ABOUT AUTHORS:
Pankaj Sharma*, Bhupendra Vyas, Yuvraj Singh Sarangdevot, Abhishek Sharma, Bhuvanesh Sharma
Department of Quality Assurance,
Bhupal Nobles’ College of Pharmacy,
Udaipur- 313002, Rajasthan, India
*pankajs.medicolite@gmail.com
ABSTRACT
Chronopharmacology involves both the investigation of drug effects as a function of biologic timing and the investigation of drug effects upon rhythm characteristics. Chronopharmacology certainly holds promise for the creation of the most favourable conditions for drug effects and safety and may therefore represent an important method of improving the treatment of many diseases. The goal of chronopharmacology is to optimize the therapeutic effect and control or reduce the adverse effects without altering the functioning of the drug in the body. Pharmacologic chronotherapy is a developing science that holds much hope for increasing the effectiveness of drug therapy and for reducing the incidence of toxic drug reaction. There is convincing scientific work to indicate that more attention should be given to the timing of drug administration. Most prescribers are currently more concerned with "what" to prescribe rather than "when" to prescribe it. Chronopharmacology certainly holds promise for the creation of the most favourable conditions for drug effects and safety and may therefore represent an important method of improving the treatment of many diseases.
REFERENCE ID: PHARMATUTOR-ART-1749
1. INTRODUCTION
Nearly all functions of the body, including those influencing pharmacokinetic parameters such as drug absorption and distribution, drug metabolism and renal elimination, show significant daily variations: these include liver metabolism, hepatic blood flow and the first-pass effect; glomerular filtration, renal plasma flow and urine volume and pH; blood pressure, heart rate and organ perfusion rates; acid secretion in the gastro-intestinal tract and gastric emptying time.
The onset and symptoms of diseases such as asthma attacks, coronary infarction, angina pectoris, stroke and ventricular tachycardia are circadian phase dependent. In humans, variations during the 24 h day in pharmacokinetics (chrono-pharmacokinetics) have been shown for cardiovascularly active drugs (propranolol, nifedipine, verapamil, enalapril, isosorbide 5-mononitrate and digoxin), anti-asthmatics (theophylline and terbutaline), anticancer drugs, psychotropics, analgesics, local anaesthetics and antibiotics, to mention but a few. Even more drugs have been shown to display significant variations in their effects throughout the day (chronopharmacodynamics and chronotoxicology) even after chronic application or constant infusion.
Moreover, there is clear evidence that even dose/concentration-response relationships can be significantly modified by the time of day. Thus, circadian time has to be taken into account as an important variable influencing a drug's pharmacokinetics and its effects or side-effects.
There is convincing scientific work to indicate that more attention should be given to the timing of drug administration. Most prescribers are currently more concerned with "what" to prescribe rather than "when" to prescribe it.
Chronotherapeutics certainly holds promise for the creation of the most favourable conditions for drug effects and safety and may therefore represent an important method of improving the treatment of many diseases. The goal of chronotherapeutics is to optimize the therapeutic effect and control or reduce the adverse effects without altering the functioning of the drug in the body.
Pharmacologic chronotherapy is a developing science that holds much hope for increasing the effectiveness of drug therapy and for reducing the incidence of toxic drug reaction.
Chronopharmacology:
It is the science dealing with the optimizations of drug effect and the minimizations of adverse effects by timing medications in relation to biological rhythm Goal is to improve our understanding of periodic and thus predictable changes in both desired effects and tolerance of medication. Chronopharmacologyis the investigative science concerned with the biological rhythm dependencies of medications.
Chronobiology:
It is the science dealing with the phenomenon of rhythmicity in living organisms is called Chronobiology. It is derived from chronos (time), bios (life), and logos (study of)--is the objective description of biological time structures and plays an important role in medicine. Circadian rhythms regulate the behavior, physiology and function of living organisms at many biochemical levels.
Aim of chronobiology is optimization of pharmacotherapeutics, taking into consideration-rhythm dependencies in kinetics and dynamics of medications, predictable in time variability in the manifestation and severity of diseases.
Circadian rhythm:
It is defined as oscillations in the biological, physiological and behavioral function of an organism with a periodicity of 24 hrs. The concepts of chronobiology and chronopharmacology have become more and more important in medical practice nowadays. Today, the circadian variation in blood pressure and heart rate as well as in the occurrence of acute cardiovascular disease is quite obvious (ischemia, infarction, stroke and sudden death). However, biological rhythms are also present in episodes of dyspnoea in nocturnal asthma, in hormonal pulses, in the organization of the immunological system and in the processes of cellular proliferation.
SuprachtAsmatic Nuclei (SCN) located in the anterior ventral hypothalamus has been identified as that centre. That generates circadian activity. Lesion of SCN produces loss of circadian rhythm- sleep-wake cycle activity- rest cycle, skin temperature and corticosteroidSecretion. Goals of Chronotherapy are to match the timing of treatment with the intrinsic timing-of illness.
The introduction of chronopharmacology into pharmacokinetic theory leads to a clock time-dependent infusion rate. The infusion modulation depends both on type of chronophenomenon, chronopharmacokinetics or chronestesy, and plasma clearance rate of the drug. In the presence of chronestesy of a biosystem the pharmacologic effect can be maintained constant only when plasma drug clearance is fast enough to allow an adequate modulation of the plasma drug concentration.
Circadian (congruent to 24 h), circannual (congruent to 1 year) and other biological rhythms of endogenous origin, detectable at all levels of organization, constitute a temporal structure in all animal species, including man. Circadian, circannual and other rhythmic changes in biological susceptibility and response of organisms to a large variety of physical as well as chemical agents including medications and foods are rather common phenomena.
Chronergy:
It isrhythmic changes of both desired (effectiveness) and undesired (toxicity, tolerance) effects on the organism as a whole.
On a 24 hour scale there are peaks and troughs of physiological variables that are not randomly distributed, but the controlled by a set pacemaker, so called biological clocks.
Chronokinetics:
It is study of absorption, distribution, metabolism and excretion of drug according to the time of day, menstrual cycle or year.
Chronesthesy:
It is rhythmic changes in susceptibility or sensitivity of a target system to a drug, which cannot be explained by chronokinetic changes. In humans, target can be skin, bronchial tree, and stomach.
2. BIOLOGICAL RHYTHMS
Biological rhythms are innately determined rhythmic biological process or function and self-sustaining oscillation with the duration of time between successive repetitions (i.e., the period) being rather non varying under normal conditions.100 different, measurable parameters in human body exhibit rhythmic variability within 24 hours.
Circadian rhythms are particularly important in medicine. Circadian (circa about dies, day, or about 24 hour) Physiological day is about 25 hours Clock is reset daily by the environment night day social schedules. A circadian clock in the brain coordinates daily physiological cycles like sleep/wake, digestion, temperature, hormones. Biologic rhythms are endogenous nature of circadian. Lack of external synchronizers leads to free running rhythms. The period of free-running rhythms is longer or shorter than 24 hours and is characteristic for each species. Our internal clocks are genetically determined. An internal biological clock located in mammals, in the suprachiasmatic nucleus of the hypothalamus (SCN), delivering its message of time throughout the body. It is responsible for circadian rhythms and annual/seasonal rhythms. The SCN uses its connections with the autonomic nervous system for spreading its time-of-day message, either by setting the sensitivity of endocrine glands (i.e., thyroid, adrenal, ovary) or by directly controlling an endocrine output of pineal gland (i.e., melatonin synthesis).
2.1 Rhythms of Different Systems:
* Respiratory System:
Increased bronchoconsriction at night because of increased parasympathetic tone decreased adrenaline and decreased cortisol at midnight, increased sensitivity to irritants and allergens at night.
* Gastrointestinal tract:
Acid secretion is 2-3 times greater between 10 pm and 2 am.
* Cardiovascular system:
Amplitude of 24 hr variation is more for diastolic blood pressure as compared to systolic blood pressure. Blood pressure shows two peaks at 9-11 a.m. and 6-7 p.m. Blood pressure decreases (slight) at afternoon and profound dip at night.
* Endocrine system:
Cortisol- Highest secretion is just before awakening and in the morning lowest at mid-night. Growth hormone- Peaks during sleep. Testosterone- Peaks early morning. Insulin- 5-10 fold Increase after ingestion of food.
* Receptors:
Circadian rhythm has been found for receptors in brain and heart of rats and blood cells in humans. Circadian rhythm was observed for the number of binding sites rather than for binding capacity of sites.
* Plasma protein binding:
Albumin and acid glycoprotein reach their nadir during nocturnal rest and their zenith in the morning. Therefore drugs bound to plasma protein, like valproic acid, carbamazepine, diazepam, lignocaine, prednisolone show increase in free fraction at night.
