{ DOWNLOAD AS PDF }
ABOUT AUTHORS:
Pankaj Kushwah1*, D.K. Vir2, Nandu Kayande2, Ravina Patidar2
1Department of Pharmacognosy, Nimar Institute of Pharmacy, Dhar, MP
2Department of Pharmacology
Nimar Institute of Pharmacy, Dhamnod, Opp.ITI, Maheshwar Road Dhamnod, Dhar. M.P.
pankajnipd@gmail.com
ABSTRACT
The present study was undertaken to explore the phytochemical screening & antimicrobial activity of stems of Glycyrrhiza Glabra L. (Leguminaceae). Glycyrrhiza Glabra L. were screened for their antimicrobial activity by agar well diffusion method, standard screening method & disc diffusion method. In the phytochemical screening showed presence of secondary metabolites, flavonides, glycosides & terpenoids. It is also showed potent against almost all the test organism.
Study indicate that Glycyrrhiza Glabra has antibacterial, antioxidant, antimalerial, antispasmodic, anti-inflammatory and anti-hyper glycemic properties. Various other effects like antiulcer, antiviral, antihapatotoxic, antifungal and herpes simplex have also been studies. One of the most commonly reported side effects with licorice supplementation is elevated blood pressure.
REFERENCE ID: PHARMATUTOR-ART-2162
INTRODUCTION
India enjoys the privilege of having time tested traditional systems of medicines based on natural products[1]. Plant based natural products have been in use for medicinal, therapeutic or other purposes right from the dawn of history [2]. Traditional system of ultimately shows us that plants have provided humanity a large variety of patent drugs to alleviate suffering from diseases[3,4]. However, drugs used in traditional systems of medicine are all crude drugs in their natural state or their preparations [1,6]. Until19th century, even western medicine (allopathic) depends largely on crude drugs. During 20thcentury the progress in chemical techniques and with the growth of pharmaceutical industry. Chemical (synthetic) drugs replaced crude drugs gradually. Thus; synthetic drugs can produce remarkable lifesaving result in acute diseases but cannot be used often in treatment of chronic diseases [5,7, 8].
Phytochemical Screening- In the phytochemical screening showed presence of secondary metabolites, flavonoids, saponins, glycosides & terpenoids. It also showed potent against almost all the test organism [9].
Microorganism-A microorganism is a microscopic organism that comprises either a single cell or no cell at all. The study of MO is called as microbiology. It include [10]-
1. Bacteria- They are practically invisible to the naked eye. It can grow double in every 10 mins.
2. Fungi- They have several unicellular species, such as Baker’s yeast.
3. Viruses-They can replicate only inside the cell.
4. Prokaryotes- They are lack of cell nucleus.
Antimicrobial activity- The activity which work against MO is called as antimicrobial activity & those agents which is helps in this activity is called as antimicrobial agents [11].
PLANT DESCRIPTION- Glycyrrhiza Glabra L. [12]
Synonym- Liquorice, Mulethi.
Biological source- Liquorice is consist of dried root of Glycyrrhiza Glabra L
Family- Laguminaceae.
Uses-
1. As expectorant.
2. As antispasmodic.
3. As flavoring agent.
MATERIAL & METHOD
1. Collection & drying of plant [13]
Glycyrrhiza Glabracollected in August, dried, made into coarse powder & extracted by using pettrolium ether or acetone in soxlet apparatus for 72hrs.
2. Extraction of plant [4,14]-
The coarse powder of shade dried stem of G. glabra (1.5 kg) was extracted with ethanol (95%) in a Soxhlet extractor. The ethanolic extract was then concentrated on rotary flash evaporator to 1/10th volume. The concentrated ethanolic extract was then fractionated with n-butanol, ethyl acetate, solvent ether and petroleum ether (40-60ºC) in succession.
