
[103] |
|
ethyl acetate extract of the pericarp |
L. braziliensis, L. panamensis |
toxicity effect higher than Glucantime as a positive control |
[26] |
|
methanol extract of the seeds |
L. donovani, L. mexicana, L. major |
bioassay-guided isolation of annonacinone (EC50: 6.72–8.00 µg/mL) and corossolone (EC50: 16.14–18.73 µg/mL) |
[53] |
|
methanol-water extract of the seeds |
L. chagasi (promastigote amastigote) |
bioassay-guided isolation of annonacinone and corossolone |
[54] |
|
aqueous extract of the leaves |
H. contortus |
toxicity against larvae (89.08%) and egg (84.91%) |
[101] |
|
pentane extract of the leaves |
P. falciparum |
toxicity against chloroquine sensitive and (IC50: 16 µg/mL) and resistant strains (IC50: 8 µg/mL) |
[02] |
Antiplasmodial Activity
Malaria, one of the most debilitating diseases, afflicts a substantial population in tropical and subtropical zones103. The available antimalarial drugs demonstrate varying degrees of failure due to rapid spread of parasite resistance104. Therefore, research into new antiplasmodial agents against the pathogenic parasites is definitely warranted. The pentane leaf extract of A. muricata was assayed against two strains of Plasmodium falciparum: the Nigerian chloroquine-sensitive strain and FcM29-Cameroon (chloroquine-resistant strain); a promising antiplasmodial effect was obtained with an IC50 value of 16 and 8 µg/mL after 72 h, respectively103.
Hepatoprotective and Bilirubin-Lowering Activity
A. muricata is traditionally employed to treat jaundice in Ghana. A study was conducted to determine the in vivo bilirubin-lowering potential of the aqueous extract of A. muricata leaves. This study was performed on phenylhydrazine-induced jaundice in adult rats, and the levels of direct and total bilirubin were measured in rats orally treated with 50 and 400 mg/kg of the extract. The extract at both doses caused a significant reduction to hyperbilirubinemia, which was close to normal levels105.
Insecticidal Activity
Botanical insecticides can have a pivotal role in different agriculture programs, especially in small farming [106]. Due to the presence of AGEs, plants from the Annonaceae family such as A. mucosa and A. sylvatica have shown to be promising biopesticides among tropical plants72,122. An investigation on different Annona species showed the growth inhibition effect of A. muricata seeds and contact toxicity by topical administration to Trichoplusia ni larvae107.
Gastroprotective Activity
Gastroprotective activity of A. muricata leaves was examined against ethanol-induced gastric injury. The results of the oral administration of the ethyl acetate extract (200 and 400 mg/kg) showed significant antiulcer potential, which was mediated through protective effects against gastric wall mucosal damages. Immunohistochemical staining demonstrated that the leaf extract decreased the Bax protein expression and elevated the Hsp70 protein expression. The effect of the extract on the gastric tissues was accompanied with augmentation in the activity of enzymatic antioxidants and suppression of lipid peroxidation, representing the preservative effect against gastric wall mucus108.Molluscicidal Activity
To establish plant-derived molluscicides for the vector control of schistosomiasis, different parts of the Annona species were tested against Biomphalaria glabrata, both in egg masses and adult forms. Santos and colleagues, in 2001, demonstrated that the leaves of A. muricata possess significant toxicity against adult worms with an LD90 value of 8.75 ppm. Additional toxicity of the A. muricata leaves against snail egg masses was markedly noted among different Annona species109.
Wound Healing Activity
Moghadamtousi and colleagues110 investigated the wound healing activity of the ethyl acetate extract of A. muricata leaves (5% w/w and 10% w/w) against excisional wound healing in rats. Topical administration of the extract for 15 days demonstrated significant wound healing potential assessed by macroscopic and microscopic analyses. The anti-inflammatory effects of the extract were demonstrated during the healing process as shown by the up-regulation of Hsp70, as assessed by immunohistochemical evaluation.
Sustained release drug therapy:
The basic goal of therapy is to achieve a steady state blood level that is therapeutically effective and nontoxic for an extended period of time. The design of proper dosage regimens is an important element in accomplishing this goal. Sustained release, sustained action, controlled release, extended action, timed release, depot and repository dosage forms are terms used to identify drug therapy systems that are designed to achieve a prolonged therapeutic effect by continuously releasing medication over an extended period of time after administration of single dose. In the case of injectable dosage forms, this period is measured in hours and critically depends on the residence time of the dosage form in the gastrointestinal tract. The term controlled release has become associated with those systems from which therapeutic agents maybe automatically delivered at predetermined rates over a long period of time. Products of this type have been formulated for oral, injectable and topical use and inserts for placement in body cavities. Prolonged or sustained release systems only prolong therapeutic blood or tissue levels of the drug for an extended period of time111. In some sustained-release (SR) formulations, the drug dissolves into the matrix, and the matrix physically swells to form a gel, allowing the drug to exit through the gel's outer surface. There are certain considerations for the formation of SR formulation,
i. If the active compound has a long half-life (over 6 hours), it is sustained on its own.
ii. If the pharmacological activity of the active compound is not related to its blood levels, time releasing has no purpose.
iii. If the absorption of the active compound involves an active transport, the development of a time-release product may be problematic.
iv. Finally, if the active compound has a short half-life, it would require a large amount to maintain a prolonged effective dose. In this case, a broad therapeutic window is necessary to avoid toxicity; otherwise, the risk is unwarranted and another mode of administration would be recommended112.
The design of oral sustain drug delivery system(DDS) should be primarily aimed to achieve the more predictability and reproducibility to control the drug release, drug concentration in the target tissue and optimization of the therapeutic effect of a drug by controlling its release in the body with lower and less frequent dose. Conventional drug therapy typically involves the periodic dosing of a therapeutic agent that has been formulated in a manner to ensure its stability, activity and bioavailability. For most of the drugs, conventional methods of formulation are quite effective. However some drugs are unstable and toxic and have a narrow therapeutic range, exhibit extreme solubility problems, require localization to a particular site in the body or require strict compliance or long-term use. In such cases a method of continuous administration of drug is desirable to maintain fixed plasma drug levels. The goal in designing sustained or sustained delivery systems is to reduce the frequency of the dosing or to increase effectiveness of the drug by localization at the site of action, reducing the dose required or providing uniform drug delivery. So, sustained release dosage form is a dosage form that release one or more drugs continuously in a predetermined pattern for a fixed period of time, either systemically or to a specified target organ. Sustained release dosage forms provide a better control of plasma drug levels, less dosage frequency, less side effect, increased efficacy and constant delivery113.
The major Drawbacks Associated with Conventional Dosage Forms:114
- Poor patient compliance, increased chances of missing the dose of a drug with short half-life for which frequent administration is necessary.
- The unavoidable fluctuations of drug concentration may lead to under medication or over medication.
- A typical peak-valley plasma concentration-time profile is obtained which makes attainment of steady-state condition difficult.
- The fluctuations in drug levels may lead to precipitation of adverse effects especially of a drug with small Therapeutic Index (TI) whenever over medication occur.
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT editor-in-chief@pharmatutor.org
Subscribe to Pharmatutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE
Advantages:111
1. The frequency of drug administration is reduced.
2. Patient compliance can be improved.
3. Drug administration can be made more convenient.
4. The blood level oscillation characteristics of multiple dosing of conventional dosage form is reduced, because a more even blood level can be maintained.
5. Better control of drug absorption can be attained, since the high blood level peak that may be observed after administration in an extended action form.
6. The characteristic blood level variations due to multiple dosing of conventional dosage form can be reduced.
7. The total amount of drug administration can be reduced, thus
-Maximizing availability with minimum dose.
-Minimize drug accumulation with chronic dosing.
8. Safety margin of high potency drugs can be increased and the incidence of both local and systemic adverse side effects can be reduced in sensitive patients.
9. Improve efficacy in treatment,
-Cure or control condition more promptly.
-Improve/ control i.e. reduces fluctuation in drug level.
-Improve bioavailability of some drugs.
-Make use of special effect e.g. sustained release aspirin for morning relief of arthritis by dosing before bed time
Disadvantages:115
1. Administration of sustained release medication does not permit prompt termination of therapy.
2. Flexibility in adjustment in dosage regimen is limited.
3. Controlled release forms are designed for normal population i.e., on the basis of average drug biological half-lives.
4. Economy factors may also be assessed, since most costly process and equipment are involved in manufacturing so many controlled release dosage forms.
Design of oral sustained release drug delivery system: The oral route administration is mostly adopted route because of its comfortable dosage form, design and patient care. Several parameters should be kept in mind before formulating sustain release dosage form which includes various pH in GIT, the gastrointestinal motility, the enzyme system and its effect on the dosage form and the drug. Most of sustained release dosage form follows the mechanism of diffusion, dissolution or combination of both, to produce slow release of drug at predetermined rate. Hypothetically, a sustained release dosage form should release the drug by a zero-order mechanism which maintains drug plasma level time similar to intravenous infusion. Plasma drug concentration-profiles for conventional tablet or capsule formulation, a sustained release formulation, and a zero order sustained release formulation are as follow in given figure
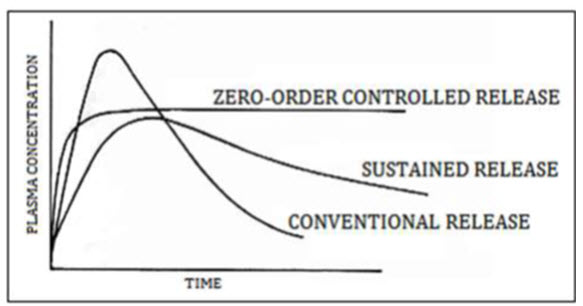
Figure 1.2: Plasma drug concentration profile for conventional release, a sustained release and zero order controlled release formulation
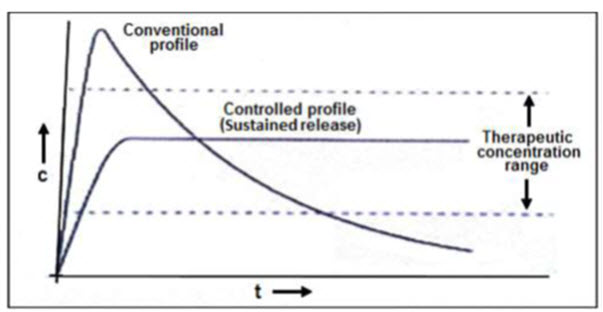
Figure 1.3: Comparison of conventional and controlled release profiles.
OBJECTIVES
AIM OF THE STUDY
Annona muricata is also called as “Graviola” (SOURSOP). Annona muricata L. is a naturally occurring plant traditionally used to treat various ailments including cancer.
The therapeutic dosage of Annona muricata leaf (which offers just as high of an amount of acetogenins as the root and almost as much as the seed) is reported to be 2-3g taken three or four times daily. The traditional preparation of Annona muricata leaf reported to be 2g, three times daily in capsule or tablets.
Elisya et; al they formulated a tablet by using wet granulation method by extracting the Annona muricata leaf by using polar solvent and made a dose of 100 mg. Acetogenin is relatively stable in the presence of moisture and to obtained a good flow property they used wet granulation method.
Since the dose of the traditional preparation was very high and a conventional tablet formulation was prepared by Elisya et; al, an attempt was made to formulate the sustained release tablet dosage form aiming to reduce the dose, improve therapeutic efficacy, to provide a standardized formulation and also to improve patient compliance.
OBJECTIVE OF THE STUDY:
- To reduce the size of the dose of Annona muricata for better therapeutic action.
- To reduce the frequency of dosing for anticancer activity.
- To formulate sustained release tablet of Annona muricata with suitable polymers and other excipients.
- To evaluate the prepared sustained release tablet containing Annona muricata.
PLAN OF WORK:
- Ethyl acetate Extraction of leaves of Annona muricata.
- Phytochemical screening of ethyl acetate leaf extract of Annona muricata.
- Biological studies of ethyl acetate leaf extract of Annona muricata.
• Brine shrimp lethality assay (BSLA) of ethyl acetate leaf extract of Annona muricata.
• MTT assay for ethyl acetate leaf extract of Annona muricata.
- Preformulation evaluation
• Fourier-transform infrared (FT-IR)
- Formulation of sustained release tablet containing ethyl acetate leaf extract of Annona muricata.
• Preparation of granules containing ethyl acetate leaf extract of Annona muricata with suitable excipients.
• Evaluation of pre-compression parameters of the granules containing ethyl acetate leaf extract of Annona muricata.
- Angle of repose
- Bulk density
- Tapped density
- Hausner ratio
• Compression of sustained release tablet containing ethyl acetate leaf extract of Annona muricata.
• Evaluation of post-compression parameters of the sustained release tablets containing ethyl acetate leaf extract of Annona muricata.
- Hardness
- Thickness
- Friability
- Analytical studies like HPLC, HPTLC, LC-MS or GC-MS.
- Stability studies
LITERATURE REVIEW
* Gajalakshmi et al., (2012) attempted the traditional, phytochemical and pharmacological studies done on an important medicinal plant Annona muricata, (Family annonaceae). Cyclo hexapeptides, acetogenins, annonaceous acetogenins were the major phytochemical compounds studied from this medicinal plant. The fruit is of economic value and hence cultivated and used widely as an edible food. The plant possess the major pharmacological activities includes cytotoxic, antileishmanial, wound healing, anti-microbial activity. It also has the anticarcinogenic and genotoxic effect. Phytochemical analysis of the plant revealed the presence of tannins, steroids and cardiac glycosides which are the major phytochemical compounds. The pulp obtained from the plant shows the thermal diffusivity property.116
* Kedari et al., (2014) summarized the information concerning the traditional uses, phytochemistry and biological activity of Annonaceous acetogenins. Extracts and metabolites from this plant exhibited pharmacological properties such as anti-inflammatory, antiulcer, anthelmintic, antibacterial, and free radical scavenging activity. Beside medicinal uses, this plant has high economic value due to its edible and nutritive fruit, leaves, bark and provides substantial livelihood support to local inhabitants. A wide range of chemical compounds including Alkaloids(acetogenins), lipids, isoquinoline, lactones, Annomuricatina (protein), Bullatacin, Muricoreacinetc. had been isolated from this species.117
* Tellez et al., (2016) focused the phytochemicals contents, bioactivity, biological actions and toxicological aspects of extracts and isolated compounds, as well as medicinal uses of A. muricata, with the objective of stimulating further studies on extracts and fruit pulp used for human consumption. Traditional medicinal uses of A. muricata have been identified in tropical regions to treat diverse ailments such as fever, pain, respiratory and skin illness, internal and external parasites, bacterial infections, hypertension, inflammation, diabetes and cancer. More than 200 chemical compounds have been identified and isolated from this plant; the most important being alkaloids, phenols and acetogenins. Using in vitro studies, extracts and phytochemicals of A. muricata have been characterized as an antimicrobial, anti-inflammatory, anti-protozoan, antioxidant, insecticide, larvicide, and cytotoxic to tumor cells. In vivo studies of the crude extracts and isolated compounds of A. muricata were shown to possess anxiolytic, anti-stress, anti-inflammatory, contraceptive, anti-tumoral, antiulceric, wound healing, hepato-protective, anti-icteric and hypoglycemic activities.118
* Mathew et al., (2016) determined the antimicrobial effect of water extracts of leaves of Annona muricata and Simarouba glauca on Enterococcus faecalis using agar diffusion method. Dried leaves of A. muricata and S. glauca were powdered and extracted in a soxhlet apparatus. Enterococcus faecalis was grown overnight in Trypticase soy agar plates. About 10 µL of each extract was placed on agar plates and incubated overnight. The zone of inhibition was measured after 24 hours. About 1% sodium hypochlorite and distilled water were used as positive and negative controls. The leaf extract of A. muricata showed similar effectiveness as that of sodium hypochlorite, whereas the leaf extract of S. glauca showed only a slight reduction in growth of E. faecalis.119
* Chang et al., (2001) isolated seven new annonaceous acetogenins, muricins A−G (1−7), as well as five known compounds, a mixture of muricatetrocin A (8) and muricatetrocin B (9), longifolicin (10), corossolin (11), and corossolone (12) from the seeds of Annona muricata. The structures of all isolates were elucidated and characterized by spectral and chemical methods. These acetogenins showed significantly selective in vitro cytotoxicities toward the human hepatoma cell lines Hep G2 and 2,2,15.120
* Gleye et al., (2000) isolated two new annonaceous acetogenins, cohibins C (1a) and D (1b), by extensive chromatography of a hexane extract of Annona muricata seeds and a cyclohexane extract of Annona nutans root bark. Their structures have been established on the basis of spectral evidence (NMR, MS) and confirmed by chemical transformation into a pair of monotetrahydrofuran (mono-THF) acetogenins. The role of these compounds in the biogenesis of mono-THF acetogenins is discussed.121.