* Liver enzymes:
Oxidative reactions peak in the middle of the (nocturnal) activity span. Conjugation catalyzed by UDP-glucuronyl transferees is greater during activity than during the ' rest phase. Sulphate conjugation is faster during the rest than during activity.
2.2 Mechanism of Circadian Rhythm:
Circadian rhythms have been documented throughout the plant and animal kingdom at every level of eukaryotic organization. Circadian rhythms are endogenous in nature, driven by oscillators or clocks, and persist under free-running (e.g. constant darkness) conditions. The genes expressing the biological clock have been identified in various species.
The important feature of endogeneous biological rhythms is their anticipatory character. Rhythmicity inherent to all living systems, allows them to adapt more easily and to better survive under changing environmental conditions during the 24 hours of a day as well as during changing seasons. Having this in mind it is easy to conceive that not only must the right amount of the right substance be at the right place, but also this must occur at the right time. Also in man nearly all functions of the body including those influencing pharmacokinetic parameters such as drug absorption and distribution, drug metabolism and renal elimination display significant daily variations.
2.3 Rhythms Affecting Our Body:
· Ultradian- Cycles shorter than a day e.g., msec. for a neuron to fire.
· Circadian- Circa-about, dies day. Lasting for about 24 hours, e.g., sleep and wake cycles.
· Infraradian- Cycles longer than 24 hours e.g. menstrual cycle.
· Seasonal- Seasonal affective disorders causing depression in people during the short day's of winter.
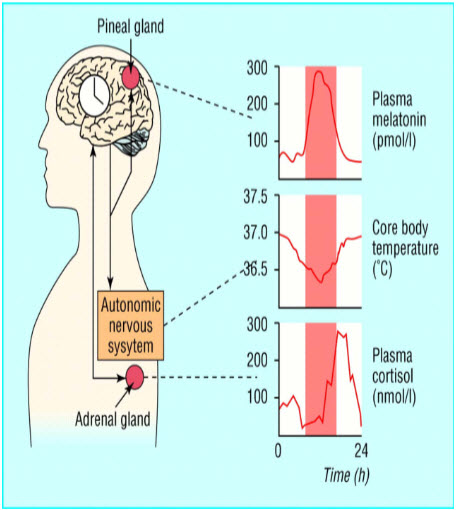
Figure 1: Rhythmicity of Body System.
2.4 Aspects of Rhythmicity:
The science dealing with the phenomenon of rhythmicity in living organisms is called Chronobiology. The branch of Chronobiology dealing with the pharmacologic aspects is termed Chronopharmacology. It may be subdivided into Chronotherapy and chronotoxicity. New technology makes possible Chronotherapy to Chrpnptherapeutics, that is, increase of the efficiency and safety of medications by proportioning their concentrations during the 24 hours in synchrony with biological rhythm determinants of disease.
The pharmacotherapy of medical conditions generally entails the scheduling of prescription medications that possess drug-delivery technologies that achieve thehomeostatic goal of constancy of blood and tissue drug concentration. It is assumed that the need for medication by patients is nonvarying throughout the 24 hours and that constancy of drug level translates into constancy in drug effect.
Knowledge of day-night and other predictable-in-time variation in the symptom intensity and risk of acute exacerbations of disease coupled with evidence of circadian rhythms in the kinetics, effects, and safety of medications constitutes the rationale for a new pharmacologic approach to treatment- Chronotherapeutics.
2.5 Mechanism of Chronopharmacology:
Circadian and other rhythmic changes in biological susceptibility and response of organisms to a large variety of physical and chemical agents including medications and foods are rather common phenomena. Time-related differences in drug effects depend upon endogenous circadian rhythms, which include metabolic pathways. In addition, chronopharmacology investigates drug effects on parameters (e.g. circadian period, peak time, amplitude, and adjusted mean) used to characterize biological rhythms. A better understanding of periodic and thus predictable changes in drug effects can be attained by consideration of complementary concepts:-
a. The chronokinetics for a drug, i.e. dosing time-dependent and predictable (rhythmic) changes in parameters used to characterize the pharmacokinetics (bioavailability) of a drug, e.g. Cmax, tmax, AUC, and t1/2;
b. The chronesthesy, i.e. rhythmic changes in susceptibility of the target biosystem to this drug, including CR in pharmacodynamic processes; and
c. The chronergy, i.e. the drug-integrated overall effect.
.Chronopharmacology involves both the investigation of drug effects as a function of biologic timing and the investigation of drug effects upon rhythm characteristics.
Circadian changes in the effects of various chemical agents have been documented: histamine, sodium salicylate, acetylcholine, halothane, prostaglandine F, reserpine, cyproheptadine, ethanol, insulin, chlorothiazide, oxymetholone, orciprenalin and SCH 1000 (bronchodilators), indomethacin, ACTH, cortisol and various synthetic corticosteroids.
Chronopharmacology is useful to solve problems of drug optimization, i.e. to enhance the desired efficiency or to reduce its undesired effects. In the human organism (among other animal species) the metabolic fate of a pharmacologic agent (as well as that of a nutrient) is not constant as a function of time. Thus, the chronobiological approach of pharmacologic phenomena involves a lesser risk of errors and/or false information than the conventional homeostatic approach.
One of the aims of chronopharmacology refers to the use of a chronopharmacological approach to clinical treatment so as to enhance both effectiveness and tolerance of a drug by determining the best biological time for its administration.
2.6 Factors Affecting Chronopharmacology:
To optimize chronotherapeutic schedules (designs), we examined the interindividual differences in chronopharmacologic effects of drugs with consideration of the following three factors:
(a) Inherited factors of direct relevance to chronopharmacology (genetic variability, gender-related differences) as well as age-related differences;
(b) Interindividual difference in chronoeffectiveness related to disease (e.g., various types and stages of cancer, affective disorders, etc.) as well as to drug-dependent alteration (phase shifts, distortion) of biological rhythms; and
(c) Means to solve problems resulting from the need of individualization in chronotherapy. These involve the use of circadian marker rhythms (MR) whose characteristics (peak or trough time, amplitude, etc.) can be precisely quantified and thus are applicable as a reference system for physiologic, pathologic, pharmacologic and therapeutic uses.
The MR has to be specific and pertinent and must be easily monitored and documented. This approach can be further advanced by the use of a battery of MRs rather than a single MR. Other suggested means relate to the fact that chronobiotics (agents capable of influencing parameters of a set of biological rhythms) should be considered (e.g., corticoids and adrenocorticotropic hormone) and/or to the subject's synchronization should be enforced by "conventional" zeitgebers (e.g., bright light, physical activity).
3. CHRONOTHERAPEUTICS
3.1 Chronotherapeutics Introduction:
It is refers to a treatment method in which in vivo drug availability is timed to match rhythms of disease in order to optimize therapeutic outcomes and minimize side effects. It is based on the observation that there is an interdependent relationship between the peak- to-trough rhythmic activity in disease symptoms and risk factors, pharmacologic sensitivity, and pharmacokinetics of many drugs. As more continues to be learned about chronobiology and chronotherapeutics, it is becoming increasingly more evident that the specific time that patients take their medication may be even more significant than was recognized in the past. The tradition of prescribing medication at evenly spaced time intervals throughout the day, in an attempt to maintain constant drug levels throughout a 24-hour period, may be changing as researchers' report that some medications may work better if their administration is coordinated with day night.
A chronotherapeutic approach is indicated when the risk of severe medical events or the intensity of diseases and their symptoms is known to vary predictably during 24 hours and or other time periods. Large scale clinical trials have shown that the efficiency and safety of certain conventional medications can be improved by dosing with reference to the circadian time structure.
This was first demonstrated during the 1960s; once-daily mornings as opposed to multiple- daily morning as opposed to multiple-daily dosing ofmethylprednisolone tablets reduces the risk of hypothalamic-pituitary-adrenal suppression and improves the treatment of sever inflammatory conditions. Other examples:- The once-daily supper-time dosing schedule of conventional H2-receptor antagonists to avert the nocturnal increase in gastric acid secretion in the treatment of peptic ulcer disease and the evening timing of HMG-CoA reductase antagonists, taking into account the evening increase of cholesterol synthesis in the liver. The risk of asthmatic attack is almost 70 times higher in patients at 04:00-05:00 in the morning, compared with the afternoon.
New technology makes possible the temporal modulation of drug levels in relation to patient requirements. During the 1980s, the Purdue Frederick Company in the United States and the Byk Gulden Company in Germany each began marketing a theophylline chadministration of these special dosage forms. Evening results in elevated chronotherapy for nocturnal asthma.
