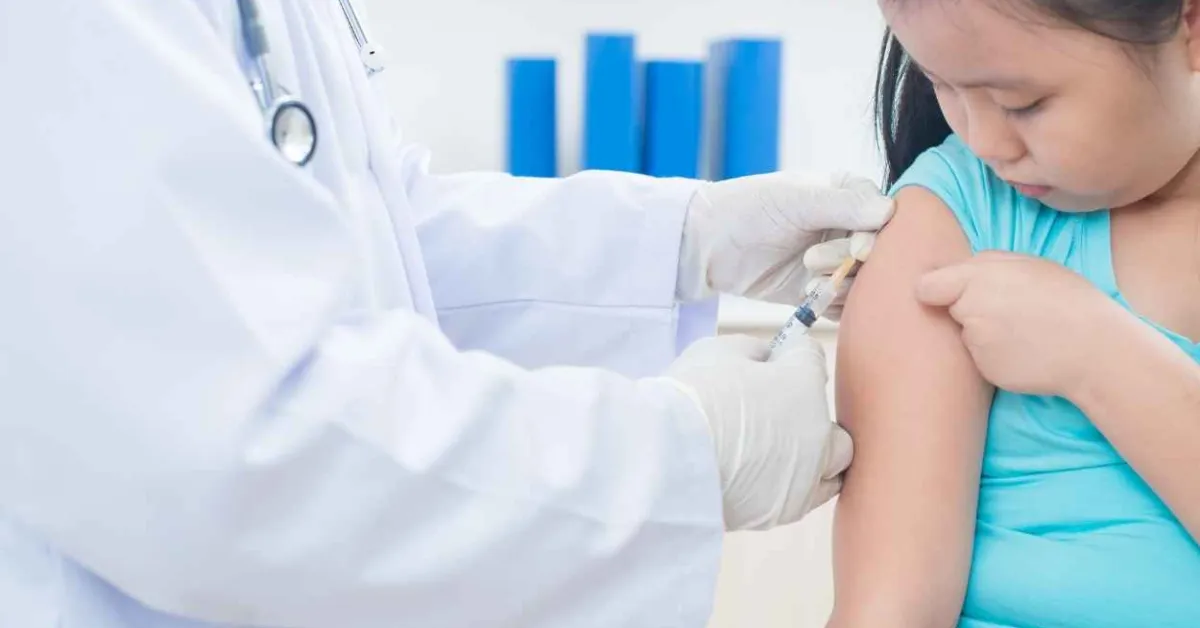
Pfizer Inc and BioNTech SE announced that the U.S. Food and Drug Administration has authorized for emergency use the Pfizer-BioNTech COVID-19 Vaccine for children 5 through 11 years of age (also referred to as 5 to <12 years). For this age group, the vaccine is to be administered in a two-dose regimen of 10-µg doses given 21 days apart. The 10-µg dose level was carefully selected based on safety, tolerability and immunogenicity data. This is the first COVID-19 vaccine authorized in the U.S. for individuals 5 through 11 years of age.
“This is a day so many parents, eager to protect their young children from this virus, have been waiting for,” said Albert Bourla, Chairman and Chief Executive Officer, Pfizer. “Over 6 million children in the U.S. have been diagnosed with COVID-19 since the start of this pandemic, and a high number of young people continue to be infected every week. With this FDA authorization, we have achieved another key marker in our ongoing effort to help protect families and communities, and to get this disease under control.”
“Today’s emergency use authorization is supported by clinical data showing a favorable safety profile and high vaccine efficacy in children, underlining its potential to address a current public health need,” said Ugur Sahin, M.D., CEO and Co-founder of BioNTech. “As children 5 through 11 get reacclimated to the new school year, both in and out of the classroom, our goal is to help keep them safe and protected and get them back to normalcy.”
The FDA based its decision on data from a Phase 2/3 randomized, controlled trial that included ~4,500 children 5 through 11 years of age (2,268 from the original group and 2,379 from the supplemental safety group). Results from this trial were reviewed by the FDA Vaccines and Related Biological Products Advisory Committee (VRBPAC). In the trial, the vaccine demonstrated a favorable safety profile, robust immune responses and a vaccine efficacy rate of 90.7% in participants without prior SARS-CoV-2 infection, measured from 7 days after the second dose. The Data Monitoring Committee for the study has reviewed the data and has not identified any serious safety concerns related to the vaccine.
The companies will begin shipping 10-µg pediatric doses immediately, as directed by the U.S. government (ages referred to as 5y to <12y on the vial and 5 to <12 years on the carton). Eligible U.S. residents will continue to receive the vaccine for free, consistent with the U.S. government’s commitment to free access to COVID-19 vaccines.
As a next step, the U.S. Centers for Disease Control and Prevention’s (CDC) Advisory Committee on Immunization Practices (ACIP) will meet next week to discuss a potential recommendation for the use and rollout of the vaccine to children 5 through 11 years of age. Pediatric vaccinations are anticipated to start, subject to, and after, CDC endorses the ACIP recommendation.
Pfizer and BioNTech have submitted requests for authorization of their COVID-19 vaccine in this age group to other regulators around the world, including the European Medicines Agency. Initial data from the other two age cohorts in the ongoing Pfizer-BioNTech clinical trial in children – those 2 to <5 years of age and those 6 months to <2 years of age – are expected as soon as fourth quarter 2021 or early first quarter 2022.
The Pfizer-BioNTech COVID-19 Vaccine, which is based on BioNTech’s proprietary mRNA technology, was developed by both BioNTech and Pfizer. BioNTech is the Marketing Authorization Holder in the United States, the European Union, the United Kingdom, Canada and the holder of emergency use authorizations or equivalents in the United States (jointly with Pfizer) and other countries. Submissions to pursue regulatory approvals in those countries where emergency use authorizations or equivalent were initially granted are planned or ongoing.