* Sulistyoningrum et al., (2017) evaluated the effect of ethanolic leaves extract of Annona muricata (ELEAM) on breast cancer histology and on proliferative indexes of DMBA-induced breast cancer rats. Eighty four female albino rats strain Sprague Dawley were divided into seven groups (of twelve each): control; DMBA-induced breast cancer; DMBA-induced breast cancer + tamoxifen 0.18 mg/kg; DMBA-induced breast cancer +ELEAM 200 mg/kg, DMBA-induced breast cancer + ELEAM 300 mg/kg, DMBA-induced breast cancer + ELEAM 400 mg/kg and DMBA-induced breast cancer + ELEAM 500 mg/kg. The research indicates that breast cancer ductal with infiltrative grade II developed in the DMBA-induced breast cancer group. The DMBA-induced breast cancer group receiving tamoxifen showed normal duct structure. The tamoxifen treated groups showed lower mAgNOR and pAgNOR compared with untreated cancer group. The ELEAM-treated group showed lower mAgNOR and pAgNOR compared with the untreated cancer group, starting from 300 mg/kg dose. The group treated with ELEAM 300 mg/kg showed no significant difference with the tamoxifen-treated group. We concluded that ELEAM improved the histological changes of breast cancer-induced DMBA. ELEAM also reduced proliferative indexes of breast cancer-induced DMBA and the most effective dose in 300 mg/kg. 122
* Acevedo et al., (2016) designed and evaluated the antiallergic effect of the atomized extract of rhizome of Curcuma longa, flowers of Cordia lutea, and leaves of Annona muricata. Twenty-four New Zealand white albino rabbits were randomized into 2 groups. Group A received the atomized extract diluted in physiological saline (APS) and group B received it diluted in Freund’s adjuvant (FA). Then, the back of each rabbit was divided into 4 quadrants. The A-I quadrant received only physiological saline. The A-I quadrants of each rabbit conformed the PS group. The following 3 quadrants received the APS in 10 μg/mL, 100 μg/mL, and 1,000 μg/mL, respectively. The B-I quadrant received only FA. The B-I quadrants of each rabbit conformed the FA group. The following 3 quadrants received the AFA in 10 μg/mL, 100 μg/mL, and 1,000 μg/mL, respectively. The occurrence of erythema and edema was recorded according to the Draize scoring system and the primary irritation index. After 72 hours, biopsies were performed. The AFA group presented significantly less erythema and edema compared to the FA group (P<0.05). The histopathologic evaluation at 72 hours showed normal characteristics in the APS group.123
* Ratnhaparkhi et al., (2013) briefly reviewed on various formulation approaches for Sustained release drug delivery system. Oral drug delivery is the most preferred and convenient option as the oral route provides maximum active surface area among all drug delivery system for administration of various drugs. The attractiveness of these dosage forms is due to awareness to toxicity and ineffectiveness of drugs when administered by oral conventional method in the form of tablets & capsules. Usually conventional dosage form produces wide range of fluctuation in drug concentration in the bloodstream and tissues with consequent undesirable toxicity and poor efficiency. The maintenance of concentration of drug in plasma within therapeutic index is very critical for effective treatment.124
* Pooja et al., (2016) briefly emphasized about the sustained release drug delivery system characteristics, formulation design and drug release mechanisms. Sustained release dosage forms are designed to release a drug at a predetermined rate by maintaining a constant drug level for a specific period of time with minimum side effects. In the recent years, focus on the development of controlled release drug delivery systems has increased. The basic rationale of controlled release drug delivery system optimizes the biopharmaceutical, pharmacokinetic, and pharmacodynamic properties of a drug in such a way that its utility is maximized, side-effects are reduced and cure or control of the condition is achieved, in the shortest possible time by using smallest quantity of drug administered by the most suitable route.125
* Hussein et al., (2015) collected fresh okra and macerated in different extracting systems in variable ratios. The extracts were dried and collected. Selected extracts were granulated and compressed into tablets using the tablet machine. The formulated tablets were characterized in terms of hardness, friability disintegration and other gross findings, Extract from HCl and NaoH (OE 7 and8) were selected to prepare SR tablet of pentoxifylline (PTX); the same tests were performed in addition to measure the rate of PTX release from the tablet over ten hours. Results: Results obtained shows a promising retardation polymer as the tablets has an elegant shape and texture without chips or cracks, and not friable (FHCl 1loses 0.03 % of its weight after friability testing), tablet strength is acceptable since tablets resist breaking strength more than 200 Newtons, in addition, time needed for tablet disintegration about 175 minutes as found in F NaoH 3. Fortunately, the prepared tablets show a slow release of PTX following zero order kinetic (90 % released in eight hours in a constant rate about 10% per hour).126
* Rastogi et al., (2011) developed the sustained release matrix tablet of Boswellia serrata, Moringa oleifera and Vitex negundo using the combination of hydrophilic and hydrophobic polymers.Tablet formulations were developed using wet granulation method. Ethanolic extracts of Boswellia serrata, Moringa oleifera and Vitex negundo were used. Addition of different diluents like talc , magnesium stearate and microcrystalline cellulose were used for improving flow ability and compressibility. The tablets were subjected to physicochemical characterization, in vitro drug release and stability studies. The physicochemical properties were found within limits. The % drug release after 12 hours for formulation F-1, F-2, F-3 was found to be 97.32, 90.12, and 73.85 respectively. The presence of Ethyl cellulose as well as HPMC as the total matrix material significantly influenced the release rate of the drug. Based on dissolution studies all the formulations showed sustained release of drugs from the formulations.127
* Tavakoli et al., (2008) collected the leaves of grape and wild strawberry, identified and dried. The content of anthocyanins present in the powdered leaves was measured based on a spectrophotometric differential pH method. To prepare chewable tablet (HepatoHeal), the same amount of powdered leaves of two plants (40 mg) was mixed to filler (mannitol or lactose) and granulated using wet granulation method. The resultant tablets were evaluated for hardness, friability, disintegration time, drug content uniformity, drug release test and organoleptic properties. The assay showed that the content of anthocyanin in grape and strawberry leaves were 0.082(w/w) % and 0.039(w/w) %, respectively. The mean weight, friability, hardness, and disintegration time of selected formulation were 262 mg, 0.23%, 59.7 N and 22.6 min, respectively. The content of active ingredient (based on anthocyanin) was 44.8 mg and the content uniformity of the selected tablet was 42.8 mg. Percent of the drug released after 30 and 60 min was 76 % and 97 % respectively. 128
* Nitin et al., (2010) developed and investigated three orally administrable dosage forms of fruits of Piper nigrum (Maricha) and leaves of Nyctanthes arbortristis (Parijataka), in combination. Tablet form of drugs from solid dosage form and two formulations from liquid class were designed and developed. By considering difficulty of solubility of herbal drugs in a vehicle, decoction form of drugs in specific vehicle was used. This form of drugs would be considered as Liquid Oral Dosage Form of drugs. Suspension form was also designed. Formulated dosage forms were then subjected to evaluation of production quality by different methods stated as per official compendia. Such evaluation had unique position in development of new formulations. 129
* Shikha et al., (2012) prepared a combined herbal formulation of the extracts of dried root powder of Plumbagozeylanica and powdered seeds of Silybummarianum for the treatment of dementia. HPTLC quantification characterization and identification of the herbal extracts of P. zeylanica and S. marianum, was performed using the standard marker compound, plumbagin and silibinin respectively of these drugs. Through the acute oral toxicity studies the dose of the extract in the formulation and a LD50 and ED50 profile of the drug combination were established. Design & development of tablet using two doses of the extract was done and evaluation was carried out on various standard parameters of formulation including accelerated stability tests, which was in turn found satisfactory and within the specified limits. Preliminary pharmacological screening of dementia was performed on formulated tablets using EPM (Elevated plus maze) and MWM (Morris water maze), two of the animal models of dementia. Here, Piracetam (400mg/kg p.o) a nootropic agent was employed as a standard drug. Sodium Nitrite (75mg/kg i.p) was used to induce amnesia in young experimental model, and which is comparable with that of the age related amnesia in old rats. The results indicate that administration of tablets produce significant dose dependant improvement of memory and were almost similar with that of standard drug Piracetam.130
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT editor-in-chief@pharmatutor.org
Subscribe to Pharmatutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE
PLANT PROFILE
ANNONA MURICATA
Annona muricata is a slender, evergreen tree, 5-10 m in height and 15 cm in diameter; trunk straight; bark smooth, dull grey or grey-brown, rough and fissured with age; inner bark pinkish and tasteless; branches at first ascending with the crown forming an inverted cone, later spreading; crown at maturity spherical due to lack of apical dominance; twigs brown or grey, bearing minute raised dots (lenticels); root system extensive and superficial, spreading beyond the diameter of the crown although shallow rooted; juvenile plants have a taproot that is eventually lost. Leaves alternate, 7.6-15.2 cm long, 2.5-7.6 cm wide, leathery, obovate to elliptic, glossy on top, glabrous on underside, simple; stipules absent; blade oblanceolate, green on top, paler and dull on under side with fine lateral nerves; a strong, pungent odour; petioles short, 3-10 mm long. Flower terminal or lateral, large; stalks stout, green, 1.3-1.9 cm long; 3 sepals, minute, inconspicuous, broad, green, 3 mm long, triangular; petals yellowish-green, 6 in 2 whorls of 3, outer petals larger, ovate-acute, valvate, cordate with pointed apex (heart shaped), 4-5 x 3-4 cm, 3 mm thick and fleshy, fitting together at edges in bud and rough on the outside; 3 inner petals, narrow, smaller, nearly 3.8 cm long, thinner, rounded, concave with fingernail-shaped base and overlapping edges; stamens numerous, shield shaped, united below; anthers parallel and opening longitudinally; carpels numerous, overtopping the stamens, each with 1 ovule; pistils white, narrow, 5 mm long, with sticky stigmas. Fruit 14-40 x 10-18 cm, weighing up to 7 kg, ovoid, heart shaped, an oblong syncarp composed of numerous united pistils, pistils end in a fleshy spine or short base of spine 1.5 mm or more in length, which grows from the style; often asymmetric due to incomplete fertilization of the ovules; epidermis often shining, dark green, with short, fleshy spines covering each carpel; pulp white, fibrous and juicy; seeds shiny, dark brown or black, oblong, up to 2 cm long, 0.7 cm wide. The genus name ‘Annona’ is from the Latin word ‘anon’, meaning ‘yearly produce’, referring to the fruit production habits of the various species in this genus.
Biology:
Flowers are protandrous, and the pollen is shed as the outer petals open towards the evening. The inner petals open much later and only very slightly, admitting small insects attracted by the fragrance of the flowers. Beetles of several species are important in carrying out natural pollination. Presumably these insects effect cross-pollination, though rather inadequately, for few flowers set fruit and many fruits are misshapen since numerous ovules are not fertilized. Hand pollination is effective in improving fruit yield and quality. Fruiting starts in the 2nd year, and 5-yearold trees produce 10-50 fruits, depending on pollination efficiency and nutrient status. Sporadic flowering and fruiting can occur all year round in favourable conditions.
EXCIPIENT PROFILE
3.2.1 CARBOPOL 974P
Carbopol® 974P NF polymer was introduced for use in oral and mucosal contact applications such as oral liquids, bioadhesive formulations, oral care formulations and extended release tablets. Additionally, Carbopol 974P NF polymer can be used to formulate viscous gels, emulsions and suspensions. It is a highly crosslinked polymer and produces highly viscous gels with rheology similar to mayonnaise. Drug release from extended release tablets is affected by differences in the rates of hydration and swelling of the polymer hydrogel, which are largely defined by the crosslinker levels. Lightly crosslinked polymers, such as Carbopol 971P NF polymer, tend to be more efficient in controlling drug release than highly crosslinked polymers such as Carbopol 974P NF polymer.
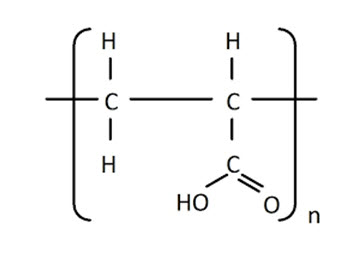
Figure No. 3.7 Structure of Carbopol 974P
Carbopol® polymers can be used alone as a controlled release agent in matrix tablets or in combination with hypromellose. Potential benefits to be derived from the polymer combination matrix (Carbopol® polymer/hypromellose) versus use of a single polymer matrix (Carbopol® polymer or hypromellose) are as follows: • Lower total polymer level needed - Formulation cost savings - Better patient compliance with smaller tablets • Performance consistency with regard to drug release • Flexibility in modulating drug release • Ability to further extend the release of some cationic drugs The synergistic effects of a polymer combination matrix have been demonstrated in a variety of APIs with different solubilities. Benefits were observed in matrix tablets manufactured by direct compression and wet granulation with both low- and mid-dose tablets.
