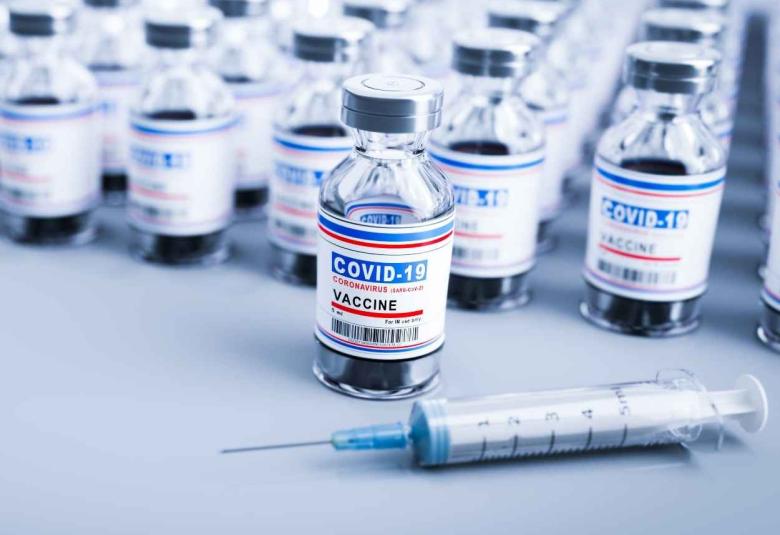
Pfizer and Moderna receives FDA approval for Omicron targeting booster vaccine
Pfizer Inc. and BioNTech announced that the U.S. Food and Drug Administration (FDA) granted Emergency Use Authorization (EUA) of a 30-µg booster dose of the Pfizer-BioNTech COVID-19 Vaccine, Bivalent for individuals ages 12 years and older.
And on the same day, Moderna, Inc. has also received emergency use authorization (EUA) from the U.S. Food and Drug Administration (FDA) for its Omicron-targeting bivalent COVID-19 booster vaccine.