ABOUT AUTHOR:
Deepika Kadodiya
Mahakal Institute Of Pharmaceutical Studies,
Ujjain M.P.
deepikakadodiya05@gmail.com
ABSTRACT
Niosomes are a novel drug delivery system that are finding application in drug targeting antineoplastic treatment, leishmaniasis treatment, delivery of peptide drugs, transdermal drug delivery systems, cosmetics, immunological application of peptide drugs, carriers for haemoglobin, sustained release etc. A niosome is a non-ionic surfactant based liposomes. It is formed by cholesterol with the use of other excipients. The various methods used in the formation of niosomes such as ether injection method, sonication, microfluidization, hand shaking method for the different types of niosomes. The goal of this article focuses on the recent advances in niosomal drug delivery, potential advantages over other delivery systems, formulation methods, methods of characterization, limitation of niosomes, comparision of niosomes v/s liposomes, application of niosomes, marketed products of niosomes and the current research in the field of niosomes.
REFERENCE ID: PHARMATUTOR-ART-1901
INTRODUCTION
The skin that covers throughout the body severs as a platform for drug delivery, but the stratum corneum the outer layer act as an obstacle for the delivery of drug through the skin1. Several technologies was developed to bypass or modulate this barrier, thereby delivers definite amount of drug at a defined rate to the dermal microcirculation; which includes physical, chemical and carrier approaches2. Thereby, making it a more prominent carrier than the oral delivery as it circumvents the variables that influence gastrointestinal absorption, overcome the hepatic metabolism and delivers the drug at controlled and constant rate thereby reducing the side effects3. Colloidal carrier have distinct advantages over conventional drug delivery as it act as drug containing reservoirs, modification of the particle composition or surface can adjusts the release rate to the target site. Even though, these carriers produce some problem with the industrial production and clinical application, are likely to play an increasingly important role in drug delivery4,5. Among the various colloid carriers, liposome and niosomes can encapsulate both hydrophilic and lipophilic drugs. The hydrophilic drug is encapsulated inside the vesicles whereas the lipophilic drug is partitioned between the hydrophilic domains. Liposomes are produced by the self-assembly of phospholipids in aqueous phase to form bilayer which may be spherical unilamellar or multilamellar vesicles. They were considered to as an efficient carrier to transdermal drug delivery as it can loosen the stratum corneum and hence help in the penetration of drug. Though they produce potent action in pharmaceutics as drug delivery, still produces significant problems. That is, liposome may undergo some problems like degradation by hydrolysis in aqueous solution, sedimentation and aggregation on storage and cannot sterilize for clinical use. Still, chemical instability like oxidation of phospholipids was not avoided. These pave the way to the discovery of non-ionic surfactant vesicles known as niosomes. Niosomes first reported in the seventies in the field of cosmetics and now used in drug targeting. In both these colloidal carrier, phospholipids and non-ionic surfactant used act as penetration enhancer and hence can overcome the barrier of transdermal drug delivery. Although the niosomes, overcame the problem associated with chemical stability on storage, has some physical problems like aggregation, fusion, leakage of drug from the vesicles and hydrolysis of drug on storage were produced6.
Non-ionic surfactant vesicles or niosomes whose structure and properties are similar to liposomes have been developed. Niosomes are microscopic lamellar structures formed on admixture of non-ionic surfactant of the alkyl or dialkyl polyglycerol ether and cholesterol with subsequent hydration in aqueous media and are capable of entrapping hydrophilic and hydrophobic solutes.7-10 Technically, niosomes are promising drug carriers because they posses greater stability, less purity variability, and lower cost. Niosomes also have the advantages of simple method of production for the routine laboratory use and the possibility of large scale production, when needed.11,12
Historical aspect
Niosomes were first reported in the seventies as a feature of the cosmetic industry by Vanlerberghe et al, Handjani-vila et al., Van Abbeexplained that the non-inonic surfactants are preferred because the irritation power of surfactants decreases in the following order:13-19 cationic > anionic > ampholytic > non-ionic.