Eophylline concentration during the night time when the likelihood of asthma is greatest and lowest drug level during the day when the risk of disease is least. Twenty-four-hour patterns in cardiovascular diseases have been documented by numerous investigators. Circadian rhythms of blood pressure in healthy subjects–nocturnal lowering of BP and a rapid elevation between 05:00-09:00 o’clock in the morning. Hypertensive patients usually present with similar patterns but the mean arterial pressure is higher.
The morning RISE in BP is associated with numerous complications of hypertension. The lack of nocturnal lowering is often associated with malignant hypertension and poor prognosis. It is smaller in males and decreases with age. Bedtime dosing of long-acting antihypertensive agents helps restore BP control in the morning and bring back normal circadian rhythm of BP (non dipper-dipper).
In diurnally active patients, angina, acute myocardial infarction (AMI), sudden cardiac death, and ischemic and hemorrhagic stroke each is several-fold more frequent in occurrence during the initial 3 to 5 hours of morning activity (about 10 a.m.) than at any other time of the day or night. In contrast, episodes of vasospasm in Prinzmetal angina are most common during the sleep span, as are the symptoms of patients with congestive heart failure.
Increased blood coagulation in the morning decreased efficacy of anticoagulants and fibrinolytic agents. Increased incidence of CAD and strokes is connected with increased sympathetic tone, the rapid increase in BP, HR, and myocardial oxygen demand (the rapid elevation in BP at the start of the activity span constitutes a shearing stress of sufficient magnitude to cause rupture of unstable coronary plaques with thrombosis and AMI as sequelae), increased blood coagulation, and coronary vessel inflammation and reactivity. The results of selected investigations that addressed circadian rhythms are the effects of antihypertensive, antiarrhythmic and anticoagulant therapies. In general, the adrenoceptor and calcium channel antagonists of propranolol, oxprenolol, atenolol, pindolol, nifedipine, and verapamil exert stronger effects on HR and BP during the day than nighttime.
Enalapril produces a greater (and more rapid onset of) effect on daytime BP when dosed once per day in the morning, whereas it has greater effect on nighttime BP when dosed once per day in the evening. Once per day at evening compared with morning ingestion of isradipine better normalizes the disturbed circadian rhythm of BP in non-dialysis chronic renal failure patients.
The effects of ramipril and quinapril on BP are also dependent on administration time; a morning once-per-day dosing schedule reduces sleep-time BP in a moderate amount, whereas an evening one reduces it excessively, especially with quinapril. The dosing-time dependencies of diltiazem on BP are different; the morning once-daily ingestion schedule exerts strong effect on nighttime BP, whereas the evening one produces a strong effect on it during the morning and afternoon. The same medication when dosed in the evening rather than morning need not have the same pharmacokinetics or efficiency of effect. Moreover, different formulations of the same therapeutic agent may vary greatly in the extent to which their pharmacokinetics are administration time-dependent because of proprietary disparities in the respective drug-delivery systems.
In the past, the effects of the body's rhythms on the behavior of medications were not well understood by the medical community; thus they were not taken into account in the design and interpretation of prevention trials.
Due to advances in chronobiology, chronopharmacology, and global market constraints, the traditional goal of pharmaceutics (e.g. design drug delivery systems with a constant drug release rate) is becoming obsolete. However, the major bottleneck in the development of drug delivery systems that match the circadian rhythm (chronopharmaceutical drug delivery systems: ChrDDS) may be the availability of appropriate technology. The last decade has witnessed the emergence of ChrDDS against several diseases. The increasing research interest surrounding ChrDDS may lead to the creation of a new sub-discipline in pharmaceutics known as chronopharmaceutics.
This review introduces the concept of chronopharmaceutics, addresses theoretical/formal approaches to this sub-discipline, underscores potential disease-targets and revisits existing technologies and examples of ChrDDS. Future development in chronopharmaceutics may be made at the interface of other emerging disciplines such as system biology and nanomedicine. Such novel and more biological approaches to drug delivery may lead to safer and more efficientdisease therapy in the future.
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT articles@pharmatutor.org
Subscribe to Pharmatutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE
3.2 Diseases and Chronotherapeutics:
Up to now, design of drug delivery systems has been governed by the homeostatic theory. This theory is based on the assumption of biological functions that display constancy over time. However, chronobiological studies have established circadian rhythm for almost all body functions, e.g., heart rate, blood pressure, body temperature, plasma concentration of various hormones, gastric pH and renal function. It has become apparent that rhythmic processes are indispensable for the treatment of human diseases. Just as physiological functions vary over time, pathological states of disease have circadian rhythms. Epidemiological studies have documented the elevated risk of disease symptoms during the 24-hour cycle.
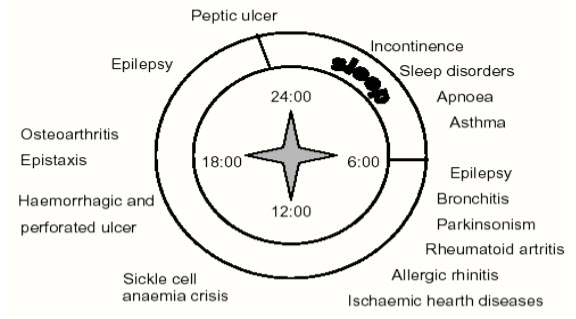
Figure 2: Diseases known to display circadian rhythm.
The potential benefits of chronotherapeutics have been demonstrated in the management of a number of diseases. In particular there is great deal of interest in how chronotherapy can particularly benefit patients suffering from allergic rhinitis, rheumatoid arthritis and related disorders, asthma, cancer, cardiovascular diseases, and peptic ulcer disease. Patients with allergic rhinitis often report that they suffer their worst symptoms when they wake up in the morning. Patients may obtain better results in controlling this morning discomfort if they were to take a long-acting antihistamine at night rather than taking the medication in the morning as is frequently recommended.
* Anti-inflammatory therapy:
Individuals suffering from rheumatoid arthritis and related painful joint disorders, NSAIDs may be more effective at relieving pain, if the drug is administered at least 6 hours before the pain reaches its peak. It will be more helpful if arthritis patients take the NSAIDs before bed time if they experience a high level of discomfort in the morning.
* Anti-asthma therapy:
It has been estimated that symptoms of asthma occur 50 to 100 times more often at night than during the day. Many circadian-dependent factors appear to contribute to the worsening of nocturnal asthmatic symptoms. For example, cortisol (an anti-inflammatory substance) levels were highest at the time of awakening and lowest in the middle of the night, and histamine (a mediator of bronchoconstriction) concentrations peaked at a level that coincided with the greatest degree of bronchoconstriction at 4:00 am. A research finding also reveals that theophylline absorption is slower at night. The enhanced understanding of the chronobiological impact upon the pathology of asthma, and the pharmacology and pharmacokinetics of the drugs used in its management, has led to new approaches to disease care.
* Chemotherapy:
Antineoplastic drugs cause cytotoxic effects on healthy and diseased tissues. As would be expected, the biological rhythms of both healthy and tumor cells may influence the susceptibility of normal and malignant cells to these agents. It has been demonstrated that “susceptibility rhythms” to drugs may differ between healthy tissue and cancerous tissue. Therefore, the “correct” timing of drug treatment may reduce host toxicity, increase maximum drug tolerance, and ultimately result in better tumor management. The pharmacologic and pharmacokinetic properties of the drug, rhythmic changes in DNA and RNA synthesis, RNA translational activity and mitotic activity may influence tumor cell susceptibility. It appears that the timing of drug administration in the treatment of cancer can have a significant impact upon treatment success.
* Cardiovascular therapy:
The differences in patterns of illness between day and night for cardiovascular disorders such as hypertension, angina, heart attack, sudden cardiac death and stroke have been documented. Medications have been formulated and dosing schedules established, in an attempt to provide appropriate concentration of a drug in the target area of the body when the drug is most needed. For example, it has often been found that the blood pressure of a hypertensive patient increases rapidly in the morning after awakening, typically peaks in the middle to late time of the day, decreases in the evening, and is lowest while the patient sleeps at night. It may also be important to recognize that the risk of heart attack appears to be greatest during the early morning hours after awakening. Currently, there are antihypertensive products in the market that are chronotherapeutic medications with novel drug delivery systems, releasing drug during the vulnerable period of 6 am to noon upon administration of medications at 10 pm.
3.3 Chronotherapeutic Drug Delivery Systems:
Controlled release formulations can be divided into subgroups of rate-controlled release, delayed-release and pulsed-release formulations. Delayed-release formulations include time-controlled release and site-specific dosage forms. When constant drug plasma levels need to be avoided, as in chronotherapy, time-controlled or pulsed-release formulations are preferable, especially in the treatment of early morning symptoms. By timing drug administration, plasma peak is obtained at an optimal time and the number of doses per day can be reduced. Saturable first-pass metabolism and tolerance development can also be avoided. Various technologies to develop time-controlled peroral drug delivery systems have been extensively studied in recent decades. Some of these systems are discussed in the following subsections:-
a. Press-coated systems:
Delayed-release and intermittent-release formulations can be achieved by press--coating. Press-coating, also known as compression coating, is relatively simple and cheap and may involve direct compression of both the core and the coat, obviating the need for a separate coating process and the use of coating solutions. Materials such as hydrophilic cellulose derivatives can be used and compression is easy on a laboratory scale. On the other hand, for large-scale manufacture, special equipment is needed.
