About Authors: Rakesh Verma
Seth G.L. Bihani S.D. college ,
sri ganganagar
1. Introduction
The discovery of a variety of pharmaceuticals in surface, ground, and drinking waters around the country is raising concerns about the potentially adverse environmental consequences of these contaminants. Minute concentrations of chemicals known as endocrine disruptors, some of which are pharmaceuticals, are having detrimental effects on aquatic species and possibly on human health and development.
The consistent increase in the use of potent pharmaceuticals, driven by both drug development and our aging population, is creating a corresponding increase in the amount of pharmaceutical waste generated. Pharmaceutical waste is not one single waste stream, but many distinct waste streams that reflect the complexity and diversity of the chemicals that comprise pharmaceuticals. Pharmaceutical waste is potentially generated through a wide variety of activities in a health care facility, including but not limited to intravenous (IV) preparation, general compounding, spills/breakage, partially used vials, syringes, and IVs, discontinued, unused preparations, unused unit dose repacks, patients’ personal medications and outdated pharmaceuticals.
In hospitals, pharmaceutical waste is generally discarded down the drain or land filled, except chemotherapy agents, which are often sent to a regulated medical waste incinerator. These practices were developed at a time when knowledge was not available about the potential adverse effects of introducing waste pharmaceuticals into the environment. Proper pharmaceutical waste management is a highly complex new frontier in environmental management for health care facilities. A hospital pharmacy generally stocks between 2,000 and 4,000 different items, each of which must be evaluated against state and federal hazardous waste regulations. Pharmacists and nurses generally do not receive training on hazardous waste management during their academic studies, and safety and environmental services managers may not be familiar with the active ingredients and formulations of pharmaceutical products.
Frequently used pharmaceuticals, such as epinephrine, warfarin, and nine chemotherapeutic agents, are regulated as hazardous waste under the Resource Conservation and Recovery Act (RCRA). Failure to comply with hazardous waste regulations by improperly managing and disposing of such waste can result in potentially serious violations and large penalties.

2. Overview of Biomedical waste
Biomedical waste is broadly defined as any solid or liquid waste that is generated in the diagnosis, treatment of immunization of human beings or animals in research pertaining thereto, or in the production or testing of biological material. According to World Health Organization (WHO) estimates 85% of hospital waste is actually non-hazardous and around 10% is infectious while the remaining 5% is non-infectious but consists of hazardous chemicals like methyl chloride and formaldehyde. Here, the main concern of infectious hospital waste is the transmission of HIV and Hepatitis B or C viruses. In this context, Syringes and needles have the highest disease transmission potential.
Hospital waste, till recently was not being managed but it was simply ‘disposed off’. The disposal of hospital waste can be very hazardous particularly when it gets mixed with municipal solid waste and is dumped in uncontrolled or illegal landfills such as vacant lots in neighboring residential areas and slums. This can lead to a higher degree of environmental pollution, apart from posing serious public health risks such as AIDS, Hepatitis, plague, cholera, etc.
In the total amount of municipal waste a city generates, only 1 to 1.5% is hospital waste, of which 10-15% is considered infectious. It is estimated, a city like New Delhi with about 40,000 beds generates about 60 metric tons of hospital waste per day. But whatever the amount of hospital waste there be, it proves to be harmful to the community. This needs immediate treatment and effective disposal. Discarded blood and blood products serve as significant foci of hazardous diseases. The waste stream from X-ray units has chemical contamination of silver bromide (Fixon), glutaraldehyde, hydroquinone and potassium hydroxide. The waste stream from the sterilization of syringes usually has infectious materials and methanol. The usual infective waste consists of bandages, gauzes, cotton waste, amputated human parts, placenta and used dialysis kits containing plastic and aluminum. The sterilization of dialysis units, operation theatres and private wards contributes formaldehyde. The waste from laboratory contains infectious materials as well as reagents and solvents used for analytical purposes.Further, unhygienic conditions in general ward toilets, coupled with frequent strikes by Class IV staff create what are virtually secondary foci of infectious diseases within the hospital premises. (Bekir Onursal, 2003)
Table 1: Percentage Constitution of Hospital Waste
|
CONSTITUENTS |
APPROX. % |
|
Pathological waste |
5 |
|
Infectious material |
10 |
|
General Non-Infectious |
50 |
|
Kitchen waste |
30 |
|
Recyclable materials (paper, plastic, Metal) |
45 |
Such areas are often stockpiles of heterogeneous infectious material and contribute greatly to the incidence of nosocomial infections. For example, according to a WHO report the excreted loads of some selected enteric diseases are as follows. The persistence of these organisms in the environment at 20-30ºC is shown to vary from 2 weeks to a month. This aggravates health hazards when associated with the biomedical wastes generated in the hospitals.
2.1. Sources of Biomedical Waste
Sources of BMWs in health care facilities include wards, delivery rooms, operating theaters, emergency and out- patient services, laboratories, and pharmaceutical and chemical stores. Persons at risk of exposure include health care facility employees (doctors, nurses, health care assistants, maintenance personnel, and support personnel for waste handling, transportation, and laundry), patients and their visitors, and waste management facility employees and scavengers.
Infectious wastes containing potentially harmful micro-organisms can infect hospital patients, health care employees, and patients’ visitors. Used needles, syringes, and other sharps present risks of injury and infection (for example, hepatitis B and C, and HIV) for health care employees. Chemical and pharmaceutical wastes may cause intoxication or injuries such as burns. Genotoxicwastes are hazardous and may have mutagenic, teratogenic, or carcinogenic properties. Radioactive sources may cause severe injuries to humans such as destruction of tissue. (Bekir Onursal, 2003)
Table 2: Excreted Loads of Infectious Agents (Dr. Saurabh Sikka)
|
INFECTIOUS AGENT |
AVERAGE NO. PER GRAM FECES |
|
Shigella spp. |
107 |
|
Salmonella typhi |
108 |
|
Escherichia coli(pathological) |
108 |
|
Hepatitis A virus |
106 |
Table 3. Hospital Waste Related Nosocomial Infections (Dr. Saurabh Sikka)
|
ORGANISM |
DISEASE CAUSED |
RELATED WASTE ITEM |
|
VIRUSES · HIV · Hepatitis B · Hepatitis A,C · Arboviruses · Enteroviruses
|
· AIDS · Infectious Hepatitis · Infectious Hepatitis · Dengue, Japanese encephalitis, tick-borne fevers, etc. · Dysentery
|
· Infected needles, body fluids · Infected needles, body fluids · Human excreta, soiled Linen · Blood, body fluids · Human excreta, soiled linen |
|
BACTERIA · Shigella spp. · Salmonella typhi · Vibrio cholerae · Clostridium tetani · Staphylococcusspp. · Pseudomonas · Streptococcus · Borrelia spp. |
· Shigellosis · Typhoid · Cholera · Tetanus · Wound infections, septicemia, rheumatic fever, endocarditis, skin and soft tissue Infections. · Louse and tick borne fevers. |
· Human excreta and body fluid in landfills and hospital wards · Sharps such as needles, surgical blades in hospital waste. · Rodent infestations of poorly managed landfills and dumping grounds.