Vesicular System
In recent years, vesicles have become the vehicle of choice in drug delivery. Lipid vesicles were found to be of value in immunology, membrane biology, diagnostic techniques, and most recently, genetic engineering. Vesicles can play a major role in modelling biological membranes, and in the transport and targeting of active agents.20-23 Conventional chemotherapy for the treatment of intracellular infections is not effective, due to limited permeation of drugs into cells. This can be overcome by use of vesicular drug delivery systems. Encapsulation of a drug in vesicular structures can be predicted to prolong the existence of the drug in systemic circulation, and perhaps, reduces the toxicity if selective uptake can be achieved. The phagocytic uptake of the systemic delivery of the drug-loaded vesicular delivery system provides an efficient method for delivery of drug directly to the site of infection, leading to reduction of drug toxicity with no adverse effects. Vesicular drug delivery reduces the cost of therapy by improved bioavailability of medication, especially in case of poorly soluble drugs. They can incorporate both hydrophilic and lipophilic drugs. Vesicular drug delivery systems delay drug elimination of rapidly metabolizable drugs, and function as sustained release systems. This system solves the problems of drug insolubility, instability, and rapid degradation. Consequently, a number of vesicular delivery systems such as liposomes, niosomes, pharmacosomes etc, were developed.24-26
Definition of Niosomes
Niosomes are a novel drug delivery system, in which the medication is encapsulated in a vesicle. The vesicle is composed of a bilayer of non-ionic surface active agents and hence the name niosomes. The niosomes are very small, and microscopic in size. Their size lies in the nanometric scale. Although structurally similar to liposomes, they offer several advantages over them. Niosomes have recently been shown to greatly increase transdermal drug delivery and also can be used in targeted drug delivery, and thus increased study in these structures can provide new methods for drug delivery.
Niosomes are non-ionic surfactant vesicles obtained on hydration of synthetic nonionic surfactants, with or without incorporation of cholesterol or other lipids.They are vesicular systems similar to liposomes that can be used as carriers of amphiphilic and lipophilic drugs. Niosomes are promising vehicle for drug delivery and being non-ionic, it is less toxic and improves the therapeutic index of drug by restricting its action to target cells.27-29
COMPONENTS OF NIOSOMES
Surfactant: Non-ionic surfactants are used; they considered the important structural component. They act as Vesicle Forming Agents. The nature of vesicles formed depends upon HLB value in addition, phase transition temperature. HLB value is a good indicator to predict the vesicle formation and entrapment efficiency. HLB number in between 4 and 8 is compatible with vesicle formation. Another important parameter is the phase transition temperature, higher TºC are more likely in the ordered gel form forming less leaky bilayer, thus having higher entrapment efficiency, while surfactants of lower T° C are more likely in the less ordered liquid form.
Cholesterol: Cholesterol acts as “vesicular cement” in the molecular space that formed by the aggregation of monomer to form the bilayer. Thereby increasing the rigidity decreases the permeability drug through the membrane and hence improves the entrapment efficiency. However, beyond certain concentration cholesterol will compete with the drug for the space within the bilayer, thereby excluding drug and can disrupt the regular linear structure of vesicular membrane. In addition to this, it can also act stabilizing agent.
Solvents: The solvent can act as penetration enhancer and in turn affect the vesicular size formation. Solvents commonly used are alcohols, mainly, ethanol, propanol, butanol, isopropanol. Researchers have reported that ethanol showed larger vesicular size due to the slow phase separation as it has greater solubility in water, whereas due to the branching of isopropanol it showed smaller vesicular size.In addition, reports suggest that the drug penetration is maximal for isopropanol due to the reason that the branched structure will act as co-surfactant and might loosen the bilayer packing resulting into the increased release of drug.30-32
STRUCTURE OF NIOSOMES
Niosomes are microscopic lamellar structures, which are formed on the admixture of non-ionic surfactant of the alkyl or dialkyl polyglycerol ether class and cholesterol with subsequent hydration in aqueous media. Structurally, niosomes are similar to liposomes, in that they are also made up of a bilayer. However, the bilayer in the case of niosomes is made up of non-ionic surface active agents rather than phospholipids as seen in the case of liposomes. Most surface active agents when immersed in water yield micellar structures, however some surfactants can yield bilayer vesicles which are niosomes. Niosomes may be unilamellar or multilamellar depending on the method used to prepare them. The niosome is made of a surfactant bilayer with its hydrophilic ends exposed on the outside and inside of the vesicle, while the hydrophobic chains face each other within the bilayer. Hence, the vesicle holds hydrophilic drugs within the space enclosed in the vesicle, while hydrophobic drugs are embedded within the bilayer itself.