ETHYL CELLULOSE
Ethyl cellulose (EC) is available under the brand name Ethocel 7 cps was sourced from DOW Chemicals Company. The chemical name of ethyl cellulose is Cellulose ethyl ether (CAS no. 9004-57-3). The product has approved regulatory status as per USP-NF, Ph.Eur. JP and BP. It is a derivative of cellulose in which some of the hydroxyl groups on the repeating glucose units are converted into ethyl ether groups. Figure 4.9 Structure of ethyl cellulose It is available as free flowing powder, white or light tan in color with a density of 0.4g/cm2 . It is practically insoluble in water, glycerol and propane-1-2-diol, but soluble in organic solvents. Ethyl cellulose containing 46-48% of ethoxyl group is freely soluble in ethanol, methanol, chloroform and ethyl acetate. Neutral to litmus, with LOD not more than 3%. It is mainly used as a thin-film coating material. In addition to being useful in a variety of pharmaceutical applications. It also features a fine particle (FP) range for use in extended release matrix systems and provides improved lipophilic properties realized by the increased Chapter 4 Drug and Excipient profile SPP School of Pharmacy & Technology Management, SVKM’s NMIMS, Mumbai 75 surface area. This flexibility is further enhanced by the ability to modify release profiles when ETHOCEL™ is used in combination with water-soluble excipients such as Colorcon’s METHOCEL™ premium cellulose ethers175, 176 . Ethyl cellulose is a stable, slightly hygroscopic material. It is chemically resistant to alkalies. Ethyl cellulose is prone to oxidative degradation in presence of UV light. Ethyl cellulose should be stored at a temperature not exceeding 32oC in a dry area and away from heat.
MICROCRYSTALLINE CELLULOSE
CELLULOSE, MICROCRYSTALLINE (Avicel PH-101)
Nonproprietary Names
BP : Microcrystalline cellulose
JP : Microcrystalline cellulose
PhEur : Cellulosum microcristallinum
USPNF : Microcrystalline cellulose
Synonyms Avicel PH; Celex; cellulose gel; Celphere; Ceolus KG; crystalline cellulose; E460; Emcocel; Ethispheres ; Fibrocel; Pharmacel; Tabulose; Vivapur. Chemical Name and CAS Registry Number: Cellulose [9004-34-6]
Empirical Formula and Molecular Weight: (C6HO105)n = 36 000,where n =220. Functional Category: Adsorbent; suspendin agent; tablet and capsule diluent; tablet disintegrant.
Structural Formula:
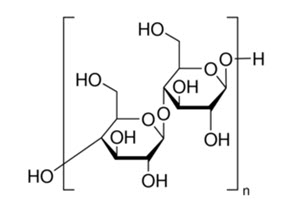
Applications in Pharmaceutical Formulation or Technology Microcrystalline cellulose is widely used in pharmaceuticals, primarily as a binder/diluent in oral tablet and capsule formulations where it is used in both wet-granulation and directcompression processes. In addition to its use as a binder/diluent, Microcrystalline cellulose also has some lubricant and disintegrant properties that make it useful in tableting. Microcrystalline cellulose is also used in cosmetics and food products.
Uses of Microcrystalline cellulose:
1. Adsorbent 20–90
2. Antiadherent 5–20
3. Capsule binder/diluents 20–90
4. Tablet disintegrant 5–15
5. Tablet binder/diluents 20–90
Description: Microcrystalline cellulose is purified, partially depolymerized cellulose that occurs as a white, odorless, tasteless, crystalline powder 69 composed of porous particles. It is commercially available in different particle sizes and moisture grades that have different properties and applications. Typical Properties Density (bulk) : 0.32 g/cm for Avicel PH-101 Density (tapped) : 0.45 g/cm for Avicel PH-101; Density (true) : 1.512–1.668 g/cm Melting point : Chars at 260–270°C. Moisture content: Typically less than 5% w/w. However, different grades may contain varying amounts of water. Microcrystalline cellulose is hygroscopic.
Particle size distribution: Typical mean particle size is 20–200 µm. Different grades may have a different nominal mean particle size.
Solubility: Slightly soluble in 5% w/v sodium hydroxide solution; practically insoluble in water, dilute acids, and most organic solvents.
Stability and Storage Conditions: Microcrystalline cellulose is a stable though hygroscopic material. The bulk material should be stored in a well-closed container in a cool, dry place.
Incompatibilities: Microcrystalline cellulose is incompatible with strong oxidizing agents. Safety Microcrystalline cellulose is widely used in oral pharmaceutical formulations and food products and is generally regarded as a relatively nontoxic and nonirritant material. Microcrystalline cellulose is not absorbed systemically following oral administration and thus has little toxic potential. Consumption of large quantities of cellulose may have a laxative effect, although this is unlikely to be a problem when cellulose is used as an excipient in pharmaceutical formulations. Deliberate abuse of formulations containing cellulose, either by inhalation or by injection, has resulted in the formation of cellulose granulomas. Regulatory Status GRAS listed. MCC is accepted for use as a food additive in Europe. Included in the FDA Inactive Ingredients Guide (inhalations; oral capsules, powders, suspensions, syrups, and tablets; topical and vaginal preparations). MCC is included in nonparenteral medicines licensed in the UK. MCC is included in the Canadian List of Acceptable Non-medicinal Ingredients.
TRIBASIC CALCIUM PHOSPHATE
Tricalcium phosphate (sometimes abbreviated TCP) is a calcium salt of phosphoric acid with the chemical formula Ca3(PO4)2. It is also known as tribasic calcium phosphate and bone phosphate of lime (BPL). It is a white solid of low solubility. Most commercial samples of "tricalcium phosphate" are in fact hydroxyapatite. It exists as three crystalline polymorphs α, α', and β. The α and α' states are stable at high temperatures. As mineral, it is found in Whitlockite.
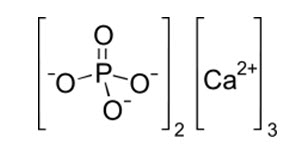
Calcium phosphate refers to numerous materials consisting of calcium ions (Ca2+) together with orthophosphates (PO43−), metaphosphates or pyrophosphates (P2O74−) and occasionally oxide and hydroxide ions. Especially, the common mineral apatite has formula Ca5(PO4)3X, where X is F, Cl, OH, or a mixture; it is hydroxyapatite if the extra ion is mainly hydroxide. Much of the "tricalcium phosphate" on the market is actually powdered hydroxyapatite.
It can be used as a tissue replacement for repairing bony defects when autogenous bone graft is not feasible or possible. It may be used alone or in combination with a biodegradable, resorbable polymer such as polyglycolic acid. It may also be combined with autologous materials for a bone graft. Porous beta-Tricalcium phosphate scaffolds are employed as drug carrier systems for local drug delivery.
3.2.5. PEG 4000
Nonproprietary Names: BP: Macrogols, JP: Macrogol 4000, PhEur: Macrogols, USP-NF: Polyethylene Glycol
Synonyms: Carbowax; CarbowaxSentry; Macrogola; PEG; Pluriol; Polyoxyethylene glycol.
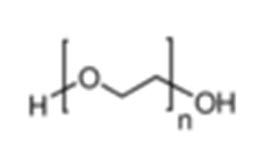
Chemical Name: α-Hydro-ω-hydroxypropyl (oxy-1,2-ethanediyl)
Empirical Formula and Molecular Weight: HOCH2(CH2OCH2) mCH2 OH where m represents the average number of oxyethylene groups. Alternatively, the general formula H(OCH2CH2)n OH may be used to represent polyethylene glycol, where n is a number m in the previous formula. Average molecular weight: 3000-4800
Structural Formula:
Description: The USP32–NF27 describes polyethylene glycol as being an addition polymer of ethylene oxide and water. Polyethylene glycol grades 200–600 are liquids; grades 1000 and above are solids at ambient temperatures.
Typical Properties:
• Density: 1.080 • Melting point: 69.0-84.0 0C
• Moisture content: Liquid polyethylene glycols are very hygroscopic, although hygroscopicity decreases with increasing molecular weight. Solid grades e.g. PEG 4000 and above, are not hygroscopic.
• Solubility: All grades of polyethylene glycol are soluble in water and miscible in all proportions with other polyethylene glycols (after melting, if necessary). Aqueous solutions of higher-molecular-weight grades may form gels. Liquid polyethylene glycols are soluble in acetone, alcohols, benzene, glycerin, and glycols. Solid polyethylene glycols are soluble in acetone, dichloromethane, ethanol (95%), and methanol. They are slightly soluble in aliphatic hydrocarbons and ether, but insoluble in fats, fixed oils, and mineral oil.
Functional Category: Ointment base; plasticizer; solvent; suppository base; tablet and Capsule lubricant.
Applications in Pharmaceutical Formulation or Technology: Polyethylene glycols (PEGs) are widely used in a variety of pharmaceutical formulations, including parenteral, topical, ophthalmic, oral, and rectal preparations. Polyethylene glycol has been used experimentally in biodegradable polymeric matrices used in controlled-release systems.
• Polyethylene glycols are stable, hydrophilic substances that are essentially nonirritant to the skin.
• They do not readily penetrate the skin, although the polyethylene glycols are water-soluble and are easily removed from the skin by washing, making them useful as ointment bases.
• Solid grades are generally employed in topical ointments, with the consistency of the base being adjusted by the addition of liquid grades of poly ethylene glycol. Stability and Storage Conditions: Polyethylene glycols are chemically stable in air and in solution, although grades with a molecular weight less than 2000 are hygroscopic. Polyethylene glycols do not support microbial growth, and they do not become rancid. Polyethylene glycols and aqueous polyethylene glycol solutions can be sterilized by autoclaving, filtration, or gamma irradiation
METHYL PARABEN
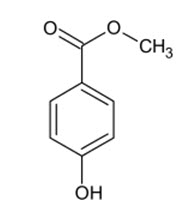
Methylparaben, also methyl paraben, one of the parabens, is a preservative with the chemical formula CH3(C6H4(OH)COO). It is the methyl ester of p-hydroxybenzoic acid.
Methylparaben serves as a pheromone for a variety of insects and is a component of queen mandibular pheromone. Some plants produce methylparaben, example thale cress. It is commonly used in the preparation of liquid dosage forms. Methylparaben is an anti-fungal agent often used in a variety of cosmetics and personal-care products. It is also used as a food preservative and has the E number E218.
Methylparaben is commonly used as a fungicide in Drosophila food media. To Drosophila, methylparaben is toxic at higher concentrations, has an estrogenic effect, and slows the growth rate in the larval and pupal stages at lower concentrations. Methylparaben and propylparaben are considered generally recognized as safe (GRAS) for food and cosmetic antibacterial preservation. Methylparaben is readily metabolized by common soil bacteria, making it completely biodegradable.
Methylparaben is readily absorbed from the gastrointestinal tract or through the skin.[6] It is hydrolyzed to p-hydroxybenzoic acid and rapidly excreted in urine without accumulating in the body. Acute toxicity studies have shown that methylparaben is practically non-toxic by both oral and parenteral administration in animals. In a population with normal skin, methylparaben is practically non-irritating and non-sensitizing; however, allergic reactions to ingested parabens have been reported. Studies indicate that methylparaben applied on the skin may react with UVB, leading to increased skin aging and DNA damage.
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT editor-in-chief@pharmatutor.org
Subscribe to Pharmatutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE
MATERIALS USED
Table No. 4.1: List of Chemicals used with Supplier
|
S. No |
Materials |
Name of the supplier |
| 1 |
Annona muricata leaves |
Sri Venkateshwara University, Tirupathi, Andhrapradesh |
| 2 |
Carbopol |
S.D Fine chem. LTD Mumbai |
| 3 |
Ethyl cellulose |
MYL CHEM Mumbai |
| 4 |
Tribasic calcium phosphate |
Kemwell, Bengaluru |
| 5 |
MicroCrystallineCellulose |
S.D Fine chem. LTD Mumbai |
| 6 |
PEG 4000 |
Merch India (P) Ltd |
| 7 |
Methyl paraben |
S.D Fine chem. LTD Mumbai |
| 8 |
Ethyl acetate |
S.D Fine chem. LTD Mumbai |
| 9 |
Ethanol |
S.D Fine chem. LTD Mumbai |
| 10 |
Brine shrimp eggs |
Aqua world, Chennai |
Instruments Used
Table No. 4.2: List of Equipments Used
|
S. No |
Instruments |
Source |
|
1 |
Electronic balance |
Shimadzu |
|
2 |
UV/Visible Spectrophotometer |
Corporation-BL-220H |
|
3 |
FTIR spectrophotometer |
Corporation japan |
|
4 |
Magnetic stirrer |
Remi motor equipments |
|
5 |
Dissolution apparatus |
Shimadzu |
|
6 |
Oven |
Biotech india. |
|
7 |
pH meter |
Shital scientific Industries |
|
8 |
Distillation unit |
Electrolab |
|
9 |
Compression machine |
Cadmach machinery |
Preparation of ethyl acetate extract from the leaves of Annona muricata:
Collection of the plant:
The plant sample (Annona muricata Linn.) was collected from Sri Venkateshwara University, Tirupathi, Andhrapradesh. The plant was authentified by Dr. Madhava Chetty, Assistant Professor, Department of Botany, Sri Venkateshwara University, Tirupathi, Andhrapradesh. The leaves were washed thoroughly in tap water and air dried in the shade for 4weeks. The leaves were grinded into coarse powder with the help of milling machine.
Ethyl acetate extraction of Annona muricata leaves:
Samples were extracted using maceration method by weighing a number (10.0 kg) soursop leaves (Annona muricata L.) was dried and crushed, then put into containers maceration then added thereto a number of 96% ethanol (80.0 liters) to soak the entire sample. The samples were left in the container for several days maceration (5 days) and then dilute extract obtained was concentrated by using a rotary evaporator, and then insert it into the oven temperature up to 50°C thick extract is obtained. Viscous extract obtained by adding the number of partitioned into solvent extracts (ethanol: n-hexane) with a ratio of 1:1, then added distilled water with the same ratio and mix using an electric mixer until a few moments. After mixing is complete, it is obtained that does not mix the two layers, separated by a layer of water on the solvent and subsequent water fraction. Perform the same process with the new solvent (ethyl acetate). The process is complete when the solvent used was clear. Ethyl acetate fraction obtained followed by concentration using a rotary evaporator, then put into an oven temperature of 50°C to obtain a viscous extract (VEAM).
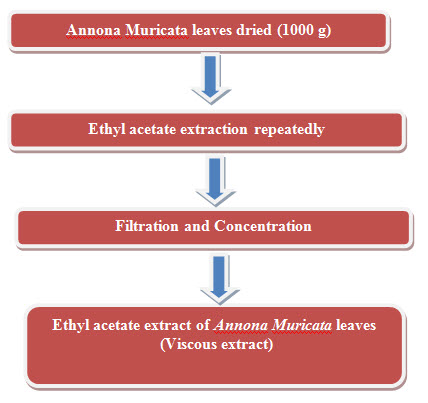
Flow chart No. 4.1: Ethyl acetate extraction of Annona muricata leaves
Preparation of free flowing powder of ethyl acetate extract leaves of Annona muricata:
The prepared ethyl acetate extract was highly viscous and hence it was not suitable for the preparation of tablets which was confirmed by the screening studies. Hence the ethyl acetate extract leaves of Annona muricata was further treated with various solvents like petroleum ether, ethanol to obtain a free flowing powder of ethyl acetate leaf extract of Annona muricata (PEAM).
Phytochemical screening of VEAM and PEAM:
The phytochemical screening tests of the viscous ethyl acetate leaf extract of Annona muricata (VEAM) and powdered ethyl acetate leaf extract of Annona muricata (PEAM) for qualitative analysis of primary and secondary metabolites was carried out as per the standard procedure131,132,133,134.
Test for alkaloids: A portion of the extract was made acidic with dilute sulphuric acid and the acidic extract was divided into two parts. With Mayer’s reagent it gives while ppt for positive test. With Dragendorff’s reagent it gives orange brown ppt. for the positive test.