|
|
PARASITES · Giardia lamblia · Wucheraria bancrofti · Plasmodium |
Giardiasis Cutaneous leishmaniasis, Kala Azar Malaria |
Human excreta, blood and body fluids in poorly managed sewage system of Hospitals. |
2.2. Classification of Bio-Medical Waste (Dr. Saurabh Sikka)
The World Health Organization (WHO) has classified medical waste into eight categories:
1. General Waste
2. Pathological waste
3. Radioactive
4. Chemical
5. Infectious to potentially infectious waste
6. Sharps
7. Pharmaceuticals
8. Pressurized containers
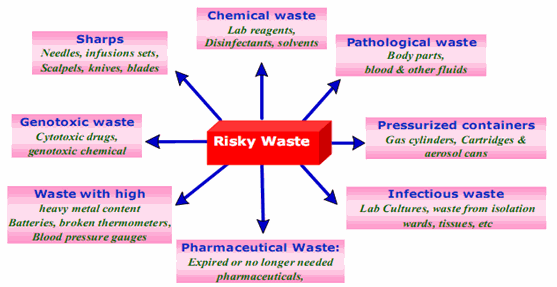
Figure.3 Different Types of Risky Waste (C. Visvanathan,2006)
3. Pharmaceutical Waste
Pharmaceutical waste is potentially generated thorough a wide variety of activities is a health care facility , including but syringes, and not limited to intravenous (IV) preparation , general compounding , spills/breakage, partially used vials, syringes, and IVs, discontinued , unused preparations, unused unit dose repacks, patients personal medications and outdated pharmaceuticals. Health care industries can generate hazardous waste from many sources, including disposal of pharmaceuticals. Pharmaceutical waste may also include expired drugs, patients personal medications, waste materials containing excess drugs (syringes, IV bags, tubing, vials, etc.)
Table 4: Overview of the Types of Health Care Wastes (Bekir Onursal, 2003)
|
Types of Health Care Wastes |
Examples |
|
Communal waste (solid wastes that are not infectious, chemical, or radioactive) |
Cardboard boxes, paper, food waste, plastic and glass bottles |
|
Biomedical wastes Infectious waste (wastes suspected of containing pathogens) |
Cultures, tissues, dressings, swabs, and other blood-soaked items; waste from isolation wards |
|
Anatomical waste |
Recognizable body parts |
|
Sharps |
Needles, scalpels, knives, blades, broken glass |
|
Pharmaceutical waste |
Expired or no longer needed medicines or pharmaceuticals |
|
Genotoxic waste |
Wastes containing genotoxic drugs and chemicals (used in cancer therapy) |
|
Chemical waste |
Laboratory reagents, film developer, solvents, expired or no longer needed disinfectants, and organic chemical wastes (for example, formaldehyde, phenol-based cleaning solutions) |
|
Pressurized containers |
Aerosol cans, gas cylinders (that is, anesthetic gases such as nitrous oxide, halothane, enflurane, and ethylene oxide; oxygen, compressed air) |
|
Radioactive waste |
Unused liquids from radiotherapy; waste materials from patients treated or tested with unsealed radionuclide’ |
3.1Defining Pharmaceutical Hazardous Waste Categories
Hazardous wastes are divided into two categories: (1) listed wastes, and (2) characteristic wastes. Listed wastes appear on one of four lists of hazardous waste (F, K, P and U).Pharmaceuticals are found on two of these lists, the P and U lists which both contain commercial chemical products. Characteristic wastes are regulated because they exhibit certain hazardous properties – ignitability, corrosivity, reactivity and toxicity.
3.1.1 P- Listed Wastes
Pharmaceuticals are chemicals first and therapeutic agents second. P-listed wastes are commercial chemical products that are categorized as acutely hazardous under RCRA. One of the primary criteria for including a drug on the P-list as acutely hazardous is an oral lethal dose of 50 mg/kg (LD50) or less. LD50 is the amount of a material, given all at once, which causes the death of 50% of a group of test animals. Eight chemicals on the P-list are used as pharmaceuticals
Table 5: P-listed Pharmaceuticals (Eydie Pines, 2006)
|
Constituent of Concern |
Waste Code |
|
Arsenic trioxide |
P012 |
|
Phentermine(CIV) |
P046 |
|
Epinephrine |
P042 |
|
Physostigmine |
P204 |
|
Nicotine |
P075 |
|
Physostigmine salicylate |
P188 |
|
Nitroglycerin |
P081 |
|
Warfarin>0.3% |
P001 |
Identifying Waste Pharmaceuticals
Some drugs have more than one trade name. The underlying chemical name, not the trade name, is regulated under RCRA. To be sure you do not miss a chemical due to using a trade name or generic name, use the Chemical Abstracts Service registry numbers that can be obtained from the Merck Index or other chemical references and compare them to the CAS numbers in the Code of Federal Regulations.
Ø Phentermine is a good example of the use of the CAS number, since it is listed in the 40 CFR 261.33 only as Benzeneethanamine, alpha, alpha-dimethyl-. By looking up phentermine in the Merck Index, its CAS number of 122-09-8 would tie to the chemical name in 40 CFR 261.33(e) to P046.
Ø Trisenox® is the trade name for arsenic trioxide which is regulated as P012.
3.1.2 U-Listed Wastes
Two Necessary Conditions
There are some drugs on the U-list. These chemicals are listed primarily for their toxicity. Similar to a P-listed waste, when a drug waste containing one of these chemicals is discarded, it must be managed as hazardous waste if two conditions are satisfied:
(1) The discarded drug waste contains a sole active ingredient that appears on the U list, and
(2) It has not been used for its intended purpose.
Table 6: U-listed Pharmaceuticals
|
Constituent of Concern |
Waste Code |
Constituent of Concern |
Waste Code |
|
Chloral hydrate (CIV) |
U034 |
Paraldehyde (CIV) |
U182 |
|
Chlorambucil |
U035 |
Phenol |
U188 |
|
Cyclophosphamide |
U058 |
Reserpine |
U200 |
|
Daunomycin |
U059 |
Resorcinol |
U201 |
|
Dichlorodifluoromethane |
U075 |
Saccharin |
U202 |
|
Diethylstilbestrol |
U089 |
Selenium sulfide |
U205 |
|
Lindane |
U129 |
Trichloromonofluromethane |
U121 |
|
Mitomycin C |
U010 |
Warfarin <0.3% |
U248 |
|
Mercury |
U151 |
Uracil mustard |
U237 |
|
Melphalan
|
U150 |
Streptozotocin |
U206 |
3.1.3 D-Listed Wastes
Table 7: D-listed Chemicals Used in Drug Formulations
|
Ingredient
|
Waste Code |
Regulatory Level (mg/l) |
Drugs Formulations Containing These Ingredients |
|
Arsenic |
D004 |
5.0 |
Arsenic trioxide (also P-listed) |
|
Barium |
D005 |
100.0 |
Barium sulfate (used in radiology) |
|
Cadmium |
D006 |
1.0 |
Multiple mineral preparations |
|
Chloroform |
D022 |
6.0 |
No longer commonly used |
|
Chromium |
D007 |
5.0 |
Multiple mineral preparations |
|
Mercury |
D009 |
0.2 |
Vaccines with thimerosal |
|
Silver |
D011 |
5.0 |
Silver sulfadiazine cream |
3.2 Characteristic Hazardous Waste
(1) Ignitability,
(2) Corrosivity,
(3) Reactivity, and
(4) Toxicity.
3.2.1 Ignitability:
The objective of the ignitability characteristic is to identify wastes that either present a fire hazard under routine storage, disposal, and transportation or are capable of exacerbating a fire once it has started. There are several ways that a drug formulation can exhibit the ignitability characteristic
Table 8. Types of Ignitability with Resources.
|
Ignitable Properties |
Resources |
Ignitable Drug Formulations |
|
Aqueous drug formulation containing 24 % or more alcohol by volume and having a flashpoint of less than 140 º F or 60 º C (261.21(a)(1)) |
Ø Material Safety Data Sheet Ø Common pharmacy references such as Facts and Comparisons or their on-line database, E-Facts |
Ø Erythromycin Gel 2% Ø Texacort Solution 1% Ø Taxol Injection |
|
Liquid drug formulations, other than aqueous solutions containing less than 24 % alcohol, with a flashpoint of less than 140 º F or 60 º C |
Ø MSDS Ø Standard laboratory test procedure for measuring flashpoint |
Ø Flexible collodion used as a base in wart removers is not an aqueous solution and has a flashpoint = 45 degrees C
|
|
Oxidizers or materials that readily supply oxygen to a reaction in the absence of air as defined by the DOT
|
Ø 40 CFR 264 Appendix V Examples of Potentially Incompatible Waste Group 6-A Oxidizers Ø Test methods in 49 CFR 173.151
|
Ø Amyl nitrite inhalers, used for the rapid relief of angina pain Ø Silver nitrate applicators, used for cauterizing Ø Bulk chemicals found in the compounding section of the pharmacy such as potassium permanganate |
|
Flammable aerosol propellants meeting the DOT definition of compressed gas (261.21(a)(3)) |
Ø Test methods in 49 CFR 173.300
|
Ø Primatene aerosol
|
3.2.2 Corrosivity:
Any waste which has a pH of less than or equal to 2 (highly acidic) or greater than or equal to12.5 (highly basic) exhibits the characteristic of corrosivity and must be managed as a hazardous waste. Generation of corrosive pharmaceutical wastes is generally limited to compounding chemicals in the pharmacy. Compounding chemicals include strong acids, such as glacial acetic acid and strong bases, such as sodium hydroxide.
3.2.3 Reactivity:
Reactive wastes are unstable under "normal" conditions. They can cause explosions, toxic fumes, gases, or vapors when heated, compressed, or mixed with water. Nitroglycerin is the only drug that is potentially reactive. Refer to the section above, entitled Nitroglycerin Exclusion, for an understanding of the regulatory status of medicinal nitroglycerin.
3.2.4 Toxicity:
Wastes that exceed these concentrations must be managed as hazardous waste. The test that determines the ability of these chemicals and heavy metals to leach in a landfill environment is called the Toxicity Characteristic Leaching Procedure, or TCLP. If the concentration determined by the TCLP exceeds the stated limits, the waste must be managed as hazardous waste. (Eydie Pines, 2006)
4. Pharmaceutical waste Management
4.1 Pharmaceutical waste management Scope
There are four (4) categories of pharmaceutical waste that need to be managed and they are defined as follows:
4.1.1 RCRA Hazardous Waste:
Waste Pharmaceuticals that meet the definition of a hazardous waste and must be segregated and managed as hazardous waste. These include nine antineoplastic agents. Determine if any stocked pharmaceuticals meet the definitions of hazardous waste by applying the criteria noted below:
1) The generic or chemical name is listed in 40 CFR 261.33 (e) (P list) or (f) (U list) or comparable state regulations
i. Empty containers which have held pharmaceuticals listed in 40 CFR 261.33 (e), or P-listed wastes, must be managed as hazardous waste except for used epinephrine syringes.
ii. Common P-listed pharmaceuticals used in the military treatment facility setting are:
2) The formulation exhibits a characteristic of hazardous waste as defined by the following:
i. 40 CFR 261.21 Characteristic of ignitability
ii. 40 CFR 261.22 Characteristic of corrosivity
iii. 40 CFR 261.23 Characteristic of reactivity
iv. 40 CFR 261.24 Toxicity characteristic
3) The formulation exhibits a characteristic of hazardous waste as defined by state hazardous waste regulations
4.1.2Non-RCRA Antineoplastic Hazardous Waste:
The Navy Bureau of Medicine and Surgery (BUMED) Environmental Programs (EP) Directorate has made the decision to manage all antineoplastic agents as hazardous waste regardless of whether or not they are technically listed as RCRA antineoplastic hazardous waste due to their inherent toxicity. Non-RCRA antineoplastic hazardous waste includes all antineoplastic agents used for the treatment of cancer that are not regulated by RCRA.
4.1.3BMP Hazardous Waste:
Pharmaceuticals which meet the criteria in these guidelines should be evaluated for possible management as RCRA hazardous waste as a best management practice. The decision will be made at the facility level. All drugs that do not meet the criteria stated above are categorized as BMP Non-Hazardous Pharmaceuticals. BMP Non-Hazardous Pharmaceuticals that meet any of the criteria listed below must be incinerated. If a regulated medical waste incinerator is not available, they should be disposed of as hazardous waste.
1) Combo P-,U-: Formulations containing more than one P or U-listed drug or combinations of P or U-listed drugs with other active ingredients.
2) OSHA: Drugs listed in the Occupational Safety and Health Administration (OSHA) Technical Manual Section 6, Chapter 2, Appendix VI: 2 -1. This list is included as
3) CARCIN: Drugs listed in the US Department of Health and Human Services National Toxicology Program's Report on Carcinogens (11th Edition).
4) OSHA or NIOSH: Additional drugs meeting OSHA or NIOSH criteria.
5) LD50: Drugs with LD50s at or below 50mg/kg.
6) EDC: Endocrine disrupting compounds not listed in any of the above references.
7) VITAMIN: Vitamin/mineral preparations that may fail the toxicity characteristic due to chromium, selenium, or cadmium for which there is inadequate data to make a hazardous waste determination.
4.1.4BMP Non-Hazardous Pharmaceutical Waste:
All other pharmaceutical waste not included in one of the above three definitions. As a best management practice, consider managing all pharmaceuticals not managed as hazardous waste through incineration at a regulated medical waste or municipal incinerator permitted to accept non-hazardous pharmaceutical waste. This decision can be made at the facility level.
(Pharmaceutical Waste Management Guidelines)
4.2 Blueprint of Pharmaceutical waste management
4.2.1 This Blueprint focuses primarily on three aspects of pharmaceutical waste management:
(1) Management of regulated hazardous pharmaceutical waste;
(2) Management of non-regulated hazardous pharmaceutical waste applying best management practices; and,
(3) Minimization of pharmaceutical waste
4.2.2 Getting Started
Given the complexity of implementation and the potential budgetary impacts (e.g., purchase of pharmaceutical waste containers and potentially increased disposal costs), the newly formed committee may find it valuable to arrange a presentation to senior management explaining the opportunities, challenges and financial implications of proper pharmaceutical waste management
· Pharmaceutical Waste (non controlled substances) may be disposed of by placing the material in a cardboard box (no larger than 12in x 12in x 12in). The box must be securely taped shut and labeled “Pharmaceutical Waste”. Arrangements can be made to bring it to the Student Health Pharmacy. Pharmaceutical waste that is a controlled substance must be returned to the Student Health Pharmacy.
· A copy of the proof of use log must accompany the controlled substance to the Student Health Pharmacy. Materiel Management arranges for both non-controlled and controlled substance waste to be picked up and disposed of by an outside vendor. Authorized personnel must transport controlled substances.(Eydie Pines, 2006)
4.3 Regulated Household Hazardous Waste
Expired or unwanted prescription or over-the- counter medications from households have traditionally been disposed of by ?ushing them down the toilet or a drain. Although this method of disposal prevents immediate accidental ingestion, has been demonstrated to cause adverse effects to ?sh and other aquatic wildlife. When the water is eventually reused, it can also cause unintentional human exposure to chemicals in medications.
4.3.1. Disposing of medications at home
Your unwanted medications may be disposed of in your trash. Follow these precautions to prevent accidental or intentional ingestion.
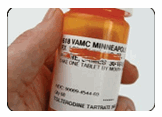
Figure 4- Keep Medication in its original container
The labels may contain safety information and the caps are typically childproof. Leaving the content information clearly visible, scratch the patient’s name out or cover it over with permanent maker.
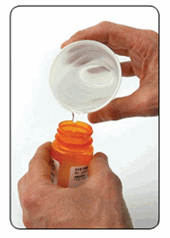
Figure 5-Modify the Medications to discourage consumption
For solid medications, such as pills or capsules:add a small amount of water to at least partially dissolve them.
For liquid medications: add enough table salt, ?our, charcoal, or nontoxic powdered spice, such as turmeric or mustard to make a pungent, unsightly mixture that discourages anyone from eating it.
For blister packs:Wrap the blister packages containing pills in multiple layers of duct or other opaque tape.

Figure 6-Seal and conceal
Seal and conceal.Tape the medication container lid shut with packing or duct tape, places it inside a non-transparent bag or container such as an empty yogurt or margarine tub to ensure that the contents cannot be seen.

Figure 7-Discard the Container
Discard the container in your garbage can—do not place in the recycling bin.
4.3.2. Managing other types of pharmaceutical waste
Unused ampoules, vials, and IV bagsshould not be opened (other than to scratch out the patient’s name). Wrap the container with tape to minimize breakage, and then place in an opaque plastic container (such as an empty yogurt or margarine tub). Wrap the outside of the container or bag with additional duct or shipping tape to prevent leakage and further obscure the contents. Dispose of the container in the trash.
Chemotherapy drugsmay require special handling. Work with your healthcare provider on proper disposal options for this type of medication. (Anonymous, 2008)
4.4 Understanding the Regulations
Focuses primarily on how the federal RCRA regulations apply to hazardous pharmaceutical waste management. It is divided into four major sections to provide a broad overview of the applicable regulations and an awareness of the overlap between RCRA and other statutes.(Eydie Pines, 2006)
4.4.1.Regulatory Bodies that Oversee Pharmaceutical Waste Management (Natasha Sharma et al,2010)
ü Environmental Protection Agency (EPA)
ü Department of Transportation (DOT)
ü Drug Enforcement Administration (DEA)
ü Occupational Safety and Health Administration (OSHA)
ü State Environmental Agencies,
ü State Pharmacy Boards, and
ü Local Publicly Owned Treatment Works (POTW)
The Biomedical Waste Rules of 1998 India’s Biomedical Waste Rules of 1998, which were amended twice in 2000, are based on the principle of segregation of communal waste from BMWs, followed by containment, treatment, and disposal of different categories of BMW The rules classify BMWs into 10 categories and require specific containment, treatment,
Main Features of India’s Biomedical Waste Rules of 1998 (amended twice in 2000
In 1998, India’s Ministry of Environment and Forests pre- pared and issued the Biomedical Waste (Handling and Management) Rules. The main features of the current rules are summarized here and in the below:
• Definition of biomedical waste:Any waste that is generated during the diagnosis, treatment, or immunization of human beings or animals, or in research activities pertain-ing to or in the production or testing of biological.
• Application of the Biomedical Waste Rules:The rules apply to all persons who generate, collect, receive, store, transport, treat, dispose, or handle BMWs in any form.
• Duty of occupier (operator):It is the duty of the occupier (operator) of a health care facility—that is, hospital, nursing home, clinic, dispensary, veterinary institution, animal house, pathological laboratory, blood bank—to ensure that BMWs are handled without any adverse effect to human health and the environment, and according to the prescribed treatment and disposal requirements in the Biomedical Waste Rules.
• Prescribed authority:State Pollution Control Boards (SPCBs) in states and Pollution Control Committees in territories are responsible for permitting and enforcing the requirements of the Biomedical Waste Rules.
• Permitting: Each occupier (operator) handling BMWs and providing services to 1,000 or more patients per month are required to obtain a permit from the prescribed authority.
• Recordkeeping:Each occupier (operator) is required to maintain records on the generation, collection, reception, storage, transportation, treatment, and disposal of BMWs. All records are subject to inspection and verification by the prescribed authority at any time.
• Accident reporting:Each occupier (operator) is required to report any accident related to the management of BMWs.
• Annual reporting: Each occupier is required to submit an annual report to the prescribed authority to provide information about categories and amounts of wastes generated and treated, and modes of treatment.
• Common disposal/incineration sites: Local public entities are required to provide common disposal/incineration sites, and the occupiers (operators) of such sites are required to comply with the Biomedical Waste Rules.
• Segregation, packaging, transportation, and storage:BMWs are not to be mixed with other waste. According to the Rules, BMWs are to be segregated into labeled bags/containers. Transportation of BMWs is to be con-ducted in authorized vehicles No untreated waste is to be stored more than 48 hours, unless special permission is obtained from the regulatory authorities.
• Standards:Technology and discharge standards for incineration, autoclaving, microwaving, liquid waste discharges, and deep burial are prescribed in the Biomedical Waste Rules. (Bekir Onursal, 2003)
4.4.2. The minimum criteria of SB 966 and of the Pharmaceutical Working Group for home-generated pharmaceutical
Pharmaceutical waste collection model programs are as follows:
1. Requires, at no additional cost to the consumer, the safe and environmentally sound take back and disposal of unused or expired home generated pharmaceuticals;
2. Ensures protection of the public’s health and safety and the environment;
3. Ensures protection of the health and safety of consumers, and employees;
4. Report to the Board the amounts of home-generated pharmaceutical waste collected for purposes of program evaluation for safety, efficiency, effectiveness and funding sustainability, and incidents of diversion of drugs for use or sale;
5. Protects against the potential for the diversion of drug waste for unlawful use or sale;
6. Provides notices and informational materials about potential impacts of improper disposal of pharmaceutical waste and options for proper disposal;
7. Subjects persons or businesses to consequences for failure to comply with model programs per SB 966 and related state and federal pharmaceutical and waste management statutes at the point of transportation, deposition, and consolidation;
8. Requires that once home-generated pharmaceutical waste has been consolidated at a facility or place of business, the waste must be managed as medical or hazardous waste. This would include all statutory requirements for storage and handling as medical or hazardous waste, the use of registered medical or hazardous waste haulers and approved treatment technology for disposal; and
9. Requires collection locations to have written policies and procedures to document their operations and compliance with this home-generated pharmaceutical waste collection program. (Eydie Pines, 2006)
4.4.3. Considering Best Management Practices for Non-Regulated Pharmaceutical Wastes
Incinerate as Hazardous Waste
· Formulations With a Listed Active Ingredient That is Not the Sole Active Ingredient
· All Chemotherapeutic Agents
· Drugs Meeting NIOSH Hazardous Drug Criteria
· Drugs Listed in Appendix VI of OSHA Technical Manual
· Drugs with LD50s Less Than or Equal to 50 mg/kg
· Carcinogenic Drugs
· Combination Vitamin/Mineral Preparations with Heavy Metals
· Endocrine Disruptors
4.5 Grappling with Hazardous Waste Combinations
This section provides guidance on how to manage combinations of hazardous waste and:
1. Personal Protective Equipment (PPE) and spill materials,
2. Regulated Medical Waste (RMW),
3. Sharps, and
4. Controlled substances.
4.5.1. Contaminated Personal Protective Equipment and Spill Materials
a) Listed Waste
PPE worn to protect employees from exposure to hazardous chemicals, materials used to perform routine cleaning or decontamination of Biological Safety Cabinets and glove boxes, and spill cleanup materials may become contaminated with hazardous waste.
b) Characteristic Waste
The contained-in policy applies differently to characteristic hazardous wastes. PPE and spill materials contaminated with characteristic wastes are hazardous only if the PPE and spill material exhibit a characteristic. However, it is best to be conservative and manage PPE that has been contaminated with a flammable waste or a highly corrosive waste as hazardous waste
4.5.2. Regulated Medical Waste
There will be situations where a combination waste that is both infectious Regulated Medical Waste and hazardous waste must be managed by a limited number of vendors that are permitted to handle both waste streams. The type of dispensing instrument used and the type of drug being administered both play an important role in determining how the resulting waste must be manage
4.5.3. Sharps
Often partially used syringes, vials or ampoules containing P- or U-listed hazardous chemicals or characteristic hazardous wastes are erroneously discarded in RMW sharps containers. Generally speaking, most vendors that manage sharps are not legally permitted to manage RCRA hazardous waste. These vendors are permitted to treat only infectious waste. As the generator, it is your responsibility to train staff that these distinct types of waste are managed differently and must be segregated (e.g., not to discard hazardous waste or waste that is both RMW and hazardous waste in sharps containers unless the containers are specially marked as both infectious and hazardous waste). If hazardous waste is improperly placed in a sharps container, the container should be relabeled as RMW and hazardous waste and managed by a vendor that is permitted to handle both waste streams.
4.5.4. Controlled Substances
Controlled substances are those drugs regulated by the Drug Enforcement Administration. Controlled substances must be destroyed so that they are beyond reclamation and two health care professionals must document the destruction. Since most hospitals no longer have ready access to incinerators in which to burn the drugs, the next most efficient way to accomplish this is through drain disposal.(Eydie Pines, 2006)
5. Minimizing Pharmaceutical Waste
As design and implement your pharmaceutical waste management program, there are inherent limitations on the substitution of a less hazardous drug since the hazardous nature of the chemical often provides the therapeutic effect. However, waste reduction can minimize compliance hassles, costs and risks. The following section provides a number of minimization opportunities to consider and explore.
5.1 Considering Lifecycle Impacts in the Purchasing Process
Implement a purchasing policy that includes restrictions and preferable purchasing practices. Examples include but are not limited to:
· Specifying that will not accept any drugs with less than one year dating unless they are only available with shorter expiration dates.
· Selecting products with less packaging. This is particularly relevant if the drug contains a P-listed constituent of concern. Packaging that comes in contact with drugs that contain P-listed chemicals must be managed as hazardous waste.
· Selecting products without preservatives whenever possible. Drugs such as some multi-dose vaccines, and eye and ear preparations, may contain the preservatives thimerosal or phenylmercuric acetate. Manufacturers are moving away from these controversial mercury based chemicals to less toxic alternatives. Always check references, such as eFacts, manufacturers’ websites, and group purchasing organization (GPO) to see if mercury free alternatives are available.
· Consider single dose containers, which do not need a preservative.
5.2. Maximizing the Use of Opened Chemotherapy Vials
Sometimes opened chemotherapy vials are retained for possible use in oncology pharmacies until they expire. However, this is not always the case especially in lower volume pharmacies. Consider possible ways to maximize usage of these partial vials to minimize waste and save money.
5.3. Implementing a Samples Policy
The ability of the pharmacy to control sampling within the organization is often based on the political realities of the organization and who controls the medical staff and who owns the associated clinics. The fact that samples are outdated and need to be discarded indicates that pharmaceutical representatives are over-supplying samples, physicians’ offices are not rotating sample inventory, physicians are not providing samples to patients, or drug usage patterns have changed for particular drugs. Whatever the cause, the organization is incurring added costs and liabilities managing waste drug samples and should adopt a policy that addresses the acceptance and end of life management of samples.
5.4. Labeling Drugs for Home Use
These patient-specific medications are returned to the pharmacy for destruction when the patient is discharged. In the current system, these procedures would cause delays in the discharge process that would be unacceptable to the patient. Consider how the system could be changed to include preauthorized discharge orders for maintenance medications and possible label production in the units. This would reduce waste and save money for both the health care system and the patient. Smaller hospitals in particular should consider relabeling for home use. This is more difficult logistically in large facilities, and patients often will not wait for the new orders to be communicated to the pharmacy and new labels attached
5.5. Priming and Flushing IV Lines with Saline Solution
Pharmacies should prime all chemotherapy IVs with saline prior to dispensing and nurses should flush the tubing after administration. These practices not only insure the patient receives the full dosage but also reduces the opportunity for employee exposure and enables IV tubing and bags to be managed as trace chemotherapy waste.
5.6. Examining the Size of Containers Relative to Use
Certain medications are routinely administered in doses that result in waste or in dispensing devices that could be “lightweighted.” Consider conducting a survey of all drugs routinely wasted in your facility due to the prepared product being too large for complete administration.
Through an on-site review, one hospital found that Lopressor is purchased in 100 mg tablets but only 50 mg were routinely administered. Therefore, 50 mg of Lopressor were routinely wasted. Lopressor is available in 50 mg tablets and the generic version, metoprolol, is available in 50 mg or 25 mg tablets. Changes in purchasing patterns can save your hospital money by reducing the amount of pharmaceutical waste that you generate.
There are variations in the weight of dispensing instruments. For example, consider using two -part polyolefin IV devices to administer antibiotics (e.g. Duplex) that weigh one-third less than traditional glass vial/PVC IV bag alternatives.
5.7. Replacing Prepackaged Unit Dose Liquids with Patient-Specific Oral Syringes
To avoid having to routinely waste the remaining contents of 5 ml and 10 ml prepackaged unit dose liquids, consider moving to patient-specific oral syringes, especially in the neonatal and pediatrics units where doses are very customized and patient-specific. This practice is especially useful for drugs like chloral hydrate, which is also a controlled substance. Eliminating waste also saves nursing time while preventing the usual drain disposal of a hazardous waste.
5.8. Controlled Substances
Due to the difficulty in disposing of a controlled substance that is also a hazardous waste and the desire to avoid the drain disposal of all pharmaceutical wastes, it is best to try to eliminate the generation of these wastes to the degree possible.
5.9. Delivering Chemotherapy Drugs
Replace brown paper bags in which chemotherapy is delivered to the floor with hard plastic buckets. This will not only reduce waste but also provide greater spill and leak protection during transport.
5.10. Monitoring Dating on Emergency Syringes
Generally, hospitals replace epinephrine and nitroglycerin syringes and vials on general crash carts when they are within three months or less of their expiration date. These products can be moved from general crash carts to the Emergency Department or ICU/CCU three months prior to outdate to avoid discarding them.
5.11. Reviewing Inventory Controls to Minimize Outdates
Create a tighter inventory control program to limit the amount of original manufacturers’ containers and repacks that expire before use. Resources spent on the management of expired products are resources lost.(Eydie Pines, 2006)
5.12. Considering the Management Options
Option I: Segregating at the Point of Generation
Ideally, pharmaceutical hazardous waste will be segregated at the point of generation and discarded in hazardous pharmaceutical waste containers that are located as conveniently as practical to the point of generation. Personnel that are trained to handle hazardous waste transfer the containers from the satellite accumulation areas at the point of generation to the central storage accumulation area (described in Step 9) where they are picked up by a permitted hazardous waste vendor.
Table 9. Minimizing of Pharmaceutical waste
|
Type of Waste |
Description of Waste
|
Description of Container |
Type of Treatment |
|
Hazardous Toxic and BMP Toxic
|
P, U and toxic D wastes All bulk non-listed chemotherapy drugs Non-listed toxic drugs PPE with visible contamination |
Black or dark blue |
Incineration at RCRA hazardous waste facility
|
|
Hazardous Ignitable
|
D001 wastes |
Black or dark blue |
Incineration at RCRA hazardous waste facility |
|
Incineration at RCRA hazardous waste facility
|
Hazardous and Infectious Hazardous toxic wastes and BMP toxic wastes combined with RMW Entire contents of sharps containers if P-listed hazardous waste was properly or improperly discarded in container |
Sharps container (e.g., white/blue) with a Hazardous Waste and RMW label applied |
Incineration at a facility permitted to handle RCRA hazardous waste and RMW |
|
Trace Chemotherapy
|
“RCRA empty” vials of chemotherapy agents, syringes/needles, IVs, PPE used to prepare or Administer chemotherapy without visible contamination |
Yellow or white |
Incineration at RMW facility
|
|
Drain Disposal |
Controlled substances, NaCl, dextrose, vitamins, electrolytes |
Sewer |
Local POTW (permission required) |
|
BMP Non-Regulated
|
All other drugs |
White with blue top (most common) |
Incineration at RMW or MSW facility |
Should Ignitable Wastes Be Managed Separately
· Waste compatibility
· Vendor’s requirements
· Quantity of ignitable waste generated
· Potential cost savings from managing ignitable waste separately
· Feasibility of combining pharmaceutical ignitable waste with other ignitable waste
· Local and national fire code requirements
Option II: Centralizing Segregation
In centralized segregation, all drug waste is discarded in hazardous waste containers that are located at the point of generation. The nursing and pharmacy staffs are not required to make decisions regarding the final destination of the drug waste. However, bulk chemotherapy and trace chemotherapy waste are an exception, as these wastes should be segregated in the patient care areas to avoid employee exposure. Overall, this option minimizes the number of containers that must be maintained at the point of generation. Ultimately, the containers are moved to a central storage accumulation area and either hospital personnel that have received hazardous waste training or a hazardous waste vendor manually sort the waste into appropriate containers. Therefore, containers for all types of waste generated at the facility should be available in the manual sorting area. The requirements for a hazardous waste storage accumulation area apply to the sorting location.
It is still necessary to determine which drugs become hazardous waste when discarded. Pre-labeling may be more time efficient for segregation personnel as lists are difficult to maintain and use in this environment. However, generally speaking, hospitals that are currently implementing this approach are using an easily accessible list identifying the appropriate bin for all of the drugs and drug containers generated by the facility.
Option III: Managing All Drug Waste as Hazardous
In small facilities with fewer than 50 beds, this may be the simplest and most economical solution in the long run. For large facilities, pilot programs have documented this approach could result in added hazardous waste costs of in excess of USD 1,000,000 annually. Analysis should provide the best answer as to whether this approach is a viable alternative. Careful cost modeling or a pilot program may be the only way to make this determination.(Eydie Pines, 2006)
5.13. Getting Ready for Implementation
5.13.1. Locating Your Satellite Accumulation Areas
To maximize compliance, satellite accumulation sites should be as conveniently located to where drug waste is generated as possible. Consider locating hazardous waste containers in the sterile products clean room and in the main pharmacy and in the soiled utility rooms or near medication carts in a secure area in the nursing units. Containers cannot be stored near a working sink or open floor drain without secondary containment. A locked wall unit that could be placed in the patient’s room is under development.
Each container must be spill-proof, leak-proof, compatible with the waste to be contained, and labeled as hazardous waste with the appropriate waste stream noted (toxic or ignitable). The containers must be kept closed when active addition is not occurring. This can be a challenging requirement but one that needs to be taken seriously. During the administration of chemotherapy drugs, for example, the containers may be open, as active addition will be occurring. However, these containers must not be left open when they will not be added to for a period of time (e.g., lunch breaks, over night, when the satellite operator/nurse is not present). Violations of this requirement are often cited during compliance inspections. The simplest way to insure timely and consistent container closure is the use of wire frame trolleys or hard plastic carriers that are opened with a foot pedal, leaving the hands free and closing automatically.
5.13.2. Evaluating Your Storage Accumulation Area
When hazardous drug waste containers are removed from satellite accumulation areas in the pharmacy, nursing units, and clinics, they must be transferred to and stored in an area known as a hazardous waste storage accumulation area.
5.13. 3. Conducting a Pilot Program
The three highest profile areas, the pharmacy, any oncology units or outpatient clinics and the ICU, should be considered for an initial pilot program. Not only are they easier to control than some units, these are the areas regulatory agents will be sure to examine.
5.13.4. Putting It All Together: Pharmaceutical Waste Management Policies and Procedures
After conducting a pilot and before rolling out the program to the entire facility, new policies and procedures for hazardous drug segregation need to be drafted. Involve safety, nursing, pharmacy, and environmental services management in the drafting of these new policies and procedures. All aspects of your pharmaceutical waste management and minimization program should be incorporated into existing policies and procedures or if necessary new ones should be created. In addition, it is helpful to set up a specific manual of pharmaceutical waste management and minimization policies and procedures to have all of the steps documented in one location. This overarching operating manual can reference other appropriate policies and procedures, such as chemotherapy preparation, administration, and disposal or general hazardous waste management.
5.14. Policies and Procedures
At a minimum pharmaceutical waste management policies and procedures should be
• developed to detail the organization’s approach to:
• Identifying drugs that must be managed as hazardous waste;
• Determining which non-regulated drugs will be managed as hazardous waste
• Labeling drugs to facilitate segregation of hazardous waste;
• Segregating waste streams;
• Training staff (e.g., which staff, what information and how often);
• Setting up and managing satellite accumulation and storage accumulation areas;
• Preparing and maintaining hazardous waste manifests;
• Determining their hazardous waste generation status;
• What criteria are used for hazardous waste selection;
• Scheduling regular program reviews;
• Keeping management informed; and,
• Using pharmaceutical waste management as a stepping-stone to a facility-wide
• Environmental Management System (EMS)
(Eydie Pines, 2006)
6. Pharmaceutical Waste Treatment and Disposal
Pharmaceutical waste must be treated in one of the following ways:
1. Steam sterilized (autoclaved) at an approved autoclave facility, then disposed of as non-regulated solid waste
2. Picked up by an approved vendor and treated outside the campus
3. Discharged to a public sewage system if the waste is liquid or semi-liquid (i.e., human blood, body fluids, and biological solutions) and has been chemically decontaminated (such as use of chlorine compounds, quaternary ammonium compounds, phenolic compounds, etc.).
4. Recognizable human anatomical remains must be disposed by incineration or interment unless otherwise hazardous
6.1. Pharmaceutical Waste Treatment and Disposal Technologies Specified in India’s Pharmaceutical Waste Rules
6.1.1. Incineration.
Incineration is a high-temperature oxidation process that involves combustion of the organic portion of BMW components, producing gaseous emissions and inorganic solid residues (ash).These emissions include steam, carbon dioxide, nitrogen oxides, particulate matter, and toxic substances (for example, metals, halogenic acids). In addition, under suboptimal combustion conditions, carbon monoxide and hazardous pollutants such as dioxins and furans may be emitted. Incineration significantly reduces waste volumes (typically 85 to 95 percent), and eliminates pathogens from BMWs. Incineration is not suitable for such health care wastes as pressurized gas containers, large amounts of reactive chemical wastes, wastes treated with halogenated chemicals, halogenated plastics such as polyvinyl chloride, wastes with mercury or cadmium (such as broken thermometers, used lead or mercury batteries), or radiographic wastes. Incinerators that meet the CPCB draft incineration regulations must have a sophisticated (for example, double- chamber) design and include a scrubber as the air pollution control equipment. Ash from these incinerators must be disposed of in a secure landfill. Such incinerators are associated with high investment and operating costs and require highly skilled operating personnel.
6.1.2. Autoclaving.
Autoclaving uses saturated steam in direct contact with the BMW in a pressure vessel at time lengths and temperatures sufficient to kill the pathogens. The Biomedical Waste Rules specify the minimum temperature, pressure, and residence time for autoclaves for safe disinfection. Autoclaving is not suitable for human anatomical, animal, chemical, or pharmaceutical wastes. Before autoclaving, BMWs require shredding to an acceptable size, an operation that would involve frequent breakdown. Autoclaving produces a waste that can be land filled with municipal waste. A wastewater stream is generated that needs to be disposed of with appropriate controls. Autoclave operation requires qualified technicians, and medium investment and operating costs. Hydroclaving is similar to autoclaving except that the BMW is heated indirectly through the outer jacket of the vessel. The BMW is continuously tumbled in the chamber during the process.
6.1.3. Microwaving.
Application of an electromagnetic field over the BMW provokes the liquid in the waste to oscillate and heat up, destroying the infectious components by conduction. This technology is effective if the ultraviolet radiation reaches the waste material. Before microwaving, BMWs require shredding to an acceptable size and humidification. Microwaving is not suitable for human anatomical, animal, chemical, or pharmaceutical wastes, or for large metal parts. Microwaving produces a waste that can be land filled with municipal waste. The advantages of this treatment technology are its small electrical energy needs and no steam requirement. The disadvantages include the need for qualified technicians and frequent breakdown of shredders. This technology requires medium investment and operating costs. Chemical disinfection. Addition of strong oxidants—like chlorine compounds, ammonium salts, aldehydes, or phenol compounds—kills or inactivates pathogens in the BMW.
6.1.4. Chemical disinfection:
Chemical disinfection is most suitable for treating liquid wastes such as blood, urine, stools, or health care facility sewage. However, microbiological cultures, mutilated sharps, or shredded solids can also be treated by chemical disinfection. Disinfection efficiency depends on such factors as the type and amount of chemical used, and the extent and duration of contact between the disinfectant and the BMW. To enhance the contact, shredding of solid BMW would be needed. As chemical disinfectants have hazardous (in particular, toxic) properties, users should wear protective clothes. Chemical disinfectants should not be discharged to surface waters, and no large quantities should be allowed into sewers.
6.1.5. Deep burial.
The Biomedical Waste Rules require that human anatomical and animal wastes in cities with population less than 500,000 and in rural areas be disposed of by deep burial. Accordingly, the deep burial site should be pre- pared by digging a pit or trench of about 2 meters deep in an area that is not prone to flooding or erosion, and where the soil is relatively impermeable, there are no inhabitants or shallow wells in the vicinity, and the risk to surface water contamination is remote. The pit should be half-filled with the BMW, and then covered with lime within 50 cm of the surface, before filling the rest of the pit with soil. On each occasion when BMW is added to the pit, a layer of 10 cm of soil should be added to cover the waste.
6.1.6. Secure land filling.
Secure land filling involves disposal of solid BMWs at a landfill designed and operated to receive hazardous wastes. The Biomedical Waste Rules require disposal of discarded medicines, cytotoxic drugs, solid chemical wastes, and incineration ash in secured landfills.
6.1.7. Municipal landfilling.
Municipal land filling involves disposal of communal HCWs and disinfected solid BMWs at a land- fill designed and operated to receive municipal solid waste. (Bekir Onursal, 2003)
7. Labeling Requirements for Pharmaceutical Waste Containers
Medical waste contained in a red biohazard bag must be labeled with the words “Biohazardous Waste” or with the international biohazard symbol and the word “BIOHAZARD.”

Figure 8- Labeling of Pharmaceutical Container Waste (C. Visvanathan, 2006)
7.1 Pharmaceutical Waste Storage and Handling in BSL-2 or BSL-3 Laboratories
• Tiered biohazard bags to prevent leakage or expulsion of contents during future storage, handling, or transport.
• Place red biohazard bags for storage, handling, or transport in a rigid secondary container.
• Rigid secondary containers must be leak resistant, have tight fitting covers and be kept clean and in good repair. Containers may be any color and labeled with the words “Biohazardous Waste” or the international biohazard symbol and the word “BIOHAZARD” on the lid and on the sides so as to be visible from any lateral direction.
• BSL-1 wastes can be placed in a red bag. These wastes are considered medical waste after being placed in a red bag.
7.1.1 Labeling Pharmacy Shelf Stickers:Place hazardous waste identification labels on the shelf stickers of all pharmaceuticals that have been identified as RCRA Hazardous Waste in Section 1.a. or Non-RCRA Antineoplastic Pharmaceutical Waste in Section, if your installation chooses to follow the Section proposed guidelines for Best Management Practices then place identification labels on the respective shelf stickers for these pharmaceuticals as well.
7.1.2 Training of Pharmacy Personnel:Train pharmacy personnel to label all hazardous waste pharmaceuticals with the appropriate hazardous waste notification sticker when dispensing the pharmaceutical directly to the nursing units.
7.1.3 Labeling Compounded Items and IV Admixture:Develop a system for labeling all compounded preparations and IV admixtures that meet the criteria of a hazardous waste when discarded.
7.1.4 Medical Waste Segregation :Separate all medical waste from other waste at the point of origin.
7.1.5 Labeling Requirements for Biohazardous Waste: Non-medical waste (BSL-1 waste) may be autoclaved in autoclavable white opaque bags in any autoclave and disposed of as solid waste (Eydie Pines, 2006)

8. CONCLUSION
Pharmaceutical waste management requires:
• Segregation of the hospital wastes according to the available disposal technology.
• Employment of cost-effective and available relevant technology.
• Possibilities of recycling to be explored in a scientific and hygienic manner for permissible items.
• Setting up of common medical waste treatment facilities for/by different hospitals such as transportation of the hazardous waste to the common disposal system to reduce expenditure.
• Safety of medical staff/rag-pickers, by the use of gloves and masks and housekeeping aspects (drinking water, sewage system of the hospitals).
• Implementation of recycling etiquette by medical and paramedical personnel.
• Training of Municipality workers by medical personnel in handling of medical waste to avoid risks and health hazards.
• Implementations of legislations pertaining to hygiene of freelance workers such as rag pickers in the recycling industry.
The management of Pharmaceutical wastes poses a great challenge to the policy planners, city administrators, medical personnel and workers in the recycling industry. There is a need for adopting a cost-effective system for providing better medical waste treatment facilities and reduce the amount of waste generation by awareness and education of all concerned.
9.BIBLIOGRAPHY
* Eydie Pines, 2006, Managing Pharmaceutical Waste: A 10 Step Blueprint for Health Care Facilities In the United States, h2e-online.org/docs/h2epharmablueprint41506.pdf [Accessed 2-Feb-2011]
* Medical Waste Disposal Guidelines; ehs.uci.edu/programs/biosafety/medwasteguide.pdf
* Bekir Onursal, 2003,Health Care Waste Management in India; epa.gov
• Pharmaceutical Waste Management Guidelines
• Natasha Sharma et al,2010,Pharmaceutical Waste management: A Challenge To Make Environment Ecofriendly < ijrap.net/issue-two/332-338.pdf>[Accessed 9-Dec.-2010]
* C. Visvanathan, 2006, Medical Waste Management Issue In Asia [Accessed 25-Feb-2011]
* Pharmaceutical Waste : Disposing of Unwanted Medication [Accessed 4-march-2011]
* Dr. Saurabh Sikka, Biomedical Waste in Indian Context [ Accessed 17-jan-2011].
* Pharmaceutical Waste Management [Accessed 12-feb-2011]
Reference ID: PHARMATUTOR-ART-1028
FIND OUT MORE ARTICLES AT OUR DATABASE









