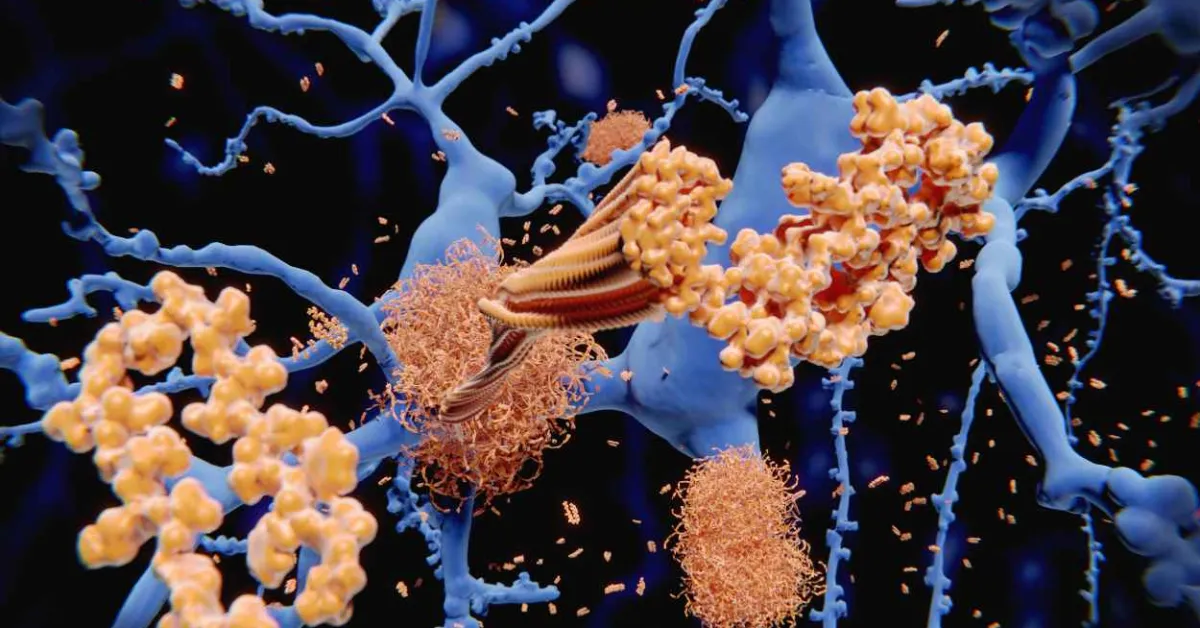
Many neurodegenerative diseases, including Alzheimer’s and Parkinson’s, are difficult to diagnose before symptoms begin to appear. However, disease-related biomarkers such as aggregated proteins called amyloids could provide important insight much earlier, if they can be readily detected. Researchers publishing in ACS Sensors have developed one such method using an array of sensor molecules that can light up amyloids. The tool could help monitor disease progression or distinguish between different amyloid-related conditions.
Neurodegenerative diseases typically involve a breakdown of communication within the brain that is often caused by “sticky” clumps of misfolded proteins called amyloids that interrupt signal transfer. These amyloids are thought to be closely related to the progression of Alzheimer’s disease, so they could be used as a means of early diagnosis to expand treatment options. Currently, radioimaging techniques including positron emission tomography (PET) scans can detect amyloids, but these methods rely on sophisticated equipment and typically focus on one of several amyloids involved in the disease. Instead, fluorescence imaging techniques have been explored as a simpler, yet still sensitive, means of detecting multiple specific amyloids. So, Margaret Sunde, Elizabeth New, Amandeep Kaur and colleagues wanted to develop a fluorescent sensor array for amyloids to monitor Alzheimer’s and other disease progression and to distinguish these atypical amyloids from similar, naturally occurring amyloid-forming proteins.
The team combined five coumarin-based molecular probes, each of which fluoresced to a different degree when it encountered the amyloids, into a sensor array. However, the team found that using just two of the probes with the strongest fluorescence responses still offered a high level of sensitivity and an identifiable fluorescent “fingerprint” for the individual amyloids.
The two-probe array was added to a sample mixture mimicking biological fluids containing molecules that could potentially interfere with sensing. Regardless, the array maintained a high sensitivity and selectivity. Its performance was also tested on samples taken from the brains of mouse models of Alzheimer’s. The team observed that the fluorescence patterns differed between early (at age 6 months) and later (at age 12 months) stages of the disease. Additionally, a unique fluorescence fingerprint was generated for three amyloids typically involved in Alzheimer’s, another disease-associated amyloid and five naturally occurring “functional amyloids” not involved in the disease.
The researchers say that this tool could be used to distinguish between closely related amyloids and could inform new approaches for earlier and more confident diagnosis of amyloid-related diseases.