Test for saponins: A small amount of the extract was boiled with water and allowed to cool. It was shaken vigorously in a test tube and left for a few minutes. The formation of persistent honey comb like froth was taken as a positive test.
Test for sterols and terpenes: A small amount of the extract was evaporated to dryness and extract was dissolved in 3 ml of chloroform. The filtrate was treated with three drops of a mixture of concentrated sulphuric acid and acetic anhydride. The production of different shade of color was recorded as a positive test.
It was furthered verified by Libermann Buchard test. A small portion of the extract was treated with hot acetic anhydride, cooled and then few drops of concentrated sulfuric acid were added, Production of bluish green color confirmed sterol while violet or pink for terpene.
Test for tannins: A small amount of the extract was treated with 5% ferric chloride solution and the production of green to blue color was taken as a positive test for tannins.
Test for proteins: Addition of very dilute copper sulphate to alkaline solution of protein gives red to violet solution that confirms protein by Biuret test. Protein produces yellow orange color when warmed with concentrated nitric acid and color gets orange when made alkaline in Xantho proteins test. Millon’s reagent gives white ppt. when a solution of mercuric nitrate containing nitrous acids is added to a protein solution.
Test for carbohydrate: Molish test is positive when on treatment with alpha napthol and concentrated sulphuric acid the extract gives purple color. Reduction of Fehling’s solution is seen when in a solution of carbohydrate equal quantity of Fehling A and B are added. After heating brick red ppt. is obtained. In Benedict’s test, the test solution gave yellow or reddish brown precipitate with Benedict’s reagent after boiling on water bath.
Test for anthraquinones: 0.5g of the extract was boiled with 10ml of H2SO4 and filtered while hot. The filtrate was shaken with 5ml of chloroform, the chloroform layer was pipette into another test tube and 1ml of dilute ammonia was added. The resulting solution was observed for colour changes.
Test for cardiac glycosides (Keller-Killiani test): To 0.5g of extract dissolved in 5ml water was added 2ml of glacial acetic acid solution containing one drop of ferric chloride solution. This was underlayed with 1ml of concentrated H2SO4. A brown ring at the interface indicated the presence of deoxysugar characteristics of cardenolides. A violet ring may appear below the brown ring while in the acetic acid layer a greenish ring may form just above the brown ring and gradually spread throughout this layer.
Test for flavonoids: There were two methods used to test for flavonoids.
(a) A portion of the extract was heated with 10ml of ethyl acetate over a steam bath for 3 minutes, the mixture was filtered and 4ml of the filtrate was shaken with 1ml of dilute ammonia solution. A yellow colouration indicated the presence of flavonoids.
(b) Dilute ammonia (5ml) was added to a portion of an acqueous filtrate of the extract. Then, concentrated sulphuric acid (1ml) was added. A yellow colouration indicated the presence of flavonoids.
Biological studies of viscous ethyl acetate leaf extract of Annona muricata (VEAM) and powdered ethyl acetate leaf extract of Annona muricata (PEAM):
The in vivo lethality in a simple zoological organism, such as the brine shrimp lethality test (BST), developed for Meyer et al., might be used as a simple tool to guide screening and fractionation of physiologically active plant extracts, where one of the simplest biological responses to monitor is lethality, since there is only one criterion: either dead or alive.
This general bioassay detects a broad range of biological activities and a diversity of chemical structures. One basic premise here is that toxicology is simply pharmacology at a higher dose, thus if we find toxic compounds, a lower, non-toxic, dose might elicit a useful, pharmaco- logical, perturbation on a physiologic system. However, it has been demonstrated that BST correlates reasonably well with cytotoxic and other biological properties.
Brine shrimp lethality assay (BSLA) of viscous ethyl acetate leaf extract of Annona muricata (VEAM) and powdered ethyl acetate leaf extract of Annona muricata (PEAM):
Brine shrimp eggs were obtained from the Aqua World, Chennai, Tamilnad, India. Filtered, artificial seawater was prepared by dissolving 38 g of sea salt in 1 liter of distilled water for hatching the shrimp eggs. The seawater was put in a small plastic container (hatching chamber) with a partition for dark (covered) and light areas. Shrimp eggs were added into the dark side of the chamber while the lamp above the other side (light) will attract the hatched shrimp. Two days were allowed for the shrimp to hatch and mature as nauplii (larva). After two days, when the shrimp larvae are ready, 4 mL of the artificial seawater was added to each test tube and 10 brine shrimps were introduced into each tube. Thus, there were a total of 30 shrimps per dilution. Then the volume was adjusted with artificial seawater up to 5 mL per test tube. The test tubes were left uncovered under the lamp. The number of surviving shrimps were counted and recorded after 24 hours. Using probit analysis, the lethality concentration (LC50) was assessed at 95% confidence intervals. LC50 of less than 100 ppm was considered as potent (active)136. As mentioned by Meyer and others137, LC50 value of less than 1000 µg/mL is toxic while LC50 value of greater than 1000 µg/mL is non-toxic. The percentage mortality (%M) was also calculated by dividing the number of dead nauplii by the total number, and then multiplied by 100%. This is to ensure that the death (mortality) of the nauplii is attributed to the bioactive compounds present in the plant extracts.

Figure 4.1: Brine shrimp lethality assay (BSLA) set-up
Samples are diluted serially 1 to 1000µg/ml and performed in triplets for individual sample to get mean average valve. Individual test tubes contains 3ml of distilled water and 1.5ml of Brine solution (72gm of NaCL) IN 500ml with distilled water. Later 0.5ml of samples of different dilution were added along with control which contain 0.5ml 1N NaoH instead of sample. It was kept over 24hours under lamp source for observation. After 24hours larvae retained same in controlled but mortality of larvae found in different test tube based on concentration used. LC50 was calculated.
MTT assay for of viscous ethyl acetate leaf extract of Annona muricata
(VEAM) and powdered ethyl acetate leaf extract of Annona muricata (PEAM):
MTT assay test is one method used in the cytotoxic test. Cytotoxic test is used to determine the IC50 value parameter. IC50 value indicates the concentration that resulted in inhibition of cell proliferation by 50% and demonstrate the potential toxicity of a compound to sel. This value is a benchmark to test cell kinetics observation. The principle of the method of MTT is a yellow tetrazolium salt reduction of MTT (3- (4,5-dimetiltiazol-2-yl) -2.5-difeniltetrazolium bromide) reductase system. Succinate tetrazolium included in the respiratory chain in the mitochondria of living cells to form purple formazan crystals and not water soluble. Reagent addition stopper (is detergenik) will dissolve these colored crystals which are then measured absorbance using an ELISA reader. Purple color intensity is proportional to the number of living cells. So if the greater intensity of the color purple, the mean number of living cells more (Dewi, 2012). Procedure: 1. Plate cells (104 – 106 cells) in 200 ml PBS in 96-well (flat bottom). 2. Add 20 ml of MTT solution, mix well. 3. Incubate for 4h in 37C in dark. 4. Remove aliquot for analysis; add 200 ml acidic isopropanol and mix well. 5. Incubate additional 1h in 37C in dark. 6. Read plate in ELISA Reader – measure OD in 570 nm (background wavelength is 630nm).
Preformulation evaluation:
Fourier-transform infrared (FT-IR)
FT-IR was used to identify the functional group of the active components based on the peak value in the region of infrared radiation. All spectra were obtained with the aid of an OMNI-sampler attenuated total reflectance (ATR) accessory on a FTIR spectrophotometer. A small amount of powdered leaves was placed directly on the germanium piece of the infrared spectrometer with constant pressure applied and data of infrared absorbance, collected over the wave number ranged from 4000 cm–1 to 675 cm–1 and computerized for analyses). The melting points were measured on a Leica Gallen III Kofter micro melting point apparatus. The optical rotations were measured in CHCl3 with Perkin Elmer 241 polarimeter. The ultraviolet (UV) spectra were recorded on Shimadzu UV 1601PC spectrophotometer.
Formulation of sustained release tablet containing ethyl acetate leaf extract of Annona muricata.
Preparation of granules containing ethyl acetate leaf extract of Annona muricata with suitable excipients.
Tablets extracts from ethyl acetate fraction soursop leaf is made with a dose of 400 mg, then formulated using wet granulation method. Wet granulation method was selected since the nature acetogenin relatively was stable in the presence of moisture and to obtain a good flowability to be easily compressed.
The ethyl acetate leaf extract of Annona muricata and other ingredients in each formula were weighed, ground and screened through sieve number 80 separately. All the ingredients (Table No. 4.3) were mixed together except talc and magnesium stearate milled in a pestle mortar and sieved again through sieve number 80. The material was mixed with the acacia gum solution, which was added slowly. After mixing, the powder mass was screened through sieve number 18 to get the granules and dried at 35°C in vacuum dryer.
Table No. 4.3: Composition of Sustained release tablet containing Annona muricata leaf extract (PEAM)
|
Ingredients |
Formulations (mg) |
|||||
|
F1 |
F2 |
F3 |
F4 |
F5 |
F6 |
|
|
Plant extract |
300 |
300 |
300 |
300 |
300 |
300 |
|
Carbopol |
20 |
30 |
40 |
- |
- |
- |
|
Ethyl cellulose |
- |
- |
- |
20 |
30 |
40 |
|
Microcrystalline cellulose |
40 |
40 |
40 |
40 |
40 |
40 |
|
Tribasic calcium phosphate |
30 |
20 |
1 |
30 |
20 |
10 |
|
PEG 4000 |
10 |
10 |
10 |
10 |
10 |
10 |
|
Methyl paraben (%) |
0.1 |
0.1 |
0.1 |
0.1 |
0.1 |
0.1 |
|
Weight per tablet |
400 |
400 |
400 |
400 |
400 |
400 |
Evaluation of Precompression parameters of granules containing ethyl acetate leaf extract of Annona muricata:
Angle of Repose:
The flow property was determined by measuring the Angle of Repose. In order to determine the flow property, the Angle of Repose was determined. It is the maximum angle that can be obtained between the free standing surface of a powder heap and the horizontal.
Angle of repose= tan-¹ (h/r)
Where,
h = height of a pile (2 cm)
r = radius of pile base.
Procedure: 20gms of the sample was taken. The sample was passed through the funnel slowly to form a heap. The height of the powder heap formed was measured. The circumference formed was drawn with a pencil on the graph paper. The radius was measured and the angle of repose was determined. This was repeated three times for a sample.
Bulk density:
Bulk density is ratio of given mass of powder and its bulk volume. Bulk density was determined by measuring the volume of known mass of powder sample that has been passed through the screen in to graduated cylinder or through volume measuring apparatus in to cup.
Bulk density = M / V0
Where M= mass of the powder;
V0=bulk volume of the powder.
Limits: It has been stated that the bulk density values having less than 1.2 g/cm3 indicates good packing and values greater than 1.5 g/cm3 indicates poor packing.
Tapped density:
A known quantity of powder was transferred to a graduated cylinder and volume V0 was noted. The cylinder fixed to a density determination apparatus, tapped for 500 times then reading was observed. The density is achieved by mechanically tapped by a measuring cylinder containing the powder sample. After observing the initial volume the cylinder is mechanically tapped and volume reading were taken until little further volume changes is observed.
Tap density = M / Vr
Where M = mass of the powder,
Vr = final tapping volume of the powder.
Compressibility index and Hausner ratio:
The compressibility index and hausner ratio may be calculated using measured values of bulk density and tapped density as follows:
Compressibility index = 100 × tapped density / bulk density.
Hausner ratio = tapped density / bulk density.
|
S. No |
Flow properties |
Angle of repose(θ) |
Compressibility Index (%) |
Hausner ratio |
|
1. |
Excellent |
25-30 |
<10 |
1.00-1.11 |
|
2. |
Good |
31-35 |
11-15 |
1.12-1.18 |
|
3. |
Fair |
36-40 |
16-20 |
1.19-1.25 |
|
4. |
Passable |
41-45 |
21-25 |
1.26-1.34 |
|
5. |
Poor |
46-55 |
26-31 |
1.35-1.45 |
|
6. |
Very poor |
56-65 |
32-37 |
1.46-1.59 |
|
7. |
Very very poor |
> 66 |
>38 |
>1.6 |
Compression of sustained release tablet containing ethyl acetate leaf extract of Annona muricata (PEAM).
The dried granules containing ethyl acetate leaf extract of Annona muricata (PEAM) was were compressed to 400 mg tablet on hand rotating single punch tablet presses using 11 X 8 mm punch set with appropriate compression pressure. The granules were mixed with talc and magnesium stearate before punching and the die cavity was adjusted for required weight and the granules were punched to tablets.
Evaluation of postcompression parameters of sustained release tablet containing ethyl acetate leaf extract of Annona muricata (PEAM).
The quantitative evaluation and assessment of a tablets chemical, physical and bioavailability properties are important in the design of tablets and to monitor product quality. There are various standards that have been set in the various pharmacopoeias regarding the quality of pharmaceutical tablets. These include the diameter, size, shape, thickness, weight, hardness, Friability and invitro-dissolution characters.
Hardness:
Hardness of the tablet was determined by using the Monsanto hardness tester. The lower plunger was placed in contact with the tablet and a zero reading was taken. The plunger was then forced against a spring by turning a threaded bolt until the tablet fractured. As the spring was compressed a pointer rides along a gauge in the barrel to indicate the force.
Thickness:
Control of physical dimensions of the tablets such as size and thickness is essential for consumer acceptance and tablet-tablet uniformity. The diameter size and punch size of tablets depends on the die and punches selected for making the tablets. The thickness of tablet is measured by Vernier Callipers scale. The thickness of the tablet related to the tablet hardness and can be used an initial control parameter. Tablet thickness should be controlled within a ±5%. In addition thickness must be controlled to facilitate packaging.
Friability:
Friction and shock are the forces that most often cause tablets to chip, cap or break. The friability test is closely related to tablet hardness and designed to evaluate the ability of the tablet to withstand abrasion in packaging, handling and shipping. It is usually measured by the use of the Roche friabilator.
Method:
A number of tablets are weighed and placed in the apparatus where they are exposed to rolling and repeated shocks as they fall 6 inches in each turn within the apparatus. After four minutes of this treatment or 100 revolutions, the tablets are weighed and the weight compared with the initial weight. The loss due to abrasion is a measure of the tablet friability. The value is expressed as a percentage. A maximum weight loss of not more than 1% of the weight of the tablets being tested during the friability test is considered generally acceptable and any broken or smashed tablets are not picked.
The percentage friability was determined by the formula:
% friability = (W1-W2) / W1 X 100
W1 = Weight of tablets before test
W2 = Weight of tablets after test
High performance liquid chromatography of ethyl acetate leaf extract of Annona muricata (PEAM) and the optimized batch of sustained release tablet containing ethyl acetate leaf extract of Annona muricata (PEAM):
Acetogenins qualitative screening analysis was carried out using HPLC with operating conditions that matched those reported by Yang [17] with slightly modification. A water HPLC system (Milford, MA, USA) that consisted of double pump and system controller (Model 2695), an auto-sampler and photo-diode array detector (Model 966) was used. The column configuration consisted of a reversed phase column (4.6 × 150 mm, 4 µm; Phenomemex, Torrance, CA, USA). Detection wavelength was set at 220 nm. The mobile phase consisted of A (acetonitirile) and B (deionized water), using a linear gradient: 0-40 min (85%), and 40-60 min (85-95% A). The flow rate was 1.0 ml/min. The column temperature was maintained at 30°C. High Performance of Liquid Chromatography (HPLC) was used for the screening of the acetogenins compounds present in the ethyl acetate leaf extract of Annona muricata (PEAM) and the optimized batch of sustained release tablet containing ethyl acetate leaf extract of Annona muricata (PEAM).