The major drawbacks of the technique are that relatively large amounts of coating materials are needed and it is difficult to position the cores correctly for the coating process. In recent years, various controlled release, especially time-controlled release; drug delivery systems based on compression coating technology have been studied. Most of such formulations release drug after a lag phase, followed by a rapid dissolution of the core. Conte et al have developed a press-coated device in which the inner core contains the drug and the outer coat is made of different types of polymers. The outer barrier, which controls drug release, can be either swellable or erodible. Lag times can be varied by changing the barrier formulation or the coating thickness. Matsuo et al have developed a diltiazem hydrochloride formulation intended for use in the treatment of time-related symptoms of ischaemic heart disease and hypertension. The tablet consists of a core, which contains the drug, and a coat formed by compressing hydroxy methylcellulose. Diltiazem is rapidly released after a delay of several hours. Marvolaetal have developed a press-coated tablet formulation in which most of the total amount of drug is in the tablet core. Hydrophilic polymers such as hydroxypropyl methylcellulose and sodium alginate have been used in the coat to control drug release. The extent of bioavailability of furosemide, ibuprofen and salbutamol sulphate from the system dev eloped has been found to be satisfactory.
b. Enteric-coated systems:
Enteric coatings have traditionally been used to prevent the release of a drug in the stomach. Enteric coatings are pH- sensitive and drug is released when pH is raised above 5 in the intestinal fluid. These formulations can be utilized in time-controlled drug administration when a lag time is needed. Because of the unpredictability of gastric residence, such systems cannot be the first choice when a time-controlled release is required. In the treatment of nocturnal asthma, a albutamol formulation containing a barrier coating which is dissolved in intestinal pH level above about 6, has been successfully used. The system contains a core which is film coated with two polymers, first with HPMC and then with a gastro-resistant polymer (Eudragit this system the duration of the lag phase in absorption can be controlled by the thickness of the HPMC layer.
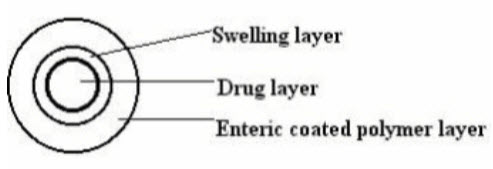
Figure 3:Schematic representation of enteric coated system Layered systems.
These are one or two impermeable or semi-permeable polymeric coatings (films or compressed) applied on both sides of the core. To allow biphasic drug release, a three- layer tablet system was developed. The two layers both contain a drug dose. The outer drug layer contains the immediately available dose of drug. An intermediate layer, made of swellable polymers, separates the drug layers. A film of an impermeable polymer coats the layer containing the other dose of drug. The first layer may also incorporate a drug- free hydrophilic polymer barrier providing delayed drug absorption. Conte et al has also studied a multi-layer tablet system (Geomatrix consists of a hydrophilic matrix core containing the drug dose. This kind of three-layer device has been used in the treatment of Parkinsonian patients using L--dopa/benserazide. Night-time problems and early-morning symptoms of Parkinsonism can be avoided by using a dual-release Geomatrix @ formulation, which allows daily doses of drug to be reduced and leads to extent of bioavailability 40% greater than when a traditional controlled release formulation is employed.
c. Time-controlled explosion systems (TES):
These have been developed for both single and multiple unit dosage forms both cases, the core contains the drug, an inert osmotic agent and suitable disintegrates. Individual units can be coated with a protective layer and then with a semi-permeable layer, which is the rate controlling membrane for the influx of water into the osmotic core. As water reaches the core, osmotic pressure is built up. The core ultimately explodes, with immediate release of the drug. The explosion of the formulation can also be achieved through the use of swelling agents. Lag time is controllable by varying the thickness of the outer polymer coating.
d. Sigmoidal release systems (SRS):
For the pellet-type multiple unit preparations, SRS containing an osmotically active organic acid have been coated with insoluble polymer to achieve different lag-times. By applying different coating thicknesses, lag times in vivo of up to 5 hours can be achieved. Release rates from SRS, beyond the lag time, has been found to be independent of coating thickness.
4. CHRONOTOXICITY
Chronotoxicity is refers as the study of time dependent toxic effects of drugs. Drugs show toxic effects according to circadian rhythm. Some examples of chronotoxicity are following:-
* Chronotoxicity of the cyclooxygenase-2 inhibitor, celecoxib:
Inhibition of the enzyme cyclooxygenase with celecoxib is cytotoxic in a variety of solid tumor cell lines. Previous work has shown that by charting circadian rhythms, it has been possible to find optimal times to deliver a dose of drug, such that it is most efficacious in killing cancer cells and least harmful to normal tissues. Therefore, we examined the time dependence of toxicity (chronotoxicity) and of antitumor effects (chronotherapy) of celecoxib to determine optimal time of day for dosing with respect to light-dark cycles. There was no clear relationship between dose administered and therapeutic response.
* Time-dependent nephrotoxicity associated with daily administration of cisplatin:
Chrononephrotoxicity induced by daily cisplatin administration does not only depend on cisplatin accumulation, but might also depend on kidney sensitivity to diurnal variations in inflammatory reaction without direct cisplatin toxicity.
* Cell cycle-dependent chronotoxicity of irinotecan hydrochloride:
The loss of body weight after an intraperitoneal injection of irinotecan hydrochloride (CPT-11,7-ethyl-10-[4-(1-piperidino)-1-piperidino]carbonyloxycamptothecin) 100mg/kg) was more serious in the late dark and the early light and milder in the late light and the early dark. The CPT-11-induced leucopenia was more serious in the late dark and milder in the late light. The lower toxicity of CPT-11 was observed when DNA synthesis and type I DNA topoisomerase activity in bone marrow cells decreased and the higher toxicity was observed when these activities began to increase. There were circadian stage- dependent changes in the concentrations of CPT-11 and its major metabolite (SN-38; 7- ethyl-10-hydroxycamptothecin) in plasma. The higher concentrations of CPT-11 and SN- 38 in plasma were observed when the level of CPT-11- induced toxicity increased.
* Chronotoxicity of methotrexate:
Methotrexate (MTX), an effective agent in treatment of cancer, is one of the most versatile antineoplastic agents in spite of severe toxicity problems.The purpose of this study was to determine the circadian variation of this toxicity in order to decrease the side effects.
* Chronotoxicity of glufosinate ammonium:
The effect of a circadian-stage dependent dosing schedule on the toxicity of glufosinate was studied in mice. A highly significant circadian rhythm occurred in the resulting mortality, with the highest mortality from doses given during the light phase and the lowest from doses administered during the dark phase. The circadian-stage dependent dosing schedule had a marked influence on the pattern of acute glufosinate toxic.
* Chronotoxicity of cyclophosphane:
Study of the chronotoxicity of cyclophosphamide injected to mice at 18, 24, 6 and 12 o'clock has shown that animals kept under the conditions of natural changes of day and night showed the circadian rhythm of the drug toxicity with the maximal survival of the animals after injection at 24 and 6 o'clock and with the minimal survival after injection at 18 o'clock. In animals maintained under the conditions of artificial constant light the toxicity rhythm was perversed within the first hours after injection and improved on subsequent observation. Moreover the earlier death was marked in these animals.
* Chronotoxicity of amikacin:
Amikacin toxicity exhibited a peak at 2 h (mean 60%) and nadir at 14 h (mean 47.75%) in november/december, whereas the opposite was true in spring (means 36.6% and 23.3% respectively at 14 h and 2 h). Thus, acute toxicity of amikacin in mice varies throughout the circadian cycle and from season to season.
* Irinotecan chronotoxicity:
In mice, minimum body weight loss is when administered at 5 pm and maximum weight loss occurred when the drug was administered between 05: OO-O9: 00 pm. Recovery from subsequent weight loss was found to be faster when injected in the late light and early dark than late dark and early light. Inhibition of acetyl cholinesterase by irinotecan causes early defaecation, diarrhoea, vomiting. Acetyl cholinesterase activity decreases in late dark and increases in late night. Maximum body weight loss occurs when acetylcholine levels increase, minimum loss occurs when acetylcholine levels decrease. Lrinotecan induced leucopenia- more serious in the late dark and milder in late light.
5. CLINICAL APPLICATIONS
A. Clinical Chronopharmacology of Intrathecal Sufentanil for Labor Analgesia:
Pharmacokinetic and pharmacodynamic characteristics of opioids vary throughout the day, as demonstrated for oral morphine in chronic pain. However, little is known about the chronobiology of intrathecal lipid soluble opioids used for labor analgesia. The aim of this prospective study was to determine whether the duration of action of intrathecally administered sufentanil is influenced by the time of administration.
The duration of intrathecal sufentanil analgesia exhibited a temporal pattern with 30% variations throughout the day period. The authors point out that the lack of consideration of chronobiological conditions in intrathecally administered analgesia studies can cause significant statistical bias. Further studies dealing with intrathecal opioids should consider the time of drug administration.
B. Clinical Chronopharmacology of Cardiovascular Disorders:
Physiological functions as well as pathophysiological events--angina pectoris, myocardial infarction and sudden cardiac death--display pronounced circadian rhythms. Clinical studies also give evidence for a circadian phase dependency in the pharmacokinetics of cardio-vascular active drugs. Ambulatory blood pressure assessment reveals marked circadian rhythms in blood pressure both in normotensive persons and hypertensive patients, whereas Holter monitoring substantiates day-night patterns in electrocardiographic events of patients with ischemic heart disease.
In most normotensives and hypertensives, blood pressure (BP) drops by 1 0-20% during sleep. Early morning increase in BP is associated with increased vascular tone, platelet aggregation and decrease in intrinsic thrombolytic activity, this favors thrombus formation and a lower is Chaemicthrs hold. In 70% cases of secondary hypertension (renal diseases, hyperthyroidism, gestational hypertension), the rhythmic pattern is abolished or even reversed.
Blood pressure and heart rate in normotensives and essential (primary) hypertensive patients display highest values during daytime followed by a nightly drop and an early morning rise. In about 70% of forms of secondary hypertension (e.g. renal disease, hyperthyroidisms, hormonal diseases, gestational hypertension), however, this rhythmic pattern is abolished or even reversed exhibiting nightly peaks in blood pressure. This form of hypertension is accompanied by increased end organ damages. Thus, different subtypes of a disease (angina pectoris, hypertension) can display different circadian patterns in symptoms. These observations are a challenge for basic and clinical research to get a better understanding on the underlying mechanisms of regulation. Moreover, they call for a circadian time-specified drug treatment.
Epidemiologic studies document the heightened morning-time risk of angina, myocardial infarction, and stroke. Circadian rhythms in coronary tone and reactivity, plasma volume, blood pressure, heart rate, myocardial oxygen demand, blood coagulation, and neuroendocrine function plus day-night patterns in the nature and strength of environmental triggers all contribute to this morning vulnerability. Circadian pattern of low BP during night (resting span) - rather than day (active span) is called as dipper and inverse 'relationship is called as no dipper. Marked nocturnal fall of more than 80% of systolic BP is more likely to have advanced silent cerebrovascular damage than dipper or nondipper.
From above it is evident that pharmacokinetics may also not be constant within a day. Chronopharmacokinetics have been shown for several cardiovascular active drugs (propranolol, nifedipine, verapamil, enalapril, isosorbide-5-mononitrate, digoxin, etc.). Far more drugs were shown to display significant daily variations in their effects (chronopharmacodynamics, chronotoxicology) even after chronic application or constant infusion.
* Aspirin:
Maximum antiparticle effect is shown in the morning, which is two times more than any other period of the day.
* Furosemide:
Administered at 9 pm shows increase in urine volume and increased excretion of Na+, CI' during first 60 minutes than when drug is administered at 9 am. Elevation of blood glucose concentration is more after an evening dose.
* Thrombolytics and Heparin:
Minimal benefit for ischaemic events is during the early morning hours.
* Labetolol:
It is more successful in controlling early morning rise in BP.
C. Clinical Chronopharmacology of Psychotropic Drugs:
The effects of many kinds of psychotropic drugs have been shown in animal studies to follow a circadian rhythm. Trials for the clinical application of this circadian rhythm have already been undertaken. Although the mechanisms underlying this phenomenon are still unclear, chronological changes in the levels of drugs in the blood and brain suggest that it is primarily due to rhythms in the brain's susceptibility to drugs. Rhythms are present in the level of intracerebral neurotransmitters, receptors and second messengers. Each of these rhythms may cause other rhythms within each system of neurotransmitters, which in turn induces a rhythm in the susceptibility to drugs.
The conception of the specific activity of antidepressants is considered from the chronobiological positions. Depression can be based on phasic dissociation between different periods and first of all circadian fluctuation of some physiological functions. Various antidepressants modified biorhythms and promoted their resynchronization. This action probably depended on its influence on the activity of the cerebral pacemaker structures (SuprachtAsmatic nuclei, hypothalamus and pineal gland).
D. Clinical Chronopharmacology with Special Reference to NSAIDs:
Both drug disposition and effects vary as a function of dosing time. A chronotherapeutic study was therefore undertaken with sustained-release indomethacin (ISR) to quantitate any potential therapeutic gain from an optimal time of administration.
At group level, time-dependent differences in drug effectiveness were small, although large inter-individual differences were documented. Evening ingestion was most effective in patients with predominant nocturnal or morning pain; conversely, morning or noon ingestion was most effective in patients with maximum afternoon or evening pain. A four-fold improvement in tolerance and a doubling of analgesic effectiveness resulted from varying the ingestion time. Chronotherapeutic studies should therefore include careful longitudinal documentation of pertinent marker rhythms such as self-rating of pain.
* Amitriptyline Chronopharmacology:
Side effects of decreased salivation and sedation were more marked when a single dose of amitriptyline was taken orally in the morning than in the evening. These dynamic differences were due to alteration in kinetics. Absorption of the drug was more rapid in the morning, although other kinetic parameters, especially total bioavailability, were unchanged. Thus, in the case of this drug, chronopharmacologic differences were due to a change in rate of absorption. The present practice of giving a single dose of drug in the evening is justified on the basis that it induces fewer side effects without a loss in therapeutic efficacy.
* Furosemide chronopharmacology:
Twelve healthy males participated in a trial to determine whether furosemide diuresis and its effects on electrolyte excretion patterns, aldosterone concentrations and renin activity are subject to diurnal variations. Six treatments were giving: placebo at 07h00, 13h00 and 19h00 and furosemide 40 mg at 07h00, 13h00 and 19h00. Each subject participated in six phases. It was found that the time of administration of furosemide does not affect the effect of the drug on sodium and potassium excretion and diuresis. There is a tendency for furosemide to elevate serum aldosterone levels and renin activity regardless of the time of administration.
E. Clinical Chronopharmacology of Bronchial Asthma:
Episodes of difficulty in breathing peak at 4 am 70% of all deaths from asthma occur between midnight and 8.00 am. Bronchial potency peaks at 4.00 pm, decreases during sleep and reaches nadir at 4.00 am.
Asthmatic's overnight fall in potency is exaggerated from 25% to 50%. In asthmatics, increase in resistance is much greater during sleep be cause of sleep associated reduction in inspiratory muscle tone decrease in pulmonary compliance increased intrapulmonary' blood pooling, which promotes airway narrowing.
The circadian rhythm of biological rhythms is particularly important in understanding the declined changes in lung function of asthmatics at night.
Chronotherapy of Corticosteroids:
Long-term oral administration of corticosteroids at 8:00am and 3:00pm are more effective in controlling nocturnal asthma than same dose at3.00pm and 8.00pm. Oral corticosteroids should be administered around 3 PM to achieve peak pulmonary anti- inflammatory efficacy between 3 AM and 6 AM corticosteroid-induced decreases in inflammatory cell infiltration in lungs clearly being associated with improvement in lung function.
Chronotherapy of Oral Nitrates:
The clinical-chronopharmacological investigations with oral nitrates (ISDN, IS-5-MN) demonstrate that the drugs; pharmacokinetics and/or hemodynamic effects are circadian phase-dependent. For both an immediate-release and a sustained-release preparation of IS-5-MN peak drug concentrations coincided with peak drug effects after morning but not after evening drug application. Results indicate a circadian phase-dependency in the dose- response relationship of oral nitrates.
Chronotherapy of Theophylline:
Chronotherapy of a once-daily evening dose of a new controlled-release theophylline preparation, which achieves to peak blood concentrations at 10-12 hours after dosage, effectively improved the values of PEF and symptoms of nocturnal asthmatics. Single night time dose of theophylline is recommended for nocturnal bronchospasm because reduction in FEV, is, only 9% compared to 28% with two or more doses. Evening dosing of special drug delivery forms of theophylline and morning methylprednisolone administration has proven to be beneficial.