An aqueous extract was also prepared by maceration method using chloroform water I.P. as vehicle. The aqueous extract was concentrated to dryness under vacuum using rotary flash evaporator. An attempt was also made to observe the presence and absence of different phytochemical constituents, viz. alkaloids (Dragendorff’s test), anthraquinones, saponins (foam formation), flavonoids (using magnesium and dil. HCl), sesquiterpenes and terpenes (Liebermann–Burchard’s test) according to standard methods.
3. Preliminary phytochemical screening [15,16]
The extracts were subjected to Preliminary phytochemical screening in order to identify the nature of chemical compounds presenting stem.
a.) Detection of alkaloids- Small portion of plant extracts + dil.HCl add, tasted with Mayers reagent, Tannic acid to detect the presence of alkaloids.
b.) Detection of carbohydrate- Small quantities of extracts were subjected to Molish test, Fehlings test, Benedict test, Iodine, Barford test, and Bial test.
c.) Detection of steroids- Small portion of plant extracts were subjected to Salkowaski test, add chloroform & conc. Sulphuric acid, Chloroform layer gives Greenish yellow fluorescence.
d.) Detection of saponins- Small amount of extricates shaken with water to check foam formation and its time of stability.
e.) Detection of tanins- Small quantity of extraction were placed between two filter papers and pressed. Oil stain on filter paper indicates the presence of fats.
4. Preparation of agar plate [17]
a.) Dissolved all chemicals in 1000ml. diss. Water.
b.) Find out the pH of the solution by pH paper.
c.) Adjust pH by NaOH or HCl solution drop by drop to pH 7.0.
d.) Now, put cotton in the mouth of the conical flask & cover it with Al foil.
e.)Then sterilize it in the autoclave.
5. Antimicrobial screening method [18,19]
Agar well diffusion method-antimicrobial activity was tested by Agar well diffusion method. The extracts were freshly reconstituted with suitable solvent & tested at various concentrations. Each ex. was carried out in triplicated and the mean diameter of the inhibitor zone was recorded.
Inhibition zone of the sample
% of Zone of inhibition = ---------------------------------- * 100
Inhibition zone of the standard
RESULTS
1. Percentage yield of extracts-
% yield of different extracts obtain by successive solvent extraction were determined & were found to be, Pet. Ether- 0.5%. Acetone- 2.0%, Ethanol- 3.5% &Aqueous- 3.6%
Table 1- Determination of % yield of extracts-

Table 1.
2. Phytochemical studies-
Phytochemical studies showed the presence of alkaloids, saponins, tannins, carbohydrates, flavonoids & fats.( Table 2)
Table 2- Phytochemical studies-
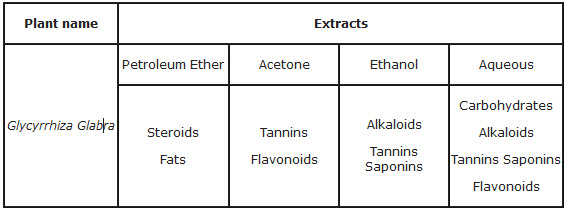
Table 2.
3. Antimicrobial activity-
Antimicrobial activity of various extracts of Glycyrrhiza Glabra was studied by measuring the zone of inhibition formed around the well &result is given at table no. 3.
Table 3-Zone of inhibition in cm
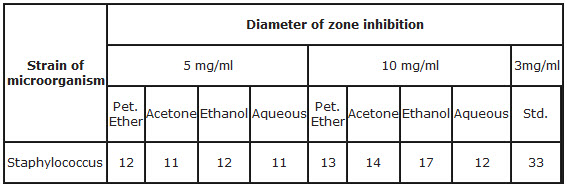
Table 3.
4. Qualitative chemical evaluation
The qualitative chemical examination showed the presence of alkaloid, steroid, fates, flavonoids, tannins, sterols, carbohydrates and saponins.