Stability studies:
Physical stability studies were conducted according to International Conference on Harmonization (ICH) guidelines. The optimized formulation was enclosed in polyethylene bottle and placed in a desicator containing saturated sodium chloride solution, which provided 75% ± 5% RH. The desicator was stored at 40°C ± 2°C for 3 months. At predetermined time intervals, the tablets were examined for hardness, friability, thickness.
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT editor-in-chief@pharmatutor.org
Subscribe to Pharmatutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE
Preparation of powder of leaves of Annona muricata: The Annona muricata leaves was a free flowing powder (Figure 5.1) which was used for further extraction.

Figure 5.1: Annona muricata dried leaf powder
Ethyl acetate extraction of Annona muricata leaves:
The ethyl acetate extraction of Annona muricata leaves were extracted using maceration method. 1000 g of Annona Muricata leaves dried gave an yield of 130 g of the extract in highly viscous form as shown in Figure.

Figure 5.2: Ethyl acetate leaf extract of Annona muricata (VEAM)
Preparation of free flowing powder of ethyl acetate extract leaves of Annona muricata:
The prepared ethyl acetate extract was highly viscous. Various combinations of excipients were used to prepare granules for compression of tablets. Unfortunately, it was quite difficult to solubilize or incorporate the viscous ethyl acetate extract. Hence an attempt was made to convert the highly viscous ethyl acetate extract containing Annona muricata leaves as semi free flowing powder or free flowing powder form. The ethyl acetate extract containing Annona muricata leaves was extracted with methanol several times and the semi free flowing powder form ( Figure xx) of ethyl acetate leaf extract of Annona muricata (PEAM).

Figure 5.3: Powder form of ethyl acetate leaf extract of Annona muricata (PEAM)
Phytochemical screening of VEAM and PEAM:
The results of phytochemical screening tests of the viscous ethyl acetate leaf extract of Annona muricata (VEAM) and powdered ethyl acetate leaf extract of Annona muricata (PEAM) for qualitative analysis of primary and secondary metabolites are as shown in the Table No. 5.1.
Table No. 5.1: Phytochemical screening of the VEAM and PEAM:
|
S.No |
Phytochemicals |
Test Performed |
VEAM |
PEAM |
|---|---|---|---|---|
| 1 |
Alkaloid |
Mayer test |
- |
+ |
| 2 |
Saponin |
Froth test |
+ |
+ |
| 3 |
Steroid |
Liberman |
+ |
+ |
| 4 |
Terpenoid |
Salkovaski test |
+ |
- |
| 5 |
Tannin and Phenolic compound |
Ferric chloride test |
- |
- |
| 6 |
Protein and Amino acids |
Ninhydin test |
+ |
+ |
| 7 |
Carbohydrate |
Benedict test |
+ |
+ |
| 8 |
Anthraquinone glycoside |
Ammonia test |
+ |
- |
| 9 |
Cardiac glycoside |
Keller killani test |
+ |
+ |
| 10 |
Flavonoid |
Shinoda test |
+ |
+ |
Biological studies of viscous ethyl acetate leaf extract of Annona muricata (VEAM) and powdered ethyl acetate leaf extract of Annona muricata (PEAM): 5.3.1 Brine shrimp lethality assay (BSLA):
Samples are diluted serially 1 to 1000µg/ml and performed in triplets for individual sample to get mean average valve. Individual test tubes contained 3ml of distilled water and 1.5ml of Brine solution (72gm of NaCL) IN 500ml with distilled water. Later 0.5ml of samples of different dilution were added along with control which contain 0.5ml 1N NaoH instead of sample. It was kept over 24hours under lamp source for observation (Figure 5.4). After 24hours larvae retained same in controlled but mortality of larvae found in different test tube based on concentration used. LC50 was calculated as shown in the Table. No 5.2.

Figure 5.4: Shrimp nauplii
Table No. 5.2: The number of shrimp nauplii that survived after treating with the three plant extracts and the percentage mortality
|
Plant Extracts |
Concentration (μg/mL) |
Number of Surviving Nauplii After 24 h |
Total Number of Survivors |
% Mortality |
||
|
I |
II |
III |
||||
|
VEAM |
1 |
7 |
8 |
8 |
23 |
23 |
|
10 |
5 |
6 |
5 |
16 |
47 |
|
|
100 |
4 |
5 |
5 |
14 |
53 |
|
|
1000 |
0 |
0 |
0 |
0 |
100 |
|
|
PEAM |
1 |
7 |
7 |
8 |
22 |
27 |
|
10 |
4 |
6 |
5 |
15 |
50 |
|
|
100 |
0 |
0 |
0 |
0 |
100 |
|
|
1000 |
0 |
0 |
0 |
0 |
100 |
|
MTT assay for of viscous ethyl acetate leaf extract of Annona muricata (VEAM) and powdered ethyl acetate leaf extract of Annona muricata (PEAM):
The Table No. 5.3 indicated the IC 50, the concentration that resulted in inhibition of cell proliferation by 50% and demonstrate the potential toxicity of the viscous ethyl acetate leaf extract of Annona muricata (VEAM) and powdered ethyl acetate leaf extract of Annona muricata (PEAM)
Table No. 5.3: MTT assay for VEAM and PEAM
|
S.No |
Cancer Cell
|
IC 50 (ppm) |
|
|
VEAM |
PEAM |
||
|
|
A549 (lung cells) |
54,66 |
52,12 |
|
|
HeLa (cervical cancer cells) |
33,98 |
30,62 |
|
|
MCF-7 (breast cells) |
32,90 |
29,98 |
Preformulation evaluation:
Fourier-transform infrared (FT-IR)
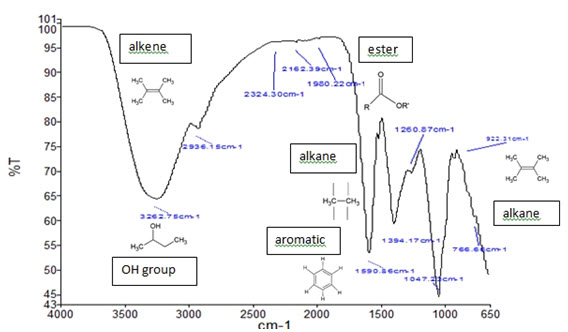
Figure 5.5: FTIR for the mixture of PEAM and other excipents
5.5 Formulation of sustained release tablet containing ethyl acetate leaf extract of Annona muricata.
5.5.1 Preparation of granules containing ethyl acetate leaf extract of Annona muricata with suitable excipients.

Figure 5.6: Granules containing PEAM
5.6 Evaluation of Precompression parameters of granules containing ethyl acetate leaf extract of Annona muricata:
Table No. 5.4: Precompression parameters of SR tablets PEAM
|
Formulation Code |
Angle of Repose (Ѳ) |
Bulk Density (g/ml) |
Tapped Density (g/ml) |
Compressibility Index (%) |
Hausner's Ratio |
|
F1 |
29.28° |
0.612±0.02 |
0.689±0.04 |
20.43±0.05 |
1.13±0.02 |
|
F2 |
27.63° |
0.675±0.02 |
0.854±0.11 |
21.07±0.02 |
1.19±0.08 |
|
F3 |
31.70° |
0.740±0.02 |
0.952±0.01 |
25.00±0.07 |
1.24±0.06 |
|
F4 |
31.28° |
0.714±0.02 |
0.909±0.23 |
22.21±0.05 |
1.20±0.05 |
|
F5 |
33.70° |
0.699±0.01 |
0.952±0.53 |
22.21±0.08 |
1.24±0.04 |
|
F6 |
31.70° |
0.751±0.05 |
0.909±0.01 |
23.25±0.03 |
1.24±0.06 |
5.7 Compression of sustained release tablet containing ethyl acetate leaf extract of Annona muricata (PEAM).

Figure 5.7: Sustained release tablet containing PEAM
5.8 Evaluation of postcompression parameters of sustained release tablet containing ethyl acetate leaf extract of Annona muricata (PEAM)
Table No. 5.5: Post-compressional parameters of SR tablets PEAM
|
Formulation Code |
Average weight (mg) |
Thickness (mm) |
Hardness (kg/cm2) |
% w/w Friability |
|
F1 |
419±0.25 |
7.43±0.30 |
3.8±0.418 |
0.163±0.02 |
|
F2 |
412±0.09 |
7.3±0.12 |
4.5±0.09 |
0.092±0.01 |
|
F3 |
418±0.12 |
7.44±0.13 |
3.9±0.041 |
0.144±0.05 |
|
F4 |
418±0.15 |
7.44±0.13 |
3.7±0.916 |
0.145±0.06 |
|
F5 |
417±0.15 |
7.43±0.30 |
3.8±0.568 |
0.145±0.06 |
|
F6 |
418±0.15 |
7.41±0.12 |
3.8±0.483 |
0.144±0.06 |
High performance liquid chromatography of ethyl acetate leaf extract of Annona muricata (PEAM) and the optimized batch of sustained release tablet containing ethyl acetate leaf extract of Annona muricata (PEAM):

Figure 5.8: HPLC of F2 batch of Sustained release tablet containing PEAM
Stability studies:
Table No. 5.6: Post-compressional parameters of F2 batch of SR tablets PEAM
|
Formulation Code |
Average weight (mg) |
Thickness (mm) |
Hardness (kg/cm2) |
% w/w Friability |
|
F2 |
412±0.13 |
7.3±0.35 |
4.5±0.19 |
0.093±0.03 |
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT editor-in-chief@pharmatutor.org
Subscribe to Pharmatutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASEDISCUSSION
Preparation of powder of leaves of Annona muricata:
The Annona muricata leaf was a free flowing powder (Figure 5.1) which was used for further extraction.
Ethyl acetate extraction of Annona muricata leaves:
The ethyl acetate extraction of Annona muricata leaves were extracted using maceration method. 1000 g of Annona Muricata leaves dried gave a yield of 130 g of the extract in highly viscous form as shown in Figure 5.2.
Preparation of free flowing powder of ethyl acetate extract leaves of Annona muricata:
The prepared ethyl acetate extract was highly viscous. Various combinations of excipients were used to prepare granules for compression of tablets. Unfortunately, it was quite difficult to solubilize or incorporate the viscous ethyl acetate extract. Hence an attempt was made to convert the highly viscous ethyl acetate extract containing Annona muricata leaves as semi free flowing powder or free flowing powder form. The ethyl acetate extract containing Annona muricata leaves was extracted with methanol several times and the semi free flowing powder form of ethyl acetate leaf extract of Annona muricata (PEAM) as shown in Figure 5.3.
Phytochemical screening of VEAM and PEAM:
VEAM showed the presence of saponin, steroid, terpenoid, phenolic compound, protein & aminoacids, carbohydrates, antraquinine and cardiac glycosides. Whereas PEAM contained alkaloids, saponin, steroid, phenolic compound, protein & aminoacids, carbohydrates and cardiac glycosides. terpenoids and anthraquinones were lost in PEAM which were found in VEAM as shown in Table No. 5.1.
Biological studies of viscous ethyl acetate leaf extract of Annona muricata (VEAM) and powdered ethyl acetate leaf extract of Annona muricata (PEAM):
Brine shrimp lethality assay (BSLA): The percentage mortality rate of VEAM showed 23, 47, 53 and 100 respectively for the concentration 1, 10, 100 and 1000 μg/mL. The percentage mortality rate of PEAM showed 27, 50, 100 and 100 respectively for the concentration 1, 10, 100 and 1000 μg/mL. The potent activity of PEAM was found to be more compared to VEAM as shown in Table No. 5.2.
Since VEAM was highly viscous and was unable to convert into granules, VEAM was extracted with alcohol to get a powder form i.e PEAM. It was very essential to check the activity to be the same or more, but not lost. From the above data comparison, it was evident that there was no loss of phytoconstituents especially which is responsible for anticancer activity. Brine shrimp lethality assay (BSLA) is a biological assay to find out whether the sample contained any biological activity and not specific about the activity. The overall conclusion of this BSLA was that both the samples had some biological activity and moreover even during the conversion of VEAM to PEAM, there was no loss of activity. Hence PEAM was subjected to further formulation development.
MTT assay for of viscous ethyl acetate leaf extract of Annona muricata (VEAM) and powdered ethyl acetate leaf extract of Annona muricata (PEAM):
The IC50 of VEAM for A549 (lung cells), HeLa (cervical cancer cells) and MCF-7 (breast cells) were found to be 54,66, 33,98 and 32,90 respectively. The IC50 of PEAM for A549 (lung cells), HeLa (cervical cancer cells) and MCF-7 (breast cells) were found to be 52,12, 30,62 and 29,98 ppm respectively as shown in Table No. 5.3.
The test results showed that the IC50 of VEAM for A549 (lung cells) being more than 50 ppm was not classified as active as anticancer. But HeLa (cervical cancer cells) and MCF-7 (breast cells) were found to be below 33 ppm and hence was approaching the threshold for active category.
The test results showed that the IC50 of PEAM for A549 (lung cells) being more than 50 ppm was not classified as active as anticancer. But HeLa (cervical cancer cells) and MCF-7 (breast cells) were found to be below 30 ppm and hence was approaching the threshold for active category. The overall conclusion of this BSLA was that both the samples had some biological activity and moreover even during the conversion of VEAM to PEAM, there was no loss of activity. Hence PEAM was subjected to further formulation development.
Preformulation evaluation:
Fourier-transform infrared (FT-IR)
Broad peak at 3262.75 cm-1 = OH groups compared to spectra of liquid form of sample 3327.21 cm-1 = OH groups, 2936.15 cm-1 = CH2 and CH alkenes groups (Figure 6a), bend peak at 1394.17 cm-1 = CH3 alkane groups (Figure 6a), 1261 cm-1 = COC ester group. As expected, the peak for OH groups remaining existed without the presence of solvent (deuterium oxide also known as water) confirming the peaks were from the sample as shown in Figure 5.5. The IR spectrum revealed the presence of broad and strong adsorption of hydroxyl functional groups at 3262.75 cm-1. From the data it was found that the PEAM was compatible with all the ingredients used for the formulation development.
Formulation of sustained release tablet containing ethyl acetate leaf extract of Annona muricata.
Preparation of granules containing ethyl acetate leaf extract of Annona muricata with suitable excipients.
The granules containing ethyl acetate leaf extract of Annona muricata was prepared with the excipients as shown in Figure 5.6. The sieving the mass after wet granulation was highly difficult. On applying more pressure during the sieving resulted in liquefaction. Hence the smooth handling was highly required till the granules are dried. It was also very difficult to standardize the drying temperature of the granules at the final stage. All the above mentioned difficulties were faced due to the wet granulation. Direct compression was not considered since it was mentioned in most of the literature that the phytochemical angiotensin was lost during direct compression.