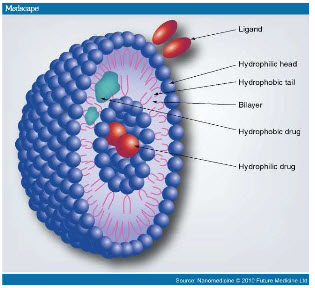
Fig.1 Structure of Niosomes
A typical niosome vesicle would consist of a vesicle forming ampiphile i.e. a non-ionic surfactant such as Span-60, which is usually stabilized by the addition of cholesterol and a small amount of anionic surfactant such as diacetyl phosphate, which also helps in stabilizing the vesicle.33
Types Of Niosomes
Small unilamellar vesicles- These are commonly produced by sonication, and French Press procedures. Ultrasonic electrocapillary emulsification or solvent dilution techniques can be used to prepare SUVs. The approximate size of these vesicles is 0.025-0.05 μm
Multilamellar vesicles- It consists of a number of bilayer surrounding the aqueous lipid compartment separately and exhibit increased-trapped volume and equilibrium solute distribution, and require hand-shaking method. They show variations in lipid compositions. The approximate size of these vesicles is 0.5-10 µm diameter
Large unilamellar vesicles- The injections of lipids solubilised in an organic solvent into an aqueous buffer, can result in spontaneous formation of LUV. But the better method of preparation of LUV is Reverse phase evaporation, or by Detergent solubilisation method. The approximate size of these vesicles is greater than 0.10 μm.
METHODS OF PREPARATION
A. Ether injection method
This method provides a means of making niosomes by slowly introducing a solution of surfactant dissolved in diethyl ether into warm water maintained at 60°C. The surfactant mixture in ether is injected through 14-gauge needle into an aqueous solution of material. Vaporization of ether leads to formation of single layered vesicles. Depending upon the conditions used, the diameter of the vesicle range from 50 to 1000 nm.
Fig.2 14 Gauge needle
B. Hand shaking method Or Thin film hydration technique
The mixture of vesicles forming ingredients like surfactant and cholesterol are dissolved in a volatile organic solvent (diethyl ether, chloroform or methanol) in a round bottom flask. The organic solvent is removed at room temperature (20°C) using rotary evaporator leaving a thin layer of solid mixture deposited on the wall of the flask. The dried surfactant film can be rehydrated with aqueous phase at 0-60°C with gentle agitation. This process forms typical multilamellar niosomes. Thermosensitive niosomes were prepared by Raja Naresh et al by evaporating the organic solvent at 60°C and leaving a thin film of lipid on the wall of rotary flash evaporator. Theaqueous phase containing drug was added slowly with intermittent shaking of flask at room temperature followed by sonication.
Twelve milligram each of span 60 and cholesterol (1:1) ratio were dissolved in ether and the solvent was evaporated at room temperature, using rotary flash evaporator. 10 ml of the aqueous phase containing drug (1.2 mg/ml) was added to this at 7 oC and shaken for about 15 min resulting in good dispersion of the mixture.

Fig.3 Rotating evaporator
C. Sonication
A typical method of production of the vesicles is by sonication of solution as described by Cable. In this method an aliquot of drug solution in buffer is added to the surfactant/cholesterol mixture in a 10-ml glass vial. The mixture is probe sonicated at 60°C for 3 minutes using a sonicator with a titanium probe to yield niosomes.34,35

Fig.4 Sonicator
D. Micro fluidization
Micro fluidization is a recent technique used to prepare unilamellar vesicles of defined size distribution. This method is based on submerged jet principle in which two fluidized streams interact at ultra high velocities, in precisely defined micro channels within the interaction chamber. The impingement of thin liquid sheet along a common front is arranged such that the energy supplied to the system remains within the area of niosomes formation. The result is a greater uniformity, smaller size and better reproducibility of niosomes formed.
E. Multiple membrane extrusion method
Mixture of surfactant, cholesterol and dicetyl phosphate in chloroform is made into thin film by evaporation. The film is hydrated with aqueous drug solution and the resultant suspension extruded through polycarbonate membranes, which are placed in series for upto 8 passages. It is a good method for controlling niosome size.36
F. Reverse Phase Evaporation Technique (REV)
Cholesterol and surfactant (1:1) are dissolved in a mixture of ether and chloroform. An aqueous phase containing drug is added to this and the resulting two phases are sonicated at 4-5°C. The clear gel formed is further sonicated after the addition of a small amount of phosphate buffered saline (PBS). The organic phase is removed at 40°C under low pressure. The resulting viscous niosome suspension is diluted with PBS and heated on a water bath at 60°C for 10 min to yield niosomes.37
G. Trans membrane pH gradient (inside acidic) Drug Uptake Process (remote Loading)
Surfactant and cholesterol are dissolved in chloroform. The solvent is then evaporated under reduced pressure to get a thin film on the wall of the round bottom flask. The film is hydrated with 300 mM citric acid (pH 4.0) by vortex mixing. The multilamellar vesicles are frozen and thawed 3 times and later sonicated. To this niosomal suspension, aqueous solution containing 10 mg/ml of drug is added and vortexed. The pH of the sample is then raised to 7.0-7.2 with 1M disodium phosphate. This mixture is later heated at 60°C for 10 minutes to give niosomes.
H. The Bubble Method
It is novel technique for the one step preparation of liposomes and niosomes without the use of organic solvents. The bubbling unit consists of round-bottomed flask with three necks positioned in water bath to control the temperature. Water-cooled reflux and thermometer is positioned in the first and second neck and nitrogen supply through the third neck. Cholesterol and surfactant are dispersed together in this buffer (pH 7.4) at 70°C, the dispersion mixed for 15 seconds with high shear homogenizer and immediately afterwards “bubbled” at 70°C using nitrogen gas.38
I. Formation of niosomes from proniosomes
Another method of producing niosomes is to coat a water-soluble carrier such as sorbitol with surfactant. The result of the coating process is a dry formulation. In which each water-soluble particle is covered with a thin film of dry surfactant. This preparation is termed “Proniosomes”. The niosomes are recognized by the addition of aqueous phase at T > Tm and brief agitation.
T=Temperature.
Tm = mean phase transition temperature
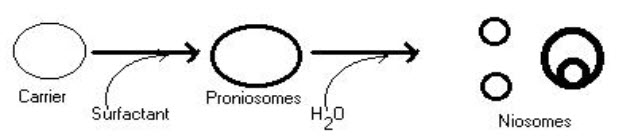
J. Lipid layer hydration method
Twelve milligram each of span 60 and cholesterol (1:1) were dissolved in chloroform and the solvent was evaporated using rotary flash evaporator. 10 ml of phosphate buffer saline PH7.4 containing drug (1.2 mg/ml) was added to the dried thin film with gentle agitation. The mixture was intermittently mixed on a vortex mixer. Sonic dispersion of the mixture was carried out at 25 0C using probe sonicator set at 200 watts for 1 minute.39
Table 1-Drugs incorporated into niosomes by various methods
|
Method of preparation |
Drug incorporated |
|
Ether Injection |
Sodium stibogluconate Doxorubicin |
|
Hand Shaking |
Methotrexate Doxorubicin |
|
Sonication |
9-desglycinamide 8-arginine Vasopressin Oestradiol |
COMPARISION OF NIOSOMES v/s LIPOSOMES
a) Niosomes are now widely studied as an alternative to liposomes, which exhibit certain disadvantages such as –they are expensive, their ingredients like phospholipids are chemically unstable because of their predisposition to oxidative degradation, they require special storage and handling and purity of natural phospholipids is variable.
b) Differences in characteristics exist between liposomes and niosomes, especially since niosomes are prepared from uncharged single-chain surfactant and cholesterol whereas liposomes are prepared from double-chain phospholipids (neutral or charged).
c) Niosomes behave in-vivo like liposomes, prolonging the circulation of entrapped drug and altering its organ distribution and metabolic stability. Encapsulation of various anti neoplastic agents in these carrier vesicles has been shown to decrease drug induced toxic side effects, while maintaining, or in some instances, increasing the anti-tumor efficacy. Such vesicular drug carrier systems alter the plasma clearance kinetics, tissue distribution, metabolism and cellular interaction of the drug. They can be expected to target the drug to its desired site of action and/or to control its release.
d) As with liposomes, the properties of niosomes depends both on the composition of the bilayer and on method of their production. It was observed by Baillie et al that the intercalation of cholesterol in the bilayers decreases the entrapment volume during formulation and thus entrapment efficiency. As the concentration of cholesterol increases, entrapment efficiency decreases.40,41
CHATACTERIZATION OF NIOSOMES
Entrapment efficiency
After preparing niosomal dispersion, unentrapped drug is separated by dialysis, centrifugation, or gel filtration as described above and the drug remained entrapped in niosomes is determined by complete vesicle disruption using 50% n-propanol or 0.1% Triton X-100 and analysing the resultant solution by appropriate assay method for the drug.
Where,
Entrapment efficiency (EF) = (Amount entrapped / total amount) x 100
Vesicle diameter
Niosomes, similar to liposomes, assume spherical shape and so their diameter can be determined using light microscopy, photon correlation microscopy and freeze fracture electron microscopy. Freeze thawing (keeping vesicles suspension at –20°C for 24 hrs and then heating to ambient temperature) of niosomes increases the vesicle diameter, which might be attributed to fusion of vesicles during the cycle.
In-vitro release
A method of in-vitro release rate study includes the use of dialysis tubing. A dialysis sac is washed and soaked in distilled water. The vesicle suspension is pipetted into a bag made up of the tubing and sealed. The bag containing the vesicles is placed in 200 ml of buffer solution in a 250 ml beaker with constant shaking at 25°C or 37°C. At various time intervals, the buffer is analyzed for the drug content by an appropriate assay method.
Separation of unentrapped drug
The removal of unentrapped solute from the vesicles can be accomplished by various techniques, which include: -
Dialysis: The aqueous niosomal dispersion is dialyzed in a dialysis tubing against phosphate buffer or normal saline or glucose solution.

Fig.5 Dialysis machine
Gel Filteration: The unentrapped drug is removed by gel filtration of niosomal dispersion through a Sephadex-G-50 column and elution with phosphate buffered saline or normal saline.42

Fig.6 Gel Filteration
Centrifugation: The niosomal suspension is centrifuged and the supernatant is separated. The pellet is washed and then resuspended to obtain a niosomal suspension free from unentrapped drug.43
Bilayer formation
Assembly of non ionic surfactants to form a bilayer vesicles by an X-cross formation under light polarization microscopy.
Membrane rigidity
Membrane rigidity can be measured by means of mobility of fluorescence probe as afunction of temperature.
Number of lamellae
This is determined by using nuclear magnetic resonance (NMR) spectroscopy, small angle Xray scattering and electron microscopy.
pH Measurement
The pH of niosomes was measured by a pH meter. The pH measurement was performed at 25°C.
Zeta Potential Measurement
Zeta potential of suitably diluted niosome dispersion was determined using zeta potential analyzer based on electrophoretic light scattering and laser Doppler velocimetry method. The temperature was set at 25°C. Charge on vesicles and their mean zeta potential values with standard deviation of 5 measurements were obtained directly from the measurement.44
STABILITY OF NIOSOMES
The factors affecting the stability of niosomes can be classified into the three categories-
• Physical stability
• Chemical stability
• Stability in biological fluids.
Physical stability
The niosomes can change their physical characteristics in several ways.
a. The particle size may change because of aggregate formation and fusion.
a. Occurrence of phase separation of bilayer components, upon storage.
b. Leakage of encapsulated material from niosomes.
The changes in particle upon storage of phosphatidyl choline containing niosomes over pharmaceutically relevant time intervals can be minimized by the selection of proper charge inducing agents. Mostly, negatively charged phospholipids (phosphatidyl glycerol) are used to stabilize the niosomes.
The phase separation may occur when the bilayer composition changes due to chemical degradation reactions or when the bilayer goes through temperature cycles. Sometimes, phase separation occurs in vivo, when bilayer components are selectively drawn from the bilayer plasma components. If this effect is undesired, thecomponents that form more rigid bilayers are preferred. In other cases, one might wish to deliberately destabilize the niosomes in vivo so that a rapid release of the encapsulated drug is induced. An example of plasma destabilized niosomes is niosomes composed of phosphatidyl ethanolamine and oleic acid. The permeability of bilayers is highly dependent on the physico-chemical properties of the bilayer, drug and the temperature. Three categories of drugs can be discussed-
• Highly hydrophilic, non-bilayer interacting drugs.
• Drugs with some lipophilicity.
• Strongly lipophilic drugs.
In category first, the presence of cholesterol in the bilayer of the egg phosphatidyl choline niosomes dramatically reduces the permeability. For gel state bilayers, permeability is low with or without cholesterol. It is clear that if in vivo performance allows ‘gel state’ bilayers to be used, the shelf life of the niosomes in aqueous media with the proper pH might easily meet industrial demands. In the second category, the drug tends to be difficult to keep entrapped over periods of months as long as outside sink conditions prevail. In the third category, strongly lipophilic drugs have high affinity for the bilayer and therefore these drugs stay there over a long period of time, independently of the state of the bilayer.
As the final remark, the presence of hydrolysis or oxidation reduction products can affect bilayer properties. Although, lysophosphatidyl choline is known to be a lipid bilayer solubilizer, the solubilizing effect of lysophosphatidyl choline in degrading niosomes is neutralized by the simultaneous appearance of fatty acids in the bilayer .
Niosomes stored in freeze dried form: The niosomes stored in freeze dried form is preferred for proper in vivo performance of niosomes with long term stability. To maintain the particle size distribution after freeze drying-rehydration cycle, a cryoprotectant needs to be added. Different types of cryoprotectants and their possible mechanisms of action have been discussed for niosome stabilization. Usually, sugars are used as cryoprotectant, although other type of excipients also have been reported to exert cryoprotective effects. A number of effects contribute to the cryoprotective action.
1) The formation of amorphous glass structures during the freeze drying process may avoid mechanical damage inflected by ice crystals. It is recommended to store these cakes below the glass transition temperature.
2) The sugars may interact with the polar head groups of the phospholipids and stabilize the membranes when the bilayer stabilizing water is removed by sublimation.
Chemical stability
The stability of niosomes depends on the chemical stability of the lipid components and the bilayer components of niosomes, designed for carrying a drug or phospholipids. Usually, hydrolysis and peroxidation are the two degradation process which occurs with phospholipids. The analytical technique to monitor hydrolysis and oxidation reactions are reviewed as under:
Lipid hydrolysis: Grit et al., in 1989 and 1993 have described different variables that influence the hydrolysis reactions of phosphatidyl choline, the major phospholipids, in the most niosomal preparations and the charge inducing phospholipids phosphatidyl glycerol. Apart from pH, other experimental conditions like temperature, ionic strength, buffer species, and ultra sonication were reported to influence hydrolysis reactions. Many investigators choose the formation of lysophosphatidyl choline as a standard measure for the chemical stability to phospholipids. Since, the presence of lysophosphatidyl choline in lipid bilayer greatly enhances the permeability of niosomes, the most important method for minimizing this problem is the proper sourcing of the phospholipid to be used. They should be essentially free from any lyso-phosphatidyl choline to start with and free of any lipases. Lipid peroxidation: Most of the phospholipid niosomal dispersions contain unsaturated acyl chains as a part of their molecular structures. These chains are vulnerable to oxidative degradation (lipid peroxidation). The peroxidation can occur during preparation, storage or actual use. Peroxidation of phospholipids produces the formation of cyclic peroxides and hydro peroxides. Peroxidation of the phospholipids may be minimized by a number of ways such as:
- Minimum use of unsaturated phospholipids.
- Use of nitrogen or argon to minimize exposure to oxygen.
- Use of light resistant container
- Removal of heavy metals (EDTA)
- Use of antioxidants such as alpha-tocopheorl or BHT.
It was reported that niosome of different lipid composition could be steam sterilized without substantial hydrolytic or oxidative degradation.
Stability in biological fluids
The inability of niosomes to retain entrapped substances when incubated in blood or plasma has been known for a decade. The instability of niosomes in plasma appears to be the result of transfer of bilayer lipids to albumin and high density lipoproteins. Both lecithin and cholesterol also exchanges with the membrane of red blood corpuscle. Niosomes are most susceptible to high density lipoprotein attack at their gel to liquid crystalline phase transition temperature. The susceptibility of niosomal phospholipids to lipoprotein and phospholipase attack is strongly dependent on niosome size and type. Generally, multilamellar vesicles are most stable whereas small lamellar vesicles are least stable. The bile salts also destabilize the bilayer membrane structure, thereby, leading to release of the entrapped material.
LIMITATION
Physical instability in niosomal dispersion during storage occurs due to vesicles aggregations, fusion and leaking. This may leads to hydrolysis of encapsulated drugs which affects the shelf life of the dispersion.
FACTORS AFFECTING VESICLES SIZE, ENTRAPMENT EFFICIENCY AND RELEASE CHARACTERISTICS
Drug
Entrapment of drug in niosomes increases vesicle size, probably by interaction of solute with surfactant head groups, increasing the charge and mutual repulsion of the surfactant bilayers, thereby increasing vesicle size. In polyoxyethylene glycol (PEG) coated vesicles, some drug is entrapped in the long PEG chains, thus reducing the tendency to increase the size. The hydrophilic lipophilic balance of the drug affects degree of entrapment. The aggregation of vesicles is prevented due to the charge development on bilayer. The encapsulation of the amphipathic drug doxorubicin has been shown to alter the electrophoretic mobility of hexadecyl diglycerol ether (C16G2) niosomes in a pH dependent manner, indicating that the amphipathic drug is incorporated in the vesicle membrane.45
Table 2-Effect of the nature of drug on the formation of niosome
|
Nature of the drug |
Leakage from the vesicle |
Stability |
Other properties |
|
Hydrophobic drug |
Decreased |
Increased |
Improved transdermal delivery |
|
Hydrophilic drug |
Increased |
Decreased |
- |
|
Amphiphilic drug |
Decreased |
- |
Increased encapsulation, altered electrophoretic mobility |
|
Macromolecules |
Decreased |
Increased |
- |
Amount and type of surfactant
The mean size of niosomes increases proportionally with increase in the HLB of surfactants like Span 85 (HLB 1.8) to Span 20 (HLB 8.6) because the surface free energy decreases with an increase in hydrophobicity of surfactant.
The bilayers of the vesicles are either in the so-called liquid state or in gel state, depending on the temperature, the type of lipid or surfactant and the presence of other components such as cholesterol. In the gel state, alkyl chains are present in a well-ordered structure, and in the liquid state, the structure of the bilayers is more disordered. The surfactants and lipids are characterized by the gel-liquid phase transition temperature (TC). Phase transition temperature (TC) of surfactant also effects entrapment efficiency i.e. Span 60 having higher TC, provides better entrapment.
A surfactant used for preparation of niosomes must have a hydrophilic head and hydrophobic tail. The hydrophobic tail may consist of one or two alkyl or perfluoroalkyl groups or in some cases a single steroidal group. The ether type surfactants with single chain alkyl as hydrophobic tail is more toxic than corresponding dialkylether chain. The ester type surfactants are chemically less stable than ether type surfactants and the former is less toxic than the latter due to ester-linked surfactant degraded by esterases to triglycerides and fatty acid in vivo. The surfactants with alkyl chain length from C12-C18 are suitable for preparation of niosome. Surfactants such as C16EO5 (poly-oxy ethylene cetyl ether) or C18EO5 (polyoxyethylenesteryl ether) are used for preparation of polyhedral vesicles. Span series surfactants having HLB number of between 4 and 8 can form vesicles.
Type of the surfactants influences encapsulation efficiency, toxicity, and stability of niosomes. The hydrophilic- lipophilic balance (HLB) is a good indicator of the vesicle forming ability of any surfactant. Uchegbu et al reported that the sorbitan monostearate (Span) surfactants with HLB values between 4 and 8 were found to be compatible with vesicle formation. Polyglycerol monoalkyl ethers and polyoxylate analogues are the most widely used single-chain surfactants. However, it must be noted that they possess less encapsulation efficiency in the presence of cholesterol. Etheric surfactants have also been used to form niosomes. These types of surfactants are composed of single-chain, monoalkyl or dialkyl chain. The latest ones are similar to phospholipids and possess higher encapsulation efficiency. Esther type amphyphilic surfactants are also used for niosome formulation. They are degraded by estherases, triglycerides and fatty acids. Although these types of surfactants are less stable than ether type ones, they possess less toxicity. Furthermore, glucosides of myristil, cethyl and stearyl alcohols form niosomes.
Cholesterol content and charge
Inclusion of cholesterol in niosomes increases its hydrodynamic diameter and entrapment efficiency. In general, the action of cholesterol is two folds; on one hand, cholesterol increases the chain order of liquid-state bilayers and on the other, cholesterol decreases the chain order of gel state bilayers. At a high cholesterol concentration, the gel state is transformed to a liquid-ordered phase. An increase in cholesterol content of the bilayers resulted in a decrease in the release rate of encapsulated material and therefore an increase of the rigidity of the bilayers obtained. Presence of charge tends to increase the interlamellar distance between successive bilayers in multilamellar vesicle structure and leads to greater overall entrapped volume.45
Methods of preparation
Methods of preparation of niosomes such as hand shaking, ether injection and sonication have been reviewed by. Hand shaking method forms vesicles with greater diameter (0.35-13nm)compared to the ether injection method (50-1000nm).
Small sized niosomes can be produced by Reverse Phase Evaporation (REV) method. Microfluidization method gives greater uniformity and small size vesicles. Niosomes obtained by trans membrane pH gradient method showed greater entrapment efficiency and better retention of drug.
Resistance to osmotic stress
Addition of a hypertonic salt solution to a suspension of niosomes brings about reduction in diameter. In hypotonic salt solution, there is initial slow release with slight swelling of vesicles probably due to inhibition of eluting fluid from vesicles, followed by faster release, which may be due to mechanical loosening of vesicles structure under osmotic stress.
Membrane composition
The stable niosomes can be prepared with addition of different additives along with surfactants and drugs. Niosomes formed have a number of morphologies and their permeability and stability properties can be altered by manipulating membrane characteristics by different additives. In case of polyhedral niosomes formed from C16G2, the shape of these polyhedral niosome remains unaffected by adding low amount of solulan C24 (cholesteryl poly-24-oxyethylene ether), which prevent saggregation due to development of steric hindrance. In contrast spherical niosomes are formed by C16G2: cholesterol: solulan (49:49:2) 2000).The mean size of niosomes is influenced by membrane composition such as polyhedral niosomes formed by C16G2: solulan C24 in ratio(91:9) having bigger size (8.0 ± 0.03mm) than spherical/tubular niosomes formed by C16G2:cholesterol:solulan C24 in ratio (49:49:2) Addition of cholesterol molecule to niosomal system provides rigidity to the membrane and reduces the leakage of drug from noisome.
Temperature of hydration-
Hydration temperature influences the shape and size of the noisome. For ideal condition it should be above the gel to liquid phase transition temperature of system. Temperature change of niosomal system affects assembly of surfactants into vesicles and also induces vesicle shape transformation. Arunothayanun et al. reported that a polyhedral vesicle formed by C16G2: solulan C24 (91:9) at 25°C which on heating transformed into spherical vesicle at 48°C, but on cooling from 55°C, the vesicle produced a cluster of smaller spherical niosomes at 49°C before changing to the polyhedral structures at 35°C. In contrast vesicle formed by C16G2: cholesterol: solulan C24(49:49:2) shows no shape transformation on heating or cooling Along with the above mentioned factors, volume of hydration medium and time of hydration of niosomes are also critical factors. Improper selection of these factors may results in formation of fragile niosomes or creation of drug leakage problems.