CONCLUSION
The plant (Glycyrrhiza Glabra Linn.) tested for antimicrobial activity was very good. In present investigation phytochemical screening showed the presence of secondary metabolites, flavonoids, saponins, glycosides & terpenoids. It also showed potent against almost all the test organism.
ACKNOWLEDGEMENT
I express my special regards to Dr. Rajesh Garg Principal Nimar Institute of Pharmacy. It’s because of his priceless intellectual help, constructive idea and for having given me complete independence, affectionate encouragement to the successful completion of this work
REFERENCES
1. ncbi.org.
2.Rambir Singh, Ramesh Chandrqa Mridula Bose and Pratibha Mehta Luthra “Antibactreial activity of Glycyrrhiza Glabra L.extract on pathogenic bacteria”, current science, 83.2002,737.
3.T.V. Sambasivam Pillai, Dictionary of Medicine, Chemistry, Botany & Allied science, 3rd volume, P.No. 212-217.
4.Bukar, A., Uba A. and Oyeyi, T.I. Antimicrobial profile of morinaoleifera lam. Extracts against some food borne microorganisms, Bayero Journal of Pure and Applied Sciences, 3(1), 2010 43-48.
5. cellbiolabs.com.
6.K.M. Elizabeth, Antimicrobial Activity of Tamarindus indica Linn. Indian Journal of Clinical Biochemistry, 2005, 20 (2), 150-153.
7.Vedpriya Arya, Sanjay Yadv, Sandeep Kumar, JP Yadav, Antimicrobial activity of cassia occidentals L (leaf) against various Human Pathogenic microbes, Life Sciences and Medicine Research. 2010, 1-6.
8. ncbi.com.
9.Anonymous 2009 mmbr.asm.org/cgi/pmidlookup?view=long&pmid=3083222.
10.R.C. Dubey & D.K. Maheshwari, A textbook of Microbiology, 3rd edition P.No.49-50.
11.S.U. Kummmaru, T. and Santosh H. Morphological diversity and its distribution. P.No. 441-416.
12.C.K. Kokate, S.K. Purohit, Gohhale D.K. Text book of Pharmacognosy, 3rd ed., Published by Nirali Prakashan Pvt. Ltd. P.No.181-183.
13.Harbone J.B., Phytochemical methods, 3rd ed., 1990, Published by Hall Londen, P.No. 123-131.
14.Segal R, Pisanty S, Wormser R, Azaz E and Sela MN, Anticariogenic activity of licorice and Glycyrrhizine I: Inhibition of in vitro plaque formation by Streptococcus mutants. Journal of Pharmaceutical Sciences. 1985; 74: 79-81.
15.Ravikumar Patil H.s, Makari, H.K. Gurumurthy H., In Vitro antimicrobial activity of Ethanol Extract of Glycyrrhiza Glabra L. EJEAF Che, 6 (9), 2007, 2318-2322.
16. whfoods.com.
17.Baba M, Shigeta S. Antiviral activity of glycyrrhizin against Varicella zoster virus in vitro .Antiviral Res 1987, 107.
18.Fenwick, G., Lutomski, J. and Nieman, C. (1990): Glycyrrhiza Glabra L. (Liquorice): Composition uses and analysis. Food Chemistry., 38(2): 119-143.
19.Nakagawa K and Asami M, Effect of glycyrrhizin on hepatic lysosomal system. Japanese Journal of Pharmacology, 1981; 31: 849–851.
|
PharmaTutor (ISSN: 2347 - 7881) Volume 2, Issue 5 Received On: 18/03/2014; Accepted On: 27/03/2014; Published On: 01/05/2014 How to cite this article: P Kushwah, DK Vir, N Kayande, R Patidar; Phytochemical Screening & Evaluation of Antimicrobial Activity of Glycyrrhiza Glabra Linn; PharmaTutor; 2014; 2(5); 144-147 |
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT articles@pharmatutor.org
FIND OUT MORE ARTICLES AT OUR DATABASE