Evaluation of Precompression parameters of granules containing ethyl acetate leaf extract of Annona muricata:
The powder blends containing Lansoprazole prepared for trilayer compression of tablets were evaluated for their flow properties as shown in Table No. 5.4. The value of bulk density and tapped density (0.612±0.02 to 0.751±0.05 g/ml and 0689±0.04. to 0.952±0.56 g/ml) indicated good packing characteristics. Carr’s index was found to be in the range of 20.43±0.05 to 25.00±0.07. The angle of repose was in the range of 27.63° to 33.70°. The compressibility index of the powder blend of F2 was less than 21 indicating good flow properties which were further confirmed by determining the angle of repose which was in the range of 25-30° indicating good flow properties.
Compression of sustained release tablet containing ethyl acetate leaf extract of Annona muricata (PEAM).
During the compression, there was much difficulty to set the hardness since Batch F2 only was found to be above 4 kg/cm2. However all the batches showed a nominal compression of into SR tablets as shown in Figure 5.7.
Evaluation of postcompression parameters of sustained release tablet containing ethyl acetate leaf extract of Annona muricata (PEAM):
The weight ranges of all the batches were found to be 412±0.09 to 419±0.25 mg. The batch F2 was having minimum weight difference 412±0.09 mg as shown in Table No. 5.5. The thickness of the tablet was ranging from 7.30±0.12mm to 7.44±0.13 mm. The hardness of F1, F2, F3, F4, F5 and F6 were found to be 3.8±0.418, 4.5±0.09, 3.9±0.041, 3.7±0.916, 3.8±0.568 and 3.8±0.483 kg/cm2respectively. Batch F2 showed nominal hardness. The friability was in the range of 0.092±0.01 to 0.163±0.02 %w/w. from the above data, it was confirmed that the batch F2 was chosen as the optimized batch for having good hardness, and friability. Hence the batch F2 was selected for HPLC studies and stability studies.
High performance liquid chromatography of ethyl acetate leaf extract of Annona muricata (PEAM) and the optimized batch of sustained release tablet containing ethyl acetate leaf extract of Annona muricata (PEAM):
The resolution between any two compounds in the sample was greater than 1.5. Other compounds in the sample did not interfere with analysis of the six acetogenins compounds analytes.
The chromatographic peaks were identified by comparing their retention time with that of each reference compounds reported from the literatures. The retention times of the compound in the extract remained the same and the coefficient of variation for retention time was less than 1% which made a confirmation of the presence of acetogenins in the extract.
Stability studies:
The best batch selected i.e F2 formulation was subjected to stability studies for 3 months to evaluate its stability and the integrity of the dosage form. The tablets were characterized for the hardness, friability, thickness and average weight. during the study stability period. There was no significant change observed in the friability, hardness at 40 °C/75 % RH for 6 months as shown in Table No. 5.6. From these results, it was concluded that optimized formulation was stable and retained their original properties with minor differences.
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT editor-in-chief@pharmatutor.org
Subscribe to Pharmatutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASESUMMARY
The Annona muricata leaf was a free flowing powder (Figure 5.1) which was used for further extraction. The ethyl acetate extraction of Annona muricata leaves were extracted using maceration method. 1000 g of Annona Muricata leaves dried gave a yield of 130 g of the extract in highly viscous form. The prepared ethyl acetate extract was highly viscous. Various combinations of excipients were used to prepare granules for compression of tablets. Unfortunately, it was quite difficult to solubilize or incorporate the viscous ethyl acetate extract. Hence an attempt was made to convert the highly viscous ethyl acetate extract containing Annona muricata leaves as semi free flowing powder or free flowing powder form. The ethyl acetate extract containing Annona muricata leaves was extracted with methanol several times and the semi free flowing powder form of ethyl acetate leaf extract of Annona muricata (PEAM).
VEAM showed the presence of saponin, steroid, terpenoid, phenolic compound, protein & aminoacids, carbohydrates, antraquinine and cardiac glycosides. Whereas PEAM contained alkaloids, saponin, steroid, phenolic compound, protein & aminoacids, carbohydrates and cardiac glycosides. terpenoids and anthraquinones were lost in PEAM which were found in VEAM.
The percentage mortality rate of VEAM showed 23, 47, 53 and 100 respectively for the concentration 1, 10, 100 and 1000 μg/mL. The percentage mortality rate of PEAM showed 27, 50, 100 and 100 respectively for the concentration 1, 10, 100 and 1000 μg/mL. The potent activity of PEAM was found to be more compared to VEAM. Since VEAM was highly viscous and was unable to convert into granules, VEAM was extracted with alcohol to get a powder form i.e PEAM. It was very essential to check the activity to be the same or more, but not lost. From the above data comparison, it was evident that there was no loss of phytoconstituents especially which is responsible for anticancer activity. Brine shrimp lethality assay (BSLA) is a biological assay to find out whether the sample contained any biological activity and not specific about the activity. The overall conclusion of this BSLA was that both the samples had some biological activity and moreover even during the conversion of VEAM to PEAM, there was no loss of activity. Hence PEAM was subjected to further formulation development.
The IC50 of VEAM for A549 (lung cells), HeLa (cervical cancer cells) and MCF-7 (breast cells) were found to be 54,66, 33,98 and 32,90 respectively. The IC50 of PEAM for A549 (lung cells), HeLa (cervical cancer cells) and MCF-7 (breast cells) were found to be 52,12, 30,62 and 29,98 ppm respectively. The test results showed that the IC50 of VEAM for A549 (lung cells) being more than 50 ppm was not classified as active as anticancer. But HeLa (cervical cancer cells) and MCF-7 (breast cells) were found to be below 33 ppm and hence was approaching the threshold for active category.
The test results showed that the IC50 of PEAM for A549 (lung cells) being more than 50 ppm was not classified as active as anticancer. But HeLa (cervical cancer cells) and MCF-7 (breast cells) were found to be below 30 ppm and hence was approaching the threshold for active category. The overall conclusion of this BSLA was that both the samples had some biological activity and moreover even during the conversion of VEAM to PEAM, there was no loss of activity. Hence PEAM was subjected to further formulation development.
Fourier-transform infrared (FT-IR) was performed as a part of preformulation studies.
Broad peak at 3262.75 cm-1 = OH groups compared to spectra of liquid form of sample 3327.21 cm-1 = OH groups, 2936.15 cm-1 = CH2 and CH alkenes groups (Figure 6a), bend peak at 1394.17 cm-1 = CH3 alkane groups (Figure 6a), 1261 cm-1 = COC ester group. As expected, the peak for OH groups remaining existed without the presence of solvent (deuterium oxide also known as water) confirming the peaks were from the sample as shown in Figure 5.5. The IR spectrum revealed the presence of broad and strong adsorption of hydroxyl functional groups at 3262.75 cm-1. From the data it was found that the PEAM was compatible with all the ingredients used for the formulation development
The sustained release tablet containing ethyl acetate leaf extract of Annona muricata was formulated. Initially granules containing ethyl acetate leaf extract of Annona muricata with suitable excipients were prepared by wet granulation method. The granules containing ethyl acetate leaf extract of Annona muricata was prepared with the excipients as shown in Figure 5.6. The sieving the mass after wet granulation was highly difficult. On applying more pressure during the sieving resulted in liquefaction. Hence the smooth handling was highly required till the granules are dried. It was also very difficult to standardize the drying temperature of the granules at the final stage. All the above mentioned difficulties were faced due to the wet granulation. Direct compression was not considered since it was mentioned in most of the literature that the phytochemical angiotensin was lost during direct compression.
The prepared granules were evaluated for pre compression parameters. The powder blends containing Lansoprazole prepared for trilayer compression of tablets were evaluated for their flow properties. The value of bulk density and tapped density (0.612±0.02 to 0.751±0.05 g/ml and 0689±0.04. to 0.952±0.56 g/ml) indicated good packing characteristics. Carr’s index was found to be in the range of 20.43±0.05 to 25.00±0.07. The angle of repose was in the range of 27.63° to 33.70°. The compressibility index of the powder blend of F2 was less than 21 indicating good flow properties which were further confirmed by determining the angle of repose which was in the range of 25-30° indicating good flow properties.
The granules which were evaluated for their pre compression parameters were compression as sustained release tablet containing ethyl acetate leaf extract of Annona muricata (PEAM). During the compression, there was much difficulty to set the hardness since Batch F2 only was found to be above 4 kg/cm2. However all the batches showed a nominal compression of into SR tablets.
The compressed tablets were evalted for their post compression parameters. The weight ranges of all the batches were found to be 412±0.09 to 419±0.25 mg. The batch F2 was having minimum weight difference 412±0.09 mg. The thickness of the tablet was ranging from 7.30±0.12mm to 7.44±0.13 mm. The hardness of F1, F2, F3, F4, F5 and F6 were found to be 3.8±0.418, 4.5±0.09, 3.9±0.041, 3.7±0.916, 3.8±0.568 and 3.8±0.483 kg/cm2respectively. Batch F2 showed nominal hardness. The friability was in the range of 0.092±0.01 to 0.163±0.02 %w/w. from the above data, it was confirmed that the batch F2 was chosen as the optimized batch for having good hardness, and friability. Hence the batch F2 was selected for HPLC studies and stability studies.
High performance liquid chromatography of ethyl acetate leaf extract of Annona muricata (PEAM) was conducted for the optimized batch of sustained release tablet containing ethyl acetate leaf extract of Annona muricata (PEAM). The resolution between any two compounds in the sample was greater than 1.5. Other compounds in the sample did not interfere with analysis of the six acetogenins compounds analytes.
The chromatographic peaks were identified by comparing their retention time with that of each reference compounds reported from the literatures. The retention times of the compound in the extract remained the same and the coefficient of variation for retention time was less than 1% which made a confirmation of the presence of acetogenins in the extract.
The stability study was conducted for the optimized batch F2. The best batch selected i.e F2 formulation was subjected to stability studies for 3 months to evaluate its stability and the integrity of the dosage form. The tablets were characterized for the hardness, friability, thickness and average weight. during the study stability period. There was no significant change observed in the friability, hardness at 40 °C/75 % RH for 6 months as shown in Table No. 5.6. From these results, it was concluded that optimized formulation was stable and retained their original properties with minor differences.
CONCLUSION
Annona muricata is also called as “Graviola” (SOURSOP). Annona muricata L. is a naturally occurring plant traditionally used to treat various ailments including cancer.
The therapeutic dosage of Annona muricata leaf (which offers just as high of an amount of acetogenins as the root and almost as much as the seed) is reported to be 2-3g taken three or four times daily. The traditional preparation of Annona muricata leaf reported to be 2g, three times daily in capsule or tablets.
Since the dose of the traditional preparation was very high and a conventional tablet formulation was prepared by Elisya et; al, an attempt made to formulate the sustained release tablet dosage form containing powder form of ethyl acetate leaf extract of Annona muricata (PEAM) would reduce the dose, improve therapeutic efficacy with a standardized formulation and improve patient compliance.
The prepared sustained release tablet dosage form containing powder form of ethyl acetate leaf extract of Annona muricata (PEAM) would
* Reduce the size of the dose of Annona muricata for better therapeutic action.
* Reduce the frequency of dosing for anticancer activity.
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT editor-in-chief@pharmatutor.org
Subscribe to Pharmatutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASEREFERENCES:
1. Moghadamtousi S.Z., Goh B.H., Chan C.K., Shabab T., Kadir H.A. Biological activities and phytochemicals of Swietenia macrophylla king. Molecules. 2013;18:10465–10483.
2. Moghadamtousi S.Z., Kamarudin M.N.A., Chan C.K., Goh B.H., Kadir H.A. Phytochemistry and biology of Loranthus parasiticus merr, a commonly used herbal medicine. Am. J. Chin. Med. 2014;42:23–35.
3. Mishra S., Ahmad S., Kumar N., Sharma B.K. Annona muricata (the cancer killer): A review. Glob. J. Pharm. Res. 2013;2:1613–1618.
4. Leboeuf M., Cavé A., Bhaumik P., Mukherjee B., Mukherjee R. The phytochemistry of the annonaceae. Phytochemistry. 1980;21:2783–2813.
5. Adewole S.O., Caxton-Martins E.A. Morphological changes and hypoglycemic effects of Annona muricata Linn. (Annonaceae) leaf aqueous extract on pancreatic B-cells of streptozotocin-treated diabetic rats. Afr. J. Biomed. Res. 2006;9:173–187.
6. De Souza R., Benassi E., da Silva R.R., Afonso S., Scarminio I.S. Enhanced extraction yields and mobile phase separations by solvent mixtures for the analysis of metabolites in Annona muricata L. Leaves. J. Sep. Sci. 2009;32:4176–4185.
7. De Sousa O.V., Vieira G.D.-V., de Pinho J.D.J.R., Yamamoto C.H., Alves M.S. Antinociceptive and anti-inflammatory activities of the ethanol extract of Annona muricata L. leaves in animal models. Int. J. Mol. Sci. 2010;11:2067–2078.
8. Adewole S., Ojewole J. Protective effects of Annona muricata linn.(annonaceae) leaf aqueous extract on serum lipid profiles and oxidative stress in hepatocytes of streptozotocin-treated diabetic rats. Afr. J. Tradit. Complement. Altern. Med. 2009;6:30–41.
9. Watt J.M., Breyer-Bnodwijk M. The Medicinal and Poisonous Plants of Southern and Eastern Africa: Being an Account of Their Medicinal and Other Uses, Chemical Composition, Pharmacological Effects Aod Toricotogy in Man and Annimal. Lívingstone Ltd.; Edinburgh, UK; London, UK: 1962.
10. Ong H., Norzalina J. Malay herbal medicine in Gemencheh, Negri Sembilan, Malaysia. Fitoterapia. 1999;70:10–14.
11. Jaramillo-Flores M., Hernandez-Sanchez H. Thermal diffusivity of soursop (Annona muricata L.) pulp. J. Food Eng. 2000;46:139–143.
12. Wu F.-E., Gu Z.-M., Zeng L., Zhao G.-X., Zhang Y., McLaughlin J.L., Sastrodihardjo S. Two new cytotoxic monotetrahydrofuran annonaceous acetogenins, annomuricins a and b, from the leaves of Annona muricata. J. Nat. Prod. 1995;58:830–836.
13. Yang C., Gundala S.R., Mukkavilli R., Vangala S., Reid M.D., Aneja R. Synergistic interactions among flavonoids and acetogenins in graviola (Annona muricata) leaves confer protection against prostate cancer. Carcinogenesis. 2015;2015
14. Matsushige A., Matsunami K., Kotake Y., Otsuka H., Ohta S. Three new megastigmanes from the leaves of Annona muricata. J. Nat. Med. 2012;66:284–291.
15. Nawwar M., Ayoub N., Hussein S., Hashim A., El-Sharawy R., Wende K., Harms M., Lindequist U. Flavonol triglycoside and investigation of the antioxidant and cell stimulating activities of Annona muricata linn. Arch. Pharm. Res. 2012;35:761–767.
16. Jiménez V.M., Gruschwitz M., Schweiggert R.M., Carle R., Esquivel P. Identification of phenolic compounds in soursop (Annona muricata) pulp by high-performance liquid chromatography with diode array and electrospray ionization mass spectrometric detection. Food Res. Int. 2014;65:42–46.
17. Pélissier Y., Marion C., Kone D., Lamaty G., Menut C., Bessière J.-M. Volatile components of Annona muricata L. J. Essent. Oil Res. 1994;6:411–414.
18. Kossouoh C., Moudachirou M., Adjakidje V., Chalchat J.-C., Figuérédo G. Essential oil chemical composition of Annona muricata L. Leaves from benin. J. Essent. Oil Res. 2007;19:307–309.
19. Rupprecht J.K., Hui Y.-H., McLaughlin J.L. Annonaceous acetogenins: A review. J. Nat. Prod. 1990;53:237–278.
20. Gyamfi K., Sarfo D., Nyarko B., Akaho E., Serfor-Armah Y., Ampomah-Amoako E. Assessment of elemental content in the fruit of graviola plant, Annona muricata, from some selected communities in ghana by instrumental neutron activation analysis. Elixir Food Sci. 2011;41:5671–5675.
21. Hasrat J., Bruyne T.D., Backer J.P., Vauquelin G., Vlietinck A. Isoquinoline derivatives isolated from the fruit of Annona muricata as 5-HTergic 5-HT1A receptor agonists in rats: Unexploited antidepressive (lead) products. J. Pharm. Pharmacol. 1997;49:1145–1149.
22. Hasrat J., Pieters L., de Backer J.-P., Vauquelin G., Vlietinck A. Screening of medicinal plants from suriname for 5-HT1A ligands: Bioactive isoquinoline alkaloids from the fruit of Annona muricata. Phytomedicine. 1997;4:133–140.
23. Melot A., Fall D., Gleye C., Champy P. Apolar annonaceous acetogenins from the fruit pulp of Annona muricata. Molecules. 2009;14:4387–4395.
24. Ragasa C.Y., Soriano G., Torres O.B., Don M.-J., Shen C.-C. Acetogenins from Annona muricata. Pharmacog. J. 2012;4:32–37.
25. Sun S., Liu J., Kadouh H., Sun X., Zhou K. Three new anti-proliferative annonaceous acetogenins with mono-tetrahydrofuran ring from graviola fruit (Annona muricata) Bioorg. Med. Chem. Lett. 2014;24:2773–2776.
26. 26. Jaramillo M., Arango G., Gonzalez M., Robledo S., Velez I.D. Cytotoxicity and antileishmanial activity of Annona muricata pericarp. Fitoterapia. 2000;71:183–186.
27. Wu F.-E., Zeng L., Gu Z.-M., Zhao G.-X., Zhang Y., Schwedler J.T., McLaughlin J.L., Sastrodihardjo S. New bioactive monotetrahydrofuran annonaceous acetogenins, annomuricin c and muricatocin c, from the leaves of Annona muricata. J. Nat. Prod. 1995;58:909–915.
28. Kim G.-S., Zeng L., Alali F., Rogers L.L., Wu F.-E., McLaughlin J.L., Sastrodihardjo S. Two new mono-tetrahydrofuran ring acetogenins, annomuricin e and muricapentocin, from the leaves of Annona muricata. J. Nat. Prod. 1998;61:432–436.
29. Wu F.-E., Zhao G.-X., Zeng L., Zhang Y., Schwedler J.T., McLaughlin J.L., Sastrodihardjo S. Additional Bioactive Acetogenins, Annomutacin and (2, 4-trans and cis)-10R-Annonacin-A-ones, from the Leaves of Annona muricata. J. Nat. Prod. 1995;58:1430–1437.
30. Zeng L., Wu F.-E., McLaughlin J.L. Annohexocin, a novel mono-THF acetogenin with six hydroxyls, from Annona muricata (annonaceae) Bioorg. Med. Chem. Lett. 1995;5:1865–1868.
31. Wu F.-E., Zeng L., Gu Z.-M., Zhao G.-X., Zhang Y., Schwedler J.T., McLaughlin J.L., Sastrodihardjo S. Muricatocins a and b, two new bioactive monotetrahydrofuran annonaceous acetogenins from the leaves of Annona muricata. J. Nat. Prod. 1995;58:902–908.
32. Li D.-Y., Yu J.-G., Zhu J.-X., Yu D.-L., Luo X.-Z., Sun L., Yang S.-L. Annonaceous acetogenins of the seeds from Annona muricata. J. Asian Nat. Prod. Res. 2001;3:267–276.
33. Yu J.-G., Gui H.-Q., Luo X.-Z., Sun L. Murihexol, a linear acetogenin from Annona muricata. Phytochemistry. 1998;49:1689–1692.
34. Zeng L., Wu F.-E., Oberlies N.H., McLaughlin J.L., Sastrodihadjo S. Five new monotetrahydrofuran ring acetogenins from the leaves of Annona muricata. J. Nat. Prod. 1996;59:1035–1042.
35. Zeng L., Wu F.-E., Gu Z.-M., McLaughlin J.L. Murihexocins A and B, two novel mono-THF acetogenins with six hydroxyls, from Annona muricata (Annonaceae) Tetrahedron Lett. 1995;36:5291–5294.
36. Kim G.-S., Zeng L., Alali F., Rogers L.L., Wu F.-E., Sastrodihardjo S., McLaughlin J.L. Muricoreacin and murihexocin C, mono-tetrahydrofuran acetogenins, from the leaves of Annona muricata in honour of professor gh neil towers 75th birthday. Phytochemistry. 1998;49:565–571.
37. Liaw C.-C., Chang F.-R., Lin C.-Y., Chou C.-J., Chiu H.-F., Wu M.-J., Wu Y.-C. New cytotoxic monotetrahydrofuran annonaceous acetogenins from Annona muricata. J. Nat. Prod. 2002;65:470–475.
38. Chang F.-R., Liaw C.-C., Lin C.-Y., Chou C.-J., Chiu H.-F., Wu Y.-C. New adjacent bis-tetrahydrofuran annonaceous acetogenins from Annona muricata. Planta Med. 2003;69:241–246.
39. Fofana S., Ziyaev R., Abdusamatov A., Zakirov S.K. Alkaloids from Annona muricata leaves. Chem. Nat. Compd. 2011;47:321–321.
40. Matsushige A., Kotake Y., Matsunami K., Otsuka H., Ohta S., Takeda Y. Annonamine, a new aporphine alkaloid from the leaves of Annona muricata. Chem. Pharm. Bull. 2012;60:257–259.
41. Gleye C., Laurens A., Hocquemiller R., Cavé A., Laprévote O., Serani L. Isolation of montecristin, a key metabolite in biogenesis of acetogenins from Annona muricata and its structure elucidation by using tandem mass spectrometry. J. Org. Chem. 1997;62:510–513.
42. Gleye C., Laurens A., Hocquemiller R., Laprévote O., Serani L., Cavé A. Cohibins a and b, acetogenins from roots of Annona muricata. Phytochemistry. 1997;44:1541–1545.
43. Gleye C., Duret P., Laurens A., Hocquemiller R., Cavé A. cis-monotetrahydrofuran acetogenins from the roots of Annona muricata L. J. Nat. Prod. 1998;61:576–579.
44. Gleye C., Raynaud S., Hocquemiller R., Laurens A., Fourneau C., Serani L., Laprévote O., Roblot F., Leboeuf M., Fournet A. Muricadienin, muridienins and chatenaytrienins, the early precursors of annonaceous acetogenins. Phytochemistry. 1998;47:749–754.
45. Gleye C., Akendengue B., Laurens A., Hocquemiller R. Coronin from roots of annona muricata, a putative intermediate in acetogenin biosynthesis (1) Planta Med. 2001;67:570–572.
46. Gleye C., Laurens A., Laprévote O., Serani L., Hocquemiller R. Isolation and structure elucidation of sabadelin, an acetogenin from roots of Annona muricata. Phytochemistry. 1999;52:1403–1408.
47. Myint S.H., Laurens A., Hocquemiller R., Cavé A., Davoust D., Cortes D. Murisolin: A new cytotoxic mono-tetrahydrofuran-γ-lactone from Annona muricata. Heterocycles. 1990;31:861–867.
48. Rieser M.J., Kozlowski J.F., Wood K.V., McLaughlin J.L. Muricatacin: A simple biologically active acetogenin derivative from the seeds of Annona muricata (annonaceae) Tetrahedron Lett. 1991;32:1137–1140.
49. Luna J.D.S., de Carvalho J., de Lima M., Bieber L., Bento E.D.S., Franck X., Sant’Ana A. Acetogenins in Annona muricata L. (annonaceae) leaves are potent molluscicides. Nat. Prod. Res. 2006;20:253–257.
50. Escobar-Khondiker M., Höllerhage M., Muriel M.-P., Champy P., Bach A., Depienne C., Respondek G., Yamada E.S., Lannuzel A., Yagi T. Annonacin, a natural mitochondrial complex I inhibitor, causes tau pathology in cultured neurons. J. Neurosci. 2007;27:7827–7837.
51. Champy P., Höglinger G.U., Féger J., Gleye C., Hocquemiller R., Laurens A., Guérineau V., Laprévote O., Medja F., Lombès A. Annonacin, a lipophilic inhibitor of mitochondrial complex I, induces nigral and striatal neurodegeneration in rats: Possible relevance for atypical parkinsonism in guadeloupe. J. Neurochem. 2004;88:63–69.
52. Cortes D., Myint S.H., Laurens A., Hocquemiller R., Leboeuf M., Cavé A. Corossolone et corossoline, deux nouvelles γ-lactones mono-tétrahydrofuraniques cytotoxiques. Can. J. Chem. 1991;69:8–11.
53. Vila-Nova N.S., de Morais S.M., Falcão M.J.C., Alcantara T.T.N., Ferreira P.A.T., Cavalcanti E.S.B., Vieira I.G.P., Campello C.C., Wilson M. Different susceptibilities of Leishmania spp. promastigotes to the Annona muricata acetogenins annonacinone and corossolone, and the Platymiscium floribundum coumarin scoparone. Exp. Parasitol. 2013;133:334–338.
54. Vila-Nova N.S., Morais S.M.D., Falcão M.J.C., Machado L.K.A., Beviláqua C.M.L., Costa I.R.S., Brasil N.V.G.P.D., Andrade Júnior H.F.D. Leishmanicidal activity and cytotoxicity of compounds from two Annonacea species cultivated in northeastern Brazil. Rev. Soc. Bras. Med. Trop. 2011;44:567–571.
55. Hla Myint S., Cortes D., Laurens A., Hocquemiller R., Lebȩuf M., Cavé A., Cotte J., Quéro A.-M. Solamin, a cytotoxic mono-tetrahydrofuranic γ-lactone acetogenin from Annona muricata seeds. Phytochemistry. 1991;30:3335–3338.
56. Gromek D., Figadère B., Hocquemiller R., Cavé A., Cortes D. Corepoxylone, a possible precursor of mono-tetrahydrofuran γ-lactone acetogenins: Biomimetic synthesis of corossolone. Tetrahedron. 1993;49:5247–5252.
57. Rieser M.J., Fang X.-P., Rupprecht J.K., Hui Y.-H., Smith D.L., McLaughlin J.L. Bioactive single-ring acetogenins from seed extracts of Annona muricata. Planta Med. 1993;59:91–92.
58. Rieser M.J., Fang X.P., Anderson J.E., Miesbauer L.R., Smith D.L., McLaughlin J.L. Muricatetrocins a and b and gigantetrocin b: Three new cytotoxic monotetrahydrofuran-ring acetogenins from Annona muricata. Helv. Chim. Acta. 1993;76:2433–2444.
59. Roblot F., Laugel T., Lebœuf M., Cavé A., Laprévote O. Two acetogenins from Annona muricata seeds. Phytochemistry. 1993;34:281–285.
60. Li C.-M., Tan N.-H., Lu Y.-P., Liang H.-L., Mu Q., Zheng H., Hao X., Wu Y., Zhou J. Annomuricatin A, a new cyclopeptide from the seeds of Annona muricata. Acta Bot. Yunnanica. 1995;17:459–462.
61. Li C.M., Tan N.H., Zheng H.L., Mu Q., Hao X.J., He Y.N., Zou J. Cyclopeptide from the seeds of Annona muricata. Phytochemistry. 1998;48:555–556.
62. Wélé A., Zhang Y., Caux C., Brouard J.-P., Pousset J.-L., Bodo B. Annomuricatin C, a novel cyclohexapeptide from the seeds of Annona muricata. Comptes Rendus Chim. 2004;7:981–988.
63. Rieser M.J., Gu Z.-M., Fang X.-P., Zeng L., Wood K.V., McLaughlin J.L. Five novel mono-tetrahydrofuran ring acetogenins from the seeds of Annona muricata. J. Nat. Prod. 1996;59:100–108.
64. Gleye C., Raynaud S., Fourneau C., Laurens A., Laprévote O., Serani L., Fournet A., Hocquemiller R. Cohibins C and D, two important metabolites in the biogenesis of acetogenins from Annona muricata and Annona nutans. J. Nat. Prod. 2000;63:1192–1196.
65. De Yu L., Yu J.G., Luo X.Z., Lan S., Yang S.L. Muricatenol, a linear acetogenin from annona muricata (Annonaceae) Chin. Chem. Lett. 2000;11:239–242.
66. Chang F.-R., Wu Y.-C. Novel cytotoxic annonaceous acetogenins from Annona muricata. J. Nat. Prod. 2001;64:925–931.
67. Hisham A., Sreekala U., Pieters L., Bruyne T.D., van den Heuvel H., Claeys M. Epoxymurins a and b, two biogenetic precursors of annonaceous acetogenins from Annona muricata. Tetrahedron. 1993;49:6913–6920.
68. Leboeuf M., Legueut C., Cavé A., Desconclois J., Forgacs P., Jacquemin H. Alcaloïdes des annonacées XXIX1: Alcaloïdes de l’Annona muricata L. (in French) Planta Med. 1981;42:37–44.
69. Fekam Boyom F., Amvam Zollo P., Menut C., Lamaty G., Bessière J. Aromatic plants of tropical central africa. Part XXVII. Comparative study of the volatile constituents of five annonaceae species growing in cameroon. Flavour Frag. J. 1996;11:333–338.
70. Thang T., Dai D., Hoi T., Ogunwande I. Study on the volatile oil contents of Annona glabra L., Annona squamosa L., Annona muricata L. and Annona reticulata L., from Vietnam. Nat. Prod. Res. 2013;27:1232–1236.
71. Jirovetz L., Buchbauer G., Ngassoum M.B. Essential oil compounds of the Annona muricata fresh fruit pulp from cameroon. J. Agric. Food Chem. 1998;46:3719–3720.
72. Alali F.Q., Liu X.-X., McLaughlin J.L. Annonaceous acetogenins: Recent progress. J. Nat. Prod. 1999;62:504–540.
73. Tempesta M.S., Kriek G.R., Bates R.B. Uvaricin, a new antitumor agent from Uvaria accuminata (annonaceae) J. Org. Chem. 1982;47:3151–3153.
74. McLaughlin J.L. Paw paw and cancer: Annonaceous acetogenins from discovery to commercial products. J. Nat. Prod. 2008;71:1311–1321.
75. Carmen Zafra-Polo M., Figadère B., Gallardo T., Tormo J., Cortes D. Natural acetogenins from annonaceae, synthesis and mechanisms of action. Phytochemistry. 1998;48:1087–1117.