Controlled-release theophylline preparations, such as Uniphyl, which achieve peak blood levels 10-12 hours after dosage, should be administered immediately after the evening meal to give greatest efficacy between 3 and 6 AM when airflow reaches its nadir. Theophylline inhibits the late asthmatic response to nighttime antigen challenge in patients with mild atopic asthma. Inhaled antigen at night causes a more pronounced late asthmatic response (LAR) when compared with daytime challenges. Chronopharmacology with controlled-release theophylline given in the evening leads to a peak serum theophylline concentration (STC) in early morning which coincides with LAR that follows an evening challenge. Nocturnal administration of controlled-release theophylline increases the tolerance to inhaled antigen and reduces severity of LAR. Because the LAR is linked to airway inflammation, these data support the possibility of anti-inflammatory effects associated with theophylline use.
A 24-h variation was also observed in albuterol-induced bronchodilation in patients with NA: maximal effectiveness occurred at night, and lower effect was obtained with the midday administration. The albuterol-induced increases in FEV1 (forced expiratory volume in 1) were not clinically significant in children without nocturnal asthma except when the beta 2-agonist was inhaled between 10 p.m. and 2 am.
F. Clinical Chronopharmacology of GIT Disorders:
This chapter discusses the influence of ultradian and circadian rhythms of gastrointestinal motor and secretory function on the action of orally administered drugs. Most drugs exhibit more rapid absorption in the morning compared to the evening due, in part, the circadian alterations in gastric emptying. Gastric acid secretion and gastrointestinal toxicity to oral drugs also display circadian rhythmicity. These observations provide a rationale for use or avoidance of drugs based on time-of-day dosing considerations. The chronopharmacological behavior of a drug may thus play an important role in the effectiveness of any oral medication treatment schedule.
G. Clinical Chronopharmacology of Gastro esophageal reflux disorders:
Daytime symptoms arise from meal triggered acid secretion. Night time symptoms result from Circadian rhythm of acid production that peaks between 10 pm and 2 am. Omeprazole when given only in the morning before breakfast, or in two divided doses- Morning and evening or only at night before dinner, are all equally effective in preventing acid production during daytime but only BD and 00 (evening dose) are effective in pre venting night symptoms.
Chronopharmacology Ulcer:
Under normal conditions, peak acid secretion occurs when other rhythms such as secretion of acid buffering substances, thickness of the protective lining and flow of, blood, naturally protects the mucosa. Ulcer pain is worst at night, usually 2 hours after food, wakes the patient, in the night around 2 am. Stomach empties 2 hours after eating, residual acid and enzymes initiate ulcer, causing pain and discomfort. Also acid secretion peaks; at night between 10 pm and 2 am. Antacids give temporary and partial relief by neutralizing stomach acid. Ulcer healing is directly related to how well acid secretion is inhibited during nighttime.
H. Clinical Chronopharmacology of Arthritis:
Rheumatoid arthritis (RA), osteoarthritis (OA), ankylosing spondylitis (AS), and gout exhibit profound circadian rhythm in manifestation and intensity of symptoms.
Chronotherapy of Rheumatoid arthritis:
It is chronic inflammatory autoimmune disorder with cardinal signs - stiffness, swelling and pain of one or more joints Severity of cardinal signs is three times more between 08:00 and 11:00 am. Hand strength is lower by 30% in the morning than at night. Long acting NSAIDs like flurbiprofen, ketoprofen, indomethacin at bedtime ensure adequate control of morning symptoms of RA.
Chronotherapy of Osteoarthritis:
Is a degenerative disease of the joints that affects nearly everyone at least to some degree by 70 yrs. of age? Pain is more intense between 2pm and 8pm. The temporal pattern of pain and stiffness varies from person to person. Individualized chronotherapy is necessary, like once a day ketoprofen, indomethacin in relation to time of day when pain is worst. If pain is worst at night evening dose is recommended but If pain is worst in afternoon- morning dose is recommended.
Chronotherapy of ankylosing spondylitis:
It is an inherited disorder, more common in males than in females; complaints are swelling and discomfort of the joint of back) Pain intensity is 2-3 times greater and stiffness 8 times more between 06:00 and 09:00am. Second, but less prominent peak, occurs between 7:00 and 9:00 pm. Backache and stiffness is 12 times more frequent in winter than summer.
Chronotherapy of Arthritis with:-
Corticosteroids:Severe forms require treatment with corticosteroids such as prednisolone, methyl- prednisolone, and triamcinalone, to control both pain and stiffness. Long-term administration leads to suppression of endogenous corticosteroids, cataract formation, ulcers, osteoporosis, and mood alteration. Evening or nighttime dose results in maximum cortisone inhibition. Morning or early afternoon once a day schedule is least detrimental 60- 70% of RA patients benefited to a great extent from the 01:00 pm dose than from 08:00 am dose. Chronic corticosteroid therapy is, or has been, a major therapeutic component for many RA patients who consequently are unable to respond adequately to the stresses of general anesthesia and surgery because of complete or partial adrenal insufficiency. These patients must be given appropriate supplemental corticosteroid therapy perioperatively.
Ketoprofen:
Solid dispersion of ketoprofen was found to be more effective in inhibiting progression of RA. The protective effect of ketoprofen and its solid dispersion was significantly higher when these were administered at 0800 hrs. Thus, it was observed that in the adjuvant induced arthritis model, inflammation and articular damage was significantly greater in the rest period of diurnally active rats than in the activity phase. KPF and its solid dispersion showed better protection from inflammation in the morning than in the evening.
NSAIDS:
Aspirin, non acetylated salicylates, and numerous other nonsteroidal anti-inflammatory drugs (NSAIDs) are used in rheumatoid arthritis (RA) patients to decrease joint inflammation and improve function. Equivalent doses of aspirin and of non acetylated salicylates are equally anti-inflammatory in RA, although the non acetylated salicylate is a poor inhibitor of prostaglandin synthesis.
Chronopharmacology studies suggest that many patients may have better efficacy and fewer side effects with evening doses than with morning doses of certain NSAIDs. The gastric, renal, and platelet adverse effects of NSAIDs are related to their inhibition of prostaglandin synthesis, and tend to be related to dose and intensity of therapy. Various strategies can minimize the impact of these side effects, such as co administration of gastric protectants or the use of short half-life NSAIDs to decrease the duration of preoperative NSAID withdrawal needed to ensure adequate platelet coagulation during surgery.
I. Clinical Chronopharmacology of Sleep Disorders:
Disturbance in circadian rhythm often result in disturbances in sleep. Pineal hormone melatonin secretion peaks during night and its release could be suppressed by exposure to light. Delayed sleep phase syndrome (DSPS) -It is a disorder in which the patient goes to bed around 4 am and wakes up around noon. This may occur after a period of late night studying, partying, or after evening or night work shifts.
Treatment:
a. Improve sleep habits.
b. Bright light therapy: This therapy needs to take total control of light and dark expo sure across whole day. Patient uses bright light exposure early in the morning and avoids light in the evening hours upon rising early in the morning, patient sits in front of a light box that emits 2500 lux- this produces an increase in alertness in one week.
c. Chronotherapy: Behavioral technique in which bedtime is systematically delayed by 3hourseach day. Procedure is maintained until the desired bedtime is reached. e.g., Day 1: sleep 4am to noon Day 2: sleep 7 am to 3:00pm Day 7-13: sleep 10pm to 6:00 am Day 14 and thereafter: sleep 11 pm to 7:00 am.
Advantage: Drug free, works rapidly.
Disadvantages:
1. Staying wake until next bedtime schedule.
2. Patient may wake up before the intended time and may be unable to sleep at the desired
time schedule.
Advanced sleep phase syndrome (ASPS)
This is a condition where a person goes to sleep an hour before sunset and is wide-awake
at 04:00 am.
Jet lag: Sleep disturbance during first night after a transmeridian flight. Period of asynchrony occurs because the traveler’s internal clock is set to early time zone and traveling across meridian imposes a new day- night phase. Resynchronization takes 24 hours and as a
consequence, individual suffers from insomnia, fatigue and irritability. Gastrointestinal disturbances occur in 60%. Delayed ovulation and associated mental disturbances occur in 30-35%. Treatment - melatonin and light therapy. Melatonin 0.5 mg/day maintains synchronization.
Rotating shift work: Impaired physical and mental performance results as sleep deprivation, inability to pay attention to minute but often-important details. Dreadful disasters have happened possibly because of this e.g., disaster of Three Mile Island, Chernobyl and Bhopal that occurred between midnight and dawn were regarded as errors committed by workers -not synchronized to work in night shifts.
J. Clinical Chronopharmacology of Skin Disorders: Some cutaneous functions of importance are governed by periodical variations according to the nycthemeral timing. Similarly to other organs, the skin is under the influence of a coherent organization of circadian and nythemeral rhythms modulating various biological cycles which usually display a wide amplitude. The cutaneous biorhythms in humans suggest that during daylight the skin boosts diverse protective functions with regard to environmental threats. In the evening and at night, the skin increases its renewal and diverse metabolic processes. Biorhythms in the skin which are in line with the organism's needs merit to be recognized. Chronophysiology, chronopathology and clinical chronopharmacology may help reach optimal therapeutic decisions. Proliferation of skin cel/svaries up to 30 folds in 24 hours, being greatest at midnight and least at noon. Oil production by skin glands is twice as great at noon than between 02:0004:00 am. Skin is more acidic during sleep than around midday.
Atopic dermatitis: Itching is most intense late in evening because skin sensitivity to histamine is highest at night.
Psoriasis: Cell proliferation rate of the affected skin is more than normal skin. Cell proliferation rate in areas of psoriasis is highest between 9 pm and 3 am and least between 09:00 and 11:00 am. In dermis, cell proliferation is maximum at 9:00 am and least at 3:00 am. Inflammatory activity is highest at night and least in the morning.
K. Clinical Chronopharmacology of Cancer: Cancer is regarded as a disease of malfunctioning internal clock because cancer cells lose their internal timekeeper and divide more rapidly than normal cells. The chronomodulation of anticancer drugs arouses our interest as a method for cancer chemotherapy. Recent clinical studies reviewed regarding circadian rhythms in 1) target tissues: healthy and cancer tissues; 2) chronopharmacology of anticancer drugs; and 3) chronotoxicity of anticancer drugs. A brief review is made of the main results of clinical phase II trials.
Studies suggest that chronobiological cycles for normal cells and tumour cells are different. Therefore treatment goal is to time the administration of cancer drugs to the chronobiological cycles of tumour cells, making it more effective against cancer cells and least toxic to normal tissues Studies in experimental models are required to understand the relation between tumor rhythms and antitumor treatments efficacy. In healthy tissues, cell proliferation, and differentiation processes are regulated precisely and exhibit marked circadian rhythmicity. Experimental and human tumors can retain circadian rhythms or display altered oscillations. Healthy tissues can also display rhythm modifications, possibly related to cancer stage. Cellular rhythms modulate the metabolism of cytotoxic agents and the cellular response to them; hence, they determine the chronopharmacology of anticancer drugs.
Circadian rhythms in host tolerability and/or cancer chemotherapy efficacy have been demonstrated with nontoxic doses of drugs in several experimental tumor models, while in other ones a circadian-time effect was only seen within a specific dose range. The usual coupling between tolerability and efficacy rhythms of anticancer agents has resulted in significant improvement of their therapeutic index. Results of laboratory animal studies have been extrapolated to the design of clinical cancer therapy trials involving a chronobiological approach.
Many physiological variables in hemato-oncology, e.g. the function of liver and kidney and hematopoiesis, show circadian changes. Consequently, the metabolism, elimination and myelotoxicity of cytostatic drugs are subjected to circadian variations in animal and man. Moreover, preliminary clinical data suggest that an optimal circadian timing may even increase the efficacy of anticancer chemotherapy. The recent development of programmable infusion pumps provided a reliable tool for the application of cytostatic drugs at any time of day.
Mechanism of Chronotherapy of Cancer: The mechanisms underlying the dosing time-dependent change in the antitumor effect of interferon-beta (IFN-beta) were investigated based on the sensitivity of tumor cells and the pharmacokinetics of the drug. Tumor-bearing mice were housed under standardized light-dark cycle conditions (lights on at 7:00 AM, off at 7:00 PM) with food and water available ad libitum. The antitumor effect of IFN-beta (0.5MI.U./kg, intratumoral) was more efficient in early light phase than in early dark phase. The higher antitumor effect of IFN-beta was observed when specific binding of IFN receptor and DNA synthesis in tumor cells increased, and the lower effect was observed when these levels decreased. The dosing time-dependent effect of IFN-beta was supported by the time-dependent expression of transcription factor (signal transducers and activators of transcription 1) and cell proliferation inhibitor (p21 wild-type p53-activated fragment 1) protein induced by IFN-beta. There was a significant dosing time-dependent change in IFN-beta concentration in tumor, with a higher level in early light phase and a lower level in early dark phase. The dosing time-dependent change of IFN-beta concentration in tumor was associated with that of IFN-beta-induced antitumor effect.
These results suggest that by choosing the most suitable dosing time for IFN-beta, the efficacy of the drug can be increased in certain experimental and clinical situations. Circadian changes in enzymatic activities involved into fluoropyrimidine (5-fluorouracil- 5-FU, or floxuridine-FUdR) catabolism (dehydropyrimidine dehydrogenase) or anabolism (thymidine kinase), rhythms in reduced glutathione, involved into cellular protection against cytotoxic effects of alkylating drugs, platinum complexes and anthracyclines, rhythms in cellular proliferation of rapidly renewing tissues, such as bone marrow or intestinal tract. Furthermore, chemotherapy injection at the least toxic time allows increasing its antitumor efficacy against several transplanted rodent tumor models. Extrapolation of these results for improving therapeutic index of chemotherapy in cancer patients was based upon the hypothesis that rhythms in metabolism or proliferation of healthy tissues were tightly coupled to the circadian sleep-wakefulness cycle, both in rodents and in man).
Chronotherapy of lung carcinoma: In nonsmall cell lung cancer (NSCLC) circadian rhythms in cell kinetics, immunological and endocrinological endpoints, and tumor markers are reviewed. Chronopharmacology and chronotolerance studies on laboratory animals and clinical observations on cancer patients involving the main drugs active in lung carcinoma have prompted prospective Phase I-III studies to evaluate the application of chronobiologic concepts in the treatment of NSCLC. Circadian rhythmicity in host tolerance (chronotolerance) to medications has been confirmed in two prospective randomized lung cancer trials; a large phase II study has established the value of chronobiologic concepts to improve the therapeutic index of advanced NSCLC.
Circadian Timing and Cancer Surgery: Cure of breast cancer is more likely if surgery is performed in the middle of menstrual cycle or following ovulation. In first half of cycle oestrogen increases, in second half progesterone increases. It is believed that progesterone may inhibit the production of some enzymes that help in cancer spread.
Chronotherapy of Various Drugs: Circadian rhythm is described for 5fluorouracil, cisplatin, oxaliplatin, 6 mercaptopurine, doxorubicin and methotrexate.
Doxorubicin: It has maximum anticancer activity when given before normal daily awakening or in the last half of the daily sleeps spans.
Cisplatin: Maximum toxicity and kidney damage is in morning; better tolerated when given late in the animals active phase.
Oxaliplatin: The fully ambulatory nature of treatment courses was an additional constraint put on chronotherapy. More than 1000 patients with metastatic gastrointestinal malignancies have now received chronotherapy protocols in several countries from North America or Europe. Results have clearly indicated that this approach improved chemotherapy tolerance and allowed safe increases in drug doses. A clinical phase III trial compared a flat versus the chronomodulated three-drug regimen, and demonstrated large,
simultaneous improvements in both tolerability and response rates in patients with metastatic colorectal cancer receiving chronotherapy. This approach may also be beneficial to patients with other gastrointestinal malignancies, and it was amenable to combination with surgery and radiotherapy. It also appeared suitable for devising potentially more active dose-intensive yet safe regimens. Incorporation of chronopharmacology into the development stages of new drugs may improve their safe use to the greatest benefit of both cancer patients and new drug development, as was done for oxaliplatin.
Fluorocil (5FU): Best timing for 5FU is in the rest phase, several hours prior to usual awakening. It is much safe when given between mid-sleep and 2 hrs prior to usual awakening.
Combined Therapy: CT is more effective in controlling cancer. Doxorubicin and cisplatin should be separated by 12 hrs with doxorubicin given in early morning e.g., 06:00 am and cisplatin given 12 hrs. Later at 06: 00pm for a patient on a normal sleep- wake schedule. Used in rectal cancer, pancreatic adenocarcinoma, and renal cell carcinoma.
L. Clinical Chronopharmacology of Diabetes: Insulin is normally secreted in humans in regular pulses every 5-15 min. In on-diabetics decreased glucose tolerance occurs in the latter part of the day because of:
*Decreased utilisation (2-fold reduction) in the latter part of the day.
*Reduced insulin sensitivity in the afternoon and evening. In case of NIDDM, glucose tolerance increases from morning to evening. It is well established that higher dose of insulin should be delivered for breakfast than for a later meal.
*Dawn phenomenon:
Insulin clearance increases in the early morning hours.
*Insulin availability decreases during the second half of the night because at" waning of insulin injected s.c.
*Increased hepatic glucose production starts around the middle of night (03: 00 am).
*Despite the presence of hyperglycemia, glucose utilisation does not begin to increase until the early morning and increase in utilisation is notcommensuratewith-accelerated rate of production. It is thus mismatch (in time and size) between increases in glucose. Duction and decreased utillistation that is responsible for mirning hypergluycaemia. Nocturnal insulinisation– portable pump is where delivery rate can be precisely adjusted to the variation in glucose levels.
*To avoid hypoglycaemia at night, it is recommended to split the evening dose of insulin by taking short acting insulin at dinner and long acting dose at bedtime, instead of both being mixed at dinnertime.
Alcohols Interaction with Circadian Rhythms: Alcohol related disorders seem to be clarified with the main concepts of chronobiology. The complex interaction between alcohol and the body's circadian rhythm has become a rapidly expanding area in chronopharmacology. This area has key implications for the field of alcohol research, because understanding alcohol's effects on the body's internal clock will aid scientists in designing medications and behavioral interventions for treating alcohol abuse and dependence.
A number of studies provide evidence that alcohol sensitivity and preference vary with circadian timing. However, only a few studies support alcohol's ability to influence the circadian phase directly. This review focuses on studies examining how alcohol and the body's circadian rhythm interact, using body temperature as an index of circadian rhythm function. Findings indicate that alcohol sensitivity and preference for drinking to indeed appear to vary with circadian timing and that alcohol may act directly on the central pacemaker to alter circadian functioning.
Thus chronopharmacology (especially chronokinetics and time-related differences in drug effects) clears up variations of blood alcohol levels and toxicity of ethanol during the 24 hrs.
M. Clinical Chronopharmacology of Renal Physiopathology: This review presents our recent knowledge in renal chronobiology, considering physiology, pathology, pharmacology and toxicology. In a first part, are described renal chronophysiological data, pointing out particularly urinary excretion rhythms, largely described for last century? Such physiological observations lead to conclude that renal structures and functions present large temporal variations. Endogenous and exogenous origins of these rhythms are widely discussed. Evidence of such circadian and circannual variations in these renal structures and functions permit to understand why different agents, pathogeneous, toxic or medicamentous will present time-dependent effects at
renal level. So, a second part presents some recent experimental and clinical data in chrononephrology, chronopharmacology and chrononephrotoxicity. Finally, the concept of renal chronesthesy is presented and discussed, in order to explain these temporal variations in renal target susceptibility to different substances acting at this level.
N. Clinical Chronopharmacology of Hematology and Immunology: The hematopoietic and the immune systems in all their components are characterized by a multifrequency time structure with prominent rhythms in cell proliferation and cell function in the circadian, infradian, and rhythms in cell proliferation and cell function in the circadian, infradian, and circannual frequency ranges. The circulating formed elements in the peripheral blood show highly reproducible circadian rhythms. The timing and the extent of these rhythms were established in a clinically healthy human population and are shown as chronograms, cosinor summaries and, for some high-amplitude rhythms, as time-qualified reference ranges (chronodesms). Not only the number but also the reactivity of circulating blood cells varies predictably as a function of time as shown for the circadian rhythm in responsiveness of human and murine lymphocytes in vitro to lectin mitogens (phytohemagglutinin and pokeweed mitogen).
Some circadian rhythms of hematologic functions appear to be innate and are presumably genetically determined but are modulated and adjusted in their timing by environmental factors, so-called synchronizers. Phase alterations in the circadian rhythms of hematologic parameters of human subjects and of mice by manipulation of the activity-rest or light- dark schedule and/or of the time of food uptake are presented. Characteristically these functions do not change their timing immediately after a shift in synchronizer phase but adapt over several and in some instances over many transient cycles. The circadian rhythm of cell proliferation in the mammalian bone marrow and lymphoid system as shown in mice in vivo and in vitro may lend itself to timed treatment with cell- cycle-specific and nonspecific agents in an attempt to maximize the desired and to minimize the undesired treatment effects upon the marrow. Differences in response, and susceptibility of cells and tissues at different stages of their circadian and circaseptan (about 7-day) rhythms and presumably of cyclic variations in other frequencies are expected to lead to the development of a chronopharmacology of the hematopoietic and immune system. Infradian rhythms of several frequencies have been described for numerous hematologic and immune functions.
CONCLUSION
Regular and thus predictable changes in biologic susceptibility and response to a large variety of physical as well as chemical agents can now be viewed as rather common phenomena. Chronopharmacology involves both the investigation of drug effects as a function of biologic timing and the investigation of drug effects upon rhythm characteristics (the period, tau, the acrophase, phi, the amplitude, a, and the mesor, M). Illustrative examples of circadian (tau congruent to 24 hrs) chronopharmacology in man are summarized in up-dated tables, keeping in mind that the objective demonstration of chronopharmacologic facts demands the use of an appropriate methodology. Circadian changes in the effects of various chemical agents have been documented: histamine, sodium salicylate, acetylcholine, halothane, prostaglandin F2alpha, reserpine, cyproheptadine, ethanol, insulin, chlorothiazide, oxymetholone, orcinprenaline and SCH 1000 (the latter being bronchodilators), Indomethacin, lignocaine, ACTH, cortisol and various synthetic corticosteroids. Despite that pharmacological facts are well documented in man for circadian rhythms, studies are not only restricted to the 24-h period but have been already extended to rhythms of periods of e.g. approximately 30 days as well as approximately 1 year. For a better understanding of chronopharmacologic findings, three new concepts must be considered: a) the chronokinetics of a drug, defined as both rhythmic (circadian) changes in the drug bioavailability (and/or pharmacokinetic effects) and its excretion (urinary, among others); b) the chronesthesy of a biosystem to a drug, or the circadian change in the susceptibility of any biosystem to a drug (including organ systems, tumors, parasites, etc.); and c) The chronergy of a drug, or the rhythmic change in the overall effects and the effectiveness of a drug. This term takes into consideration the chronokinetics and the chronesthesies of the organismic biosystems involved. Presently, one of the aims of chronopharmacology is to solve problems of drug optimization, i.e. to enhance the desired efficiency of a corticosteroid or other medications, while reducing undesired effects. The metabolic pathways are neither open permanently, nor open with a constant patency along the 24-h scale, among other bioperiodic domains. Thus, the chronobiologic approach concerning pharmacologic phenomena involves a lesser risk of errors and/or false information than the conventional homeostatic approach.
References:
1. Reinberg A E., “Concepts in Chronopharmacology”, 2 nd , Annu Rev Pharmacol, Toxicol,
1992, Page no. 51-66.
2. Abrahm J., “Timing treatment to the rhythm of disease”, Postgrad med, 2001, Page no.
108- 109.
3. Martin R J, Banks–Schlegel S., “Chronobiology of asthma”, M J Respir Crit Care Med,
1998, Page no. 702-712.
4. Anwar Y, White W B., “Chronotherapeutics for cardiovascular Disease”, Drugs 1998,
Page no. 631-643.
5. Daga M K, Prabash K., “Chronopharmacology and Chronotherapeutics”, JAPI 2000,
Page no. 17-21.
6. Lemmer B, Labrecque G. C., “Chronopharmacology and Chronotherapeutics” Chronobiol
Int 1987, Page no. 319-329.
7. Millar–Craig M W, Bishop C N, Reftery E B., “Circadian variation of blood– pressure”,
Lancet 1978, Page no. 795–799.
8. Van C E, Polonsky K S, Scheen A J., “Roles of circadian rhythmicity and sleep in human
glucose regulation”, Endocr Rev 1997, Page no. 716-738.
9. Evans RM, Marain C., “Taking Your Medication: A Question of Timing”, American
Medical Association, Chicago, IL, Page no. 1996. 3-8.
10. Ura J, Shirachi D, Ferrill M., “The Chronotherapeutic Approach to Pharmaceutical
Treatment” California Pharmacist. 1992, Page no. 46-53.
11. Lamberg L., “Chronotherapeutics: implications for drug therapy”, American Pharmacy
1991, Page no. 20-23.
12. Smolensky M H, Labreque G., “Chronotherapeutics Pharm. News”, 1997, 10-16.
Fujimura A, Tateishi T, Ohashi K, Ebihara A., “Differences of chronopharmacokinetic
profiles between propranolol and atenolol in hypertensive patients”, J Clin Pharmacol,
1993, Page no. 756-761.
13. Lemmer B, Portaluppi R., “Chronopharmacology of Cardiovascular Diseases”,
Physiology and Pharmacology of Biological Rhythms, Mannheim, Springer, Verlag;
1997, Page no. 251-283.
14. Clench J, Reinberg A, Dziewanowska Z, Ghata, Smolensky M., “Circadian changes in the
bioavailability and effects of indomethacin in healthy subjects”, Eur J Clin Pharmacol
1981, Page no. 359-369.









