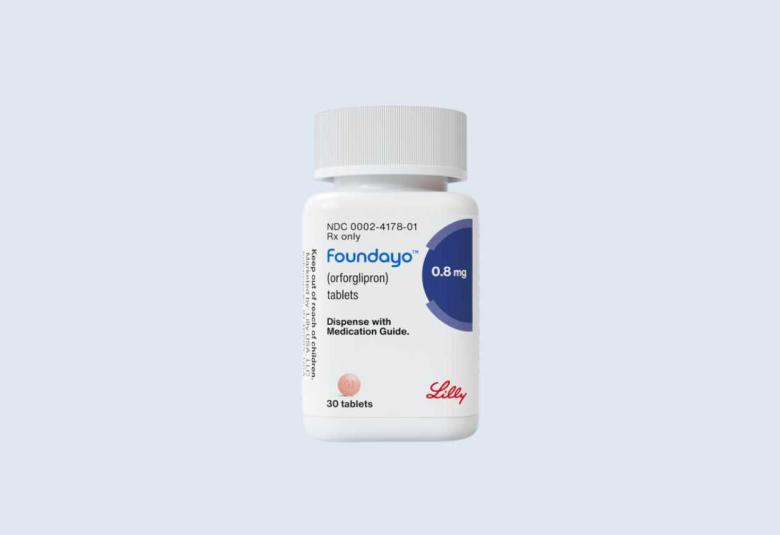
Lilly's novel weight loss medicine gets FDA approval
Eli Lilly and Company announced the U.S. Food and Drug Administration (FDA) approved Foundayo™ (orforglipron) for adults with obesity, or overweight with weight-related medical problems.
When used alongside a reduced-calorie diet and increased physical activity, Foundayo helps individuals lose excess body weight and keep the weight off. Foundayo will be available via LillyDirect®, with prescriptions accepted immediately and shipping beginning April 6, followed shortly after by broad availability through U.S. retail pharmacies and telehealth providers.