76. Zafra-Polo M.C., González M.C., Estornell E., Sahpaz S., Cortes D. Acetogenins from annonaceae, inhibitors of mitochondrial complex I. Phytochemistry. 1996;42:253–271.
77. Chih H.-W., Chiu H.-F., Tang K.-S., Chang F.-R., Wu Y.-C. Bullatacin, a potent antitumor annonaceous acetogenin, inhibits proliferation of human hepatocarcinoma cell line 2.2.15 by apoptosis induction. Life Sci. 2001;69:1321–1331.
78. Chan P., Ah R., Mh K. Anti-arthritic activities of Annona muricata L. Leaves extract on complete freund’s adjuvant (CFA)-induced arthritis in rats. Planta Med. 2010;76:P166.]
79. Arroyo J., Prashad M., Vásquez Y., Li E., Tomás G. Actividad citotóxica in vitro de la mezcla de Annona muricata y krameria lappacea sobre células cancerosas de glándula mamaria, pulmón y sistema nervioso central (in Spanish) Rev. Perú. Med. Exp. Salud Publica. 2005;22:247–253.
80. Astirin O.P., Artanti A.N., Fitria M.S., Perwitasari E.A., Prayitno A. Annonaa muricata linn leaf induce apoptosis in cancer cause virus. J. Cancer Ther. 2013;4:1244–1250.
81. Gavamukulya Y., Abou-Elella F., Wamunyokoli F., AEl-Shemy H. Phytochemical screening, anti-oxidant activity and in vitro anticancer potential of ethanolic and water leaves extracts of Annona muricata (graviola) Asian Pac. J. Trop. Med. 2014;7:S355–S363.
82. George V.C., Kumar D., Rajkumar V., Suresh P., Kumar R.A. Quantitative assessment of the relative antineoplastic potential of the n-butanolic leaf extract of Annona muricata linn. In normal and immortalized human cell lines. Asian Pac. J. Cancer Prev. 2012;13:699–704.
83. Moghadamtousi S.Z., Kadir H.A., Paydar M., Rouhollahi E., Karimian H. Annona muricata leaves induced apoptosis in A549 cells through mitochondrial-mediated pathway and involvement of NF-κB. BMC Complement. Altern. Med. 2014;14:299.
84. Moghadamtousi S.Z., Karimian H., Rouhollahi E., Paydar M., Fadaeinasab M., Kadir H.A. Annona muricata leaves induce g1 cell cycle arrest and apoptosis through mitochondria-mediated pathway in human HCT-116 and HT-29 colon cancer cells. J. Ethnopharmacol. 2014;156:277–289.
85. Ezirim A., Okachi V., James A., Adebeshi O., Ogunnowo S., Odeghe O. Induction of apoptosis in myelogenous leukemic k562 cells by ethanolic leaf extract of Annona muricata. Indian J. Drug Dis. 2013;2:142–151.
86. Asare G.A., Afriyie D., Ngala R.A., Abutiate H., Doku D., Mahmood S.A., Rahman H. Antiproliferative activity of aqueous leaf extract of Annona muricata L. On the prostate, BPH-1 cells, and some target genes. Integr. Cancer Ther. 2015;14:65–74.
87. Minari J., Okeke U. Chemopreventive effect of Annona muricata on DMBA-induced cell proliferation in the breast tissues of female albino mice. Egypt. J. Med. Hum. Genet. 2014;15:327–334.
88. Hamizah S., Roslida A., Fezah O., Tan K., Tor Y., Tan C. Chemopreventive potential of Annona muricata L leaves on chemically-induced skin papillomagenesis in mice. Asian Pac. J. Cancer Prev. 2012;13:2533–2539.
89. Eggadi V., Gundamedi S., Sheshagiri S.B.B., Revoori S.K., Jupally V.R., Kulandaivelu U. Evaluation of anticancer activity of Annona muricata in 1,2-dimethyl hydrazine induced colon cancer. World Appl. Sci. J. 2014;32:444–450.
90. Hansra D.M., Silva O., Mehta A., Ahn E. Patient with metastatic breast cancer achieves stable disease for 5 years on graviola and xeloda after progressing on multiple lines of therapy. Adv. Breast Cancer Res. 2014;3:84–87.
91. Moghadamtousi S.Z., Rouhollahi E., Karimian H., Fadaeinasab M., Firoozinia M., Abdulla M.A., Kadir H.A. The chemopotential effect of Annona muricata leaves against azoxymethane-induced colonic aberrant crypt foci in rats and the apoptotic effect of acetogenin annomuricin E in HT-29 cells: A bioassay-guided approach. PLoS ONE. 2015;10.
92. Elisya Y., Kardono L.B., Simanjuntak P. Tablet formulation of the ethyl acetate soluble extract of soursop (Annona muricata L.) leaves. Asian J. Appl. Sci. 2014;2:323–329.
93. N’gouemo P., Koudogbo B., Tchivounda H.P., Akono-Nguema C., Etoua M.M. Effects of ethanol extract of Annona muricata on pentylenetetrazol-induced convulsive seizures in mice. Phytother. Res. 1997;11:243–245.
94. Adeyemi D.O., Komolafe O.A., Adewole O.S., Obuotor E.M., Adenowo T.K. Anti hyperglycemic activities of Annona muricata (Linn) Afr. J. Tradit. Complement. Altern. Med. 2009;6:62–69.
95. Adeyemi D.O., Komolafe O.A., Adewole S.O., Obuotor E.M. Anti hyperlipidemic activities of Annona muricata (Linn) Internet J. Altern. Med. 2008;7:1.
96. Roslida A., Tay C., Zuraini A., Chan P. Anti-inflammatory and anti-nociceptive activities of the ethanolic extract of Annona muricata leaf. J. Nat. Rem. 2010;10:97–104.
97. Hamid R.A., Foong C.P., Ahmad Z., Hussain M.K. Antinociceptive and anti-ulcerogenic activities of the ethanolic extract of Annona muricata leaf. Rev. Bras. Farmacogn. 2012;22:630–641.
98. Chance B., Sies H., Boveris A. Hydroperoxide metabolism in mammalian organs. Physiol. Rev. 1979;59:527–605.
99. Liao J.-C., Deng J.-S., Chiu C.-S., Huang S.-S., Hou W.-C., Lin W.-C., Huang G.-J. Chemical compositions, anti-inflammatory, antiproliferative and radical-scavenging activities of Actinidia callosa var. Ephippioides. Am. J. Chin. Med. 2012;40:1047–1062.
100. Chen W., Weng Y.-M., Tseng C.-Y. Antioxidative and antimutagenic activities of healthy herbal drinks from Chinese medicinal herbs. Am. J. Chin. Med. 2003;31:523–532.
101. Nwokocha C.R., Owu D.U., Gordon A., Thaxter K., McCalla G., Ozolua R.I., Young L. Possible mechanisms of action of the hypotensive effect of Annona muricata (soursop) in normotensive sprague-dawley rats. Pharm. Biol. 2012;50:1436–1441.
102. Osorio E., Arango G.J., Jiménez N., Alzate F., Ruiz G., Gutiérrez D., Paco M.A., Giménez A., Robledo S. Antiprotozoal and cytotoxic activities in vitro of colombian annonaceae. J. Ethnopharmacol. 2007;111:630–635.
103. Snow R., Craig M., Deichmann U., le Sueur D. A preliminary continental risk map for malaria mortality among african children. Parasitol. Today. 1999;15:99–104.
104. Winstanley P. Chemotherapy for falciparum malaria: The armoury, the problems and the prospects. Parasitol. Today. 2000;16:146–153.
105. Arthur F.K., Woode E., Terlabi E.O., Larbie C. Bilirubin lowering potential of Annona muricata (Linn.) in temporary jaundiced rats. Am. J. Pharmacol. Toxicol. 2012;7:33–40.
106. Wezel A., Casagrande M., Celette F., Vian J.-F., Ferrer A., Peigné J. Agroecological practices for sustainable agriculture. A review. Agron. Sustain. Dev. 2014;34:1–20.
107. Moghadamtousi S.Z., Rouhollahi E., Karimian H., Fadaeinasab M., Abdulla M.A., Kadir H.A. Gastroprotective activity of Annona muricata leaves against ethanol-induced gastric injury in rats via Hsp70/Bax involvement. Drug Des. Dev. Ther. 2014;8:2099–2111.
108. Dos Santos A., Sant’Ana A. Molluscicidal properties of some species of Annona. Phytomedicine. 2001;8:115–120.
109. Moghadamtousi S.Z., Rouhollahi E., Hajrezaie M., Karimian H., Abdulla M.A., Kadir H.A. Annona muricata leaves accelerate wound healing in rats via involvement of hsp70 and antioxidant defence. Int. J. Surg. 2015;18:110–117.
110. Paarakh P.M., Chansouria J., Khosa R. Wound healing activity of Annona muricata extract. J. Pharm. Res. 2009;2:404–406.
111. N. Anuj Patnaik, T. Nagarjuna, T. V. Thulasiramaraju. Sustained release drug delivery system: A modern formulation approach. International Journal of Research in Pharmaceutical and Nano Sciences. 2(5), 2013, 586- 601.
112. Dusane Abhijit Ratilal, Gaikwad Priti D., Bankar Vidyadhar H., Pawar Sunil P. A review on: Sustained released technology. International Journal of Research in Ayurveda & Pharmacy. 2011. 2 (6) 1701-1708.
113. K. P. Sampath Kumar, Debjit Bhowmik, Shweta Srivastava, Shravan Paswan, A. S. Dutta. Sustained Release Drug Delivery System Potential. The Pharma Innovation. Online Available at www.thepharmajournal.com .Vol. 1 No. 2 2012& Vol. 1 No. 1 2012. Page no. 46-56.
114. Jaimini Manish, Kothari Abhay. Sustained release matrix type drug delivery system: A review. Journal of Drug Delivery & Therapeutics. 2012, 2(6), 142-148.
115. Satinder Kumar, Shashi Kant , Bharat Prashar. Sustained release drug delivery system: A review. International Journal of Institutional Pharmacy and Life Sciences. 2(3): May-June 2012. Page no. 356-376.
116. Gajalakshmi S, Vijayalakshmi S and Devi Rajeswari V. Phytochemical and pharmacological properties of annona muricata: a review. International Journal of Pharmacy and Pharmaceutical Sciences 2012; 4(2): 3-6.
117. Tai S. Kedari, Ayesha A. Khan. Guyabano (Annona Muricata): A review of its traditional uses phytochemistry and pharmacology. American Journal of Research Communication, 2014, 2(10): 247-268.
118. Coria-Te´llez, A.V. et al., Annona muricata: A comprehensive review on its traditional medicinal uses, phytochemicals, pharmacological activities, mechanisms of action and toxicityAnnona muricata. Arabian Journal of Chemistry 2016; 9(1): 1-170.
119. Mathew J, George R, Theruvil R, Padavil TC, Tomy L, Kurian A. Antibacterial Activity of Leaf Extract of Annona muricata and Simarouba glauca on Enterococcus faecalis. J Contemp Dent Pract 2016;17(8):650-653.
120. Fang Rong Chang and Yang Chang Wu. Novel Cytotoxic Annonaceous Acetogenins from Annona muricata. J. Nat. Prod 2001; 64 (7): 925–931.
121. Christophe Gleye , Sophie Raynaud , Christophe Fourneau , Alain Laurens , Olivier Laprévote , Laurent Serani , Alain Fournet and Reynald Hocquemiller. Cohibins C and D, Two Important Metabolites in the Biogenesis of Acetogenins from Annona muricata and Annona nutans. J. Nat. Prod., 2000; 63 (9): 1192–1196.
122. Sulistyoningrum E, Rachmani EPN, Baroroh HN, Rujito L. Annona muricata Leaves Extract Reduce Proliferative Indexes And Improve Histological Changes In Rat’s Breast Cancer. J App Pharm Sci, 2017; 7 (01): 149-155.
123. Jorge Arroyo Acevedo, Cesar Franco Quino, Eliberto Ruiz Ramirez, Roberto Chávez Asmat, Andrea Anampa Guzmán, Ernesto Raéz González, and José Cabanillas Coral. Antiallergic effect of the atomized extract of rhizome of Curcuma longa, flowers of Cordia lutea and leaves of Annona muricata. Ther Clin Risk Manag 2016; 12: 1643–1647.
124. Ratnaparkhi M P, Gupta Jyoti P. Sustained Release Oral Drug Delivery System - An Overview. International Journal of Pharma Research & Review 2013; 2(3):11-21.
125. Pooja R. Alli, Pratima B. Bargaje, Nilesh S. Mhaske, Sustained Release Drug Delivery System: A Modern Formulation Approach, Asian Journal of Pharmaceutical Technology& Innovation, 04 (17); 2016,108-118 .
126. Ahmed Hashim Hussein. Formulation and evaluation of sustained release tablets of pentoxifylline using okra extract as a novel retardant. Int J Pharm Pharm Sci 2015; 7(2): 204-208.
127. Trapti Rastogi, Khadabadi S S. Design, development and evaluation of matrix tablet containing indigenous medicinal plants. International Journal of Pharmaceutical Sciences and Research 2011; 2(11): 2806-2811.
128. Tavakoli N, Ghodrati M, Ghassemi Dehkordi N and Sadeghi Aliabadi H. Formulation and evaluation of a new herbal tablet from strawberry and grape leaves. Jundishapur Journal of Natural Pharmaceutical Products 2008; 3(1): 19-25.
129. Ghiwar Nitin B, Gattani Surendra G and Chalikwar Shailesh S. Design, Development and Evaluation of Oral Herbal Formulations of Piper nigrum and Nyctanthes arbortristis. International Journal of PharmTech Research 2010; 2(1): 171-176.
130. Gupta Shikha and Gahlot Kavita. Formulation and evaluation of herbal antidemential tablets. Asian J Pharm Clin Res 2012; 5(1): 148-153.
131. Chopra RN, Chopra K, Handa KL, Kapoor LD. Indigenous Drugs of India, Calcutta, India, 1958, 80.
132. Finar IL. Organic Chemistry, ELBS Long Mann Publishers, 1994, 517.
133. Kokate CK. Pharmacognosy, 14, Nirali Publications, 2000, 114.
134. Rutkowski M and Grzegorczyk K. Modifications of spectrophotometric methods for antioxidative vitamins determination convenient in analytic practice. Acta Sci Pol Technol Aliment 2007; 6:17-28.
135. Lilybeth F. Olowa and Olga M. Nuñeza. Brine Shrimp Lethality Assay of the Ethanolic Extracts of Three Selected Species of Medicinal Plants from Iligan City, Philippines. Int. Res. J. Biological Sci 2013; 2(11):74-7.
136. Gupta M P, Monge A et al. Screening of Panamanian medicinal plants for brine shrimp toxicity, crown gall tumor inhibition, cytotoxicity and DNA interaction. Int. J. Pharmacol 1996; 34:123-27.
137. Meyer B N, Ferrigni N R, Putnam J E, Jacobsen L B, Nichols D E and McLaughlin J L. Brine shrimp: A convenient general bioassay for active plant constituents. Plant Med 1982; 45: 31-4.
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT editor-in-chief@pharmatutor.org
Subscribe to Pharmatutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE









