{ DOWNLOAD AS PDF }
 ABOUT AUHTORS
ABOUT AUHTORS
AKASH S MALI1,2*,AUDRIUS MARUŠKA2
1University of Würzburg Germany.
2Vytautas Magnus University, Lithuania
*Akashmit97@gmail.com
ABSTRACT
Pharmaceutical gases molecules (Therapeutic medical Gases) are plays vital role in various disease conditions. In particular, Carbon monoxide, Oxygen, Nitric oxide, Hydrogen sulfide are generated in the human body and mediate signaling pathways as biological messengers and shown pharmacological effects on cardiovascular disorders, nervous system, respiratory track and gastrointestinal disorder. Appropriate oral delivery of gases is challenging concept in front of novel drug delivery system, recently microbubbles, nanobubbles, liposomes, ultrasound technology provides new oral and parenteral delivery platform for therapeutic gases. This review provides pharmacological effects of therapeutic medical gases and its novel delivery aspects.
[adsense:336x280:8701650588]
REFERENCE ID: PHARMATUTOR-ART-2459
|
PharmaTutor (ISSN: 2347 - 7881) Volume 5, Issue 1 Received On: 25/08/2016; Accepted On: 17/09/2016; Published On: 01/01/2017 How to cite this article: Mali AS, Maruška A; Therapeutic Gases Pharmacology and It’s Advanced Delivery; PharmaTutor; 2017; 5(1); 37-41 |
INTRODUCTION
“Therapeutic medical gas is pharmaceutical gaseous molecules which offer solutions to medical needs.” Recent basic and clinical research has revealed that therapeutic gases is an important physiological regulatory factor with antioxidant, anti-inflammatory and anti-apoptotic protective effects on cells and organs. A therapeutic gas has been applied by different delivery methods including straight forward inhalation, drinking hydrogen dissolved in water and injection with hydrogen-saturated saline. The gases are relatively small molecules as compare with classic drugs which give benefits towards permeability of blood brain barrier (BBB). The purpose of this review provide a comprehensive overview of the use of microbubbles and echogenic liposomes for the delivery of bioactive gasses.
Pharmacology of Gases
Oxygen (O2)
The development and growth of multicellular organisms oxygen is play essential role,it administrated to prevent hypoxic injury, the ability to sense and respond to changes in oxygen is important for the survival of prokaryotic and eukaryotic organisms. According to report 60% of solid tumor contain hypoxic tissue and its associated with radiation therapy, adverse effects of classic drugs. Resulting, Tumor hypoxia leads to poor prognosis in cancer’s patients, which is one of the deadly human cancer. Hypoxia-inducible responses are potentially regulated in normal embryonic development and are dysregulated in a tumor hypoxia. In case of respiratory failure (ARDS) require higher amount of FiO2 (fraction of inspired oxygen) with mechanical ventilation to maintain constant level of oxygenation. Oxygen rapidly covert carboxyhemoglobin to oxyhemoglobin to avoid CO2 poisoning, it dissolves in plasma thereby causing relief hypoxia to tissue. In case of anemia. There is decrease in level of O2 supply and there is a need to restore oxygen delivery to tissues For this purpose, significant efforts have been made toward developing artificial blood substitutes. These are typically perfluorocarbon emulsions or hemoglobin based oxygen carriers. Oxygen therapy is of value in cases of cerebrovascular accidents where good oxygenation of hypoxic areas of brain. O2 inhalation is also beneficial in thermotoxicosis and hyperthermia.[1]
Nitric oxide (NO)
It is colorless, poisonous gas which causes serious air pollutant. Biologically NO produced from L-arginine by iNOS and endothelial NOS. It is an important signaling molecule in the body of mammals. NO plays vital role in the vascular homeostasis by its potent vasoregulatory and immunodulatory characteristics. NO stimulate sGC(soluble guanylate cyclase) and increase cGMP content in smooth muscle cells propagating relaxation of vascular tone and vasodilatation and it is also responsible for blood vessel dilation(regulation of platelet activity), which increase tissue blood supply and diminish the inflammatory response leading protection of tissue from oxidative insults. Inhalation of NO prevent graft injury due to ischemia or reperfusion injury after human lungs and liver transplantation. [2]
Hydrogen Sulfide (H2S)
Hydrogen sulfide (H2S) is a colorless, toxic and flammable gas. Hydrogen sulfide is biologically synthesized from L-cysteine, a product methionine, by CBS and cyctathione-γ-lyase. Methionine adenosyltransferase (MAT) converts methionine to homocystine. CBS catalyses thehomocystine to cystathionine conversion. CSE, subsequently, converts cystathionine to L-cysteine, which is further metabolized to hydrogen sulfide by CBS and CSE. H2S help regulate body temperature and metabolic activity at physiological concentrations. H2S affect on cardiovascular system possibly through modulation of K+-ATP channel opening or as a cellular messenger molecule involved vascular flow regulation. Hydrogen sulphide has the potential to act as an antioxidant inhibitor of peroxy nitrite mediated processes via activation of N-methyl-D-aspartate receptor which shield cultured neurons from oxidative damage by increasing levels of glutathione, an antioxidant enzyme,H2S can induce upregulation of HO-1, anti-inflammatory and cytoprotective genes .H2S give antioxidant by its effects on cytochrome c oxidase and mitochondrial functions, while its effects on gene expression may be related to actions on the the NFkB and ERK pathways, H2S protect cardiac system from oxidative injury[3]
Methane(CH4)
It was previously assumed that methane is not utilized by humans, so it is excreted either as flatus, or it traverses the intestinal mucosa and is absorbed into the systemic circulation and excreted unchanged through the lungs as per research methane could exert anti-inflammatory effects. methanogens in the colon can use CO2 and H2 to produce methane as a by-product. Methane gas is produced by enteric bacteria in 30–60% of humans. Pulmonary methane excretion ranged from undetectable to 0.66 ml/min, and 20% of total methane produced was excreted via the lungs(basis on LBT). The therapeutic effect of methane may be confirmed in animal models to conform whether the protective effect of methane is species-specific. Inhalation of methane acts more rapidly, may be suitable for defense against acute oxidative stress. However, it may be impractical for daily application for disease prevention due to its flammability and difficulty to transport. Thus, normal saline rich in methane may be prepared as an injection which may be portable, easily administered and safe.
Helium(He)
Helium is a very light, odorless, tasteless, and colorless noble gas with a strong safety profile and multiple applications. Because of unique physical properties (MP,BP, Density) helium used in many purposes. The high thermal conductivity of helium results in lower body temperature when the body is immersed in helium, which could result in decreased metabolism and decreased energy expenditure [5]. In humans, breathing helium for short periods has not been shown to induce hypothermia. Clinical application of helium is in various areas (Figure no.1)

Figure no.1 Clinical Application of Helium[10,11]
Carbon Monoxide(CO)
It is colorless, odorless initially consider as poison, avidly binds to hemoglobin and forms carboxyhemoglobin. CO is endogenously and physiologically generated in mammalian cells via the catabolism of heme in the rate-limiting step by hemeoxygenase systems. application of CO to animals at low concentrations approximating cigarette-smoke exposure caused no apparent lung pathology. Carbon monoxide may prevent the degradation of heme proteins by binding to the heme moiety. The functions treated in UW with CO showed better renal functions and fewer inflammatory events. Ex vivo organ-targeted CO delivery during cold storage prevents CYP breakdown during the ischemia/reperfusion process. [6]
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT editor-in-chief@pharmatutor.org
Subscribe to Pharmatutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE
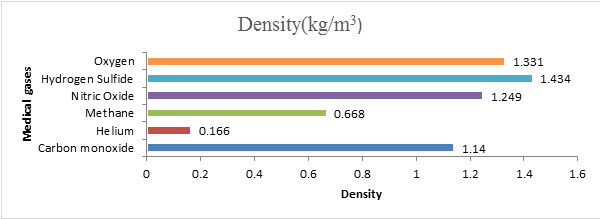
Figure No. 2 Density of Medical Gases
Delivery of Therapeutic Gases
Gases Molecules are comparatively smaller than classic drugs and are able to easily diffuse across membranes and through the blood brain barrier(BBB). The bubble or liposomal shell protects the contained gas from endogenous scavengers, this property particularly attractive for gas delivery which rapidly reacts with HB. indications of gastrointestinal Oral carbon monoxide release system controlled by the permeation of a water through the semipermeable cellulose acetate shell. providing a reliable, easy to use pharmaceutical platform serving various diseases and inflammation.[12] Gases delivery is become advanced due to microbubbles and liposomes, both are differentiating with their gas loading capacity. liposomes can only carry approximately 10% gas by volume. This low loading capacity is sufficient when delivering potent bioactive gasses However, liposomes are less suitable for the delivery of O2, which is typically required in high concentrations for a therapeutic effect. The MBs and ELIPs which recently used for delivery, the difference between both is their size and their potential to extravasate[7]. Tumor vasculature is typically characterized by large pore sizes with upper limits between 380 and 780 nm. This allows for enhanced permeability and retention of large molecules within tumors. Therapeutic microbubbles are typically 1–4 μm in diameter and are therefore unable to extravasate from either healthy or leaky tumor vasculature. The targeting molecule can either be conjugated directly to the surface of the bubble or liposome(Fig.3.A) Antibodies and proteins linked to MB or liposomes through an avidin bridge(Fig.3.B) This would be a limitation when using microbubbles in situations where tissue penetration is required for a therapeutic effect.

Figure No. 3 Targeting method
Liposomes can be fabricated on the nanoscale and have been shown to preferentially extravasate into tumor tissue. loading gas into liposomes increases their size. The ELIPs presented here have average sizes ranging from 800 nm to several microns. These particles may be slightly smaller than microbubbles but still may be too large to extravasate from tumor vasculature. However, it may be possible to reduce the size of these liposomes; liposomes loaded with non-therapeutic gasses have been fabricated with particle sizes between 400 and 600 nm.[8] The microbubble shell provides mechanical stability for the particle and enables persistence of the bubble in circulation. Lipids, which Form a monolayer around the gas core, are a popular choice of shell material. The internal structure of ELIPs is not developed. One theory is that lipid-monolayer stabilized pockets of gas are contained within the liposome core Alternatively, gas may form pockets within the lipid bilayer. Bubbles and liposomes are commonly stabilized by the addition of a hydrophilic polymer brush layer like PEG. PEG stabilizes micro/nanoparticles by introducing steric repulsion between individual particles and between particles and cells. Ultrasound may be used to target the release of a therapeutic gas in a specific area via MB or ELIP destruction at high mechanical index. Acoustic radiation force may be used to push intravenous MBs or ELIPs toward the endothelial surface. This may be used to enhance receptor–target binding efficiency (Fig.3C)
SUMMARY
Medical therapeutic gases having huge clinical applications in cardiovascular, neurobiology and respiratory inflammation. Properties of medical gases to better oxidative stress plays vital roles at the Pharmacological, cellular and physiological levels. Many gases may cause hazards effects, there are still many possible applications of these gases as therapeutic purpose for numerous diseases if the concentrations are tightly controlled. Microbubbles and liposomes offer new edges as Medical gas delivery vehicles. Gases particles are inherently echogenic, and therefore ultrasound can be used to fix them and targeted release of gas contents in via acoustic cavitation. Microbubble and liposomal delivery of medical gasses must be proven to be safe prior to clinical translation.
REFERENCES
1.Buchholz, BM1, et al.; "Hydrogen inhalation ameliorates oxidative stress in transplantation induced intestinal graft injury."; American Journal of Transplantation; (2008); 8(10) 2015-2024.
2. Mocellin, Simone, Vincenzo Bronte, and Donato Nitti;"Nitric oxide, a double edged sword in cancer biology: searching for therapeutic opportunities."; Medicinal research reviews; (2007); 27(03) 317-352.
3.Ikeda, Hideaki, et al;"Fatal and nonfatal poisoning by hydrogen sulfide at an industrial waste site."; Journal of Forensic Science; (2002); 47(3) 652-655.
4. . Liu, Wenwu, et al; "Is methane a new therapeutic gas?."; Medical gas research; (2012); 2.1; 1.
5. Fix, Samantha M., Mark A. Borden, and Paul A. Dayton; "Therapeutic gas delivery via microbubbles and liposomes."; Journal of Controlled Release; (2015); 209; 139-149.
6. Steiger, Christoph, Tessa Lühmann, and Lorenz Meinel.; "Oral drug delivery of therapeutic gases—Carbon monoxide release for gastrointestinal diseases."; Journal of Controlled Release; (2014); 189; 46-53.
7. Huang, Shao-Ling, and Robert C. MacDonald; "Acoustically active liposomes for drug encapsulation and ultrasound-triggered release."; Biochimica et Biophysica Acta (BBA)-Biomembranes; (2004); 1665(1-2) 134-141.
8. Tao, Zhimin, and P. Peter Ghoroghchian;"Microparticle, nanoparticle, and stem cell-based oxygen carriers as advanced blood substitutes."; Trends in biotechnology; (2014); 32(9); 466-473.
9. Rudic, Radu Daniel, et al; "Direct evidence for the importance of endothelium-derived nitric oxide in vascular remodeling."; Journal of Clinical Investigation; (1998); 101(4); 731.
10. Ma, D., et al; "Neuroprotective and neurotoxic properties of the ‘inert’gas, xenon."; British Journal of Anaesthesia; (2002); 89(5); 739-746.
11. Britton, George L., et al; "In vivo therapeutic gas delivery for neuroprotection with echogenic liposomes."; Circulation; (2010); 122(16); 1578-1587.
12. Roccarina, Davide, et al.; "The role of methane in intestinal diseases."; The American journal of gastroenterology; (2010); 105(6); 1250-1256.
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT editor-in-chief@pharmatutor.org
Subscribe to Pharmatutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE









