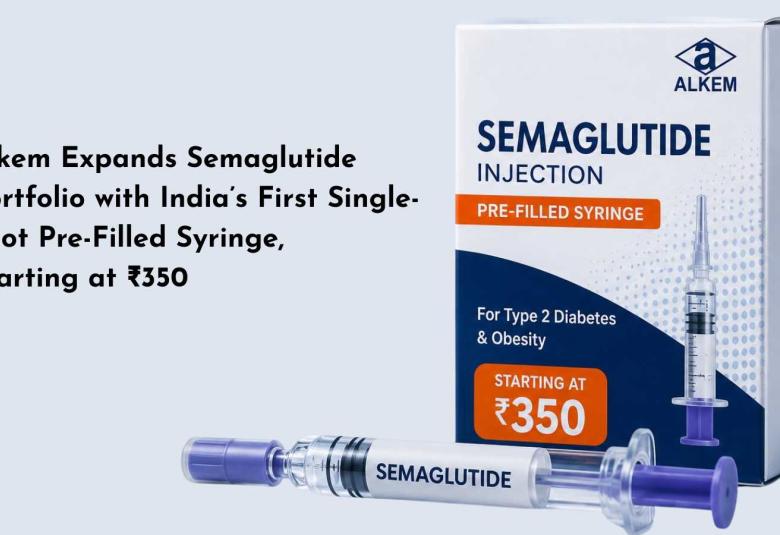
Alkem Expands Semaglutide Portfolio with India’s First Single-Shot Pre-Filled Syringe, Starting at ₹350
In a move aimed at improving access to diabetes and obesity treatment, Alkem Laboratories has launched India's first semaglutide single-shot pre-filled syringes, with prices starting at just ₹350.
The launch marks another milestone in the rapidly evolving obesity and diabetes care market, as Alkem continues to expand its semaglutide portfolio following the introduction of pre-filled disposable and reusable injection pens earlier this year.