Surfactant/Lipid and Surfactant/Water Ratios
Other important parameters are the level of surfactant/lipid and the surfactant/water ratio. The surfactant/lipid ratio is generally 10–30 mM (1–2.5% w/w). If the level of surfactant/lipid is too high, increasing the surfactant/lipid level increases the total amount of drug encapsulated. Change in the surfactant/water ratio during the hydration process may affect the system's microstructure and thus, the system's properties.
Other Additives
As is the case with liposomes, charged phospholipids such as dicethylphosphate (DCP) and stearyl amine (SA) have been used to produce Charge in niosome formulations. The former molecule provides negative charge to vesicles whereas the later one is used in the preparation of positively charged (cationic) niosomes.
APPLICATIONS OF NIOSOMES
Niosomal drug delivery is potentially applicable to many pharmacological agents for their action against various diseases. Some of their therapeutic applications are discussed below.
1) Targeting of bioactive agents
a) To reticulo-endothelial system (RES)
The cells of RES preferentially take up the vesicles. The uptake of niosomes by the cells is also by circulating serum factors known as opsonins, which mark them for clearance. Such localized drug accumulation has, however, been exploited in treatment of animal tumors known to metastasize to the liver and spleen and in parasitic infestation of liver.
b) To organs other than RES
It has been suggested that carrier system can be directed to specific sites in the body by use of antibodies. Immunoglobulins seem to bind quite readily to the lipid surface, thus offering a convenient means for targeting of drug carrier. Many cells possess the intrinsic ability to recognize and bind particular carbohydrate determinants and this can be exploited to direct carriers system to particular cells.46
2) Neoplasia
Doxorubicin, the anthracyclic antibiotic with broad spectrum anti tumor activity, shows a dose dependant irreversible cardio toxic effect. Niosomal delivery of this drug to mice bearing S-180 tumor increased their life span and decreased the rate of proliferation of sarcoma. Niosomal entrapment increased the half-life of the drug, prolonged its circulation and altered its metabolism. Intravenous administration of methotrexate entrapped in niosomes to S-180 tumor bearing mice resulted in total regression of tumor and also higher plasma level and slower elimination. Niosomes containing anti-cancer drugs, if suitably designed, will be expected to accumulate within tumors in a similar manner to liposomes.
3) Leishmaniasis
Niosomes can be used for targeting of drug in the treatment of diseases in which the infecting organism resides in the organ of reticulo-endothelial system. Leishmaniasis is such a disease in which parasite invades cells of liver and spleen. The commonly prescribed drugs are antimonials, which are related to arsenic, and at high concentration they damage the heart, liver and kidney. The study of antimony distribution in mice, performed by Hunter et al showed high liver level after intravenous administration of the carriers forms of the drug.
4) Delivery of peptide drugs
Yoshida et al investigated oral delivery of 9-desglycinamide, 8-arginine vasopressin entrapped in niosomes in an in-vitro intestinal loop model and reported that stability of peptide increased significantly.
5) Immunological application of niosomes
Niosomes have been used for studying the nature of the immune response provoked by antigens. Brewer and Alexander have reported niosomes as potent adjuvant in terms of immunological selectivity, low toxicity and greater stability.
6) Niosomes as carriers for Hemoglobin
Niosomes can be used as a carrier for hemoglobin. Niosomal suspension shows a visible spectrum superimposable onto that of free hemoglobin. Vesicles are permeable to oxygen and hemoglobin dissociation curve can be modified similarly to non-encapsulated hemoglobin.
7) Transdermal delivery of drugs by niosomes
Slow penetration of drug through skin is the major drawback of transdermal route of delivery. An increase in the penetration rate has been achieved by transdermal delivery of drug incorporated in niosomes. Jayraman et al has studied the topical delivery of erythromycin from various formulations including niosomes or hairless mouse. From the studies, and confocal microscopy, it was seen that non-ionic vesicles could be formulated to target pilosebaceous glands.
8) Other Applications
a) Sustained Release
Sustained release action of niosomes can be applied to drugs with low therapeutic index and low water solubility since those could be maintained in the circulation via niosomal encapsulation.
b) Localized Drug Action
Drug delivery through niosomes is one of the approaches to achieve localized drug action, since their size and low penetrability through epithelium and connective tissue keeps the drug localized at the site of administration.
Localized drug action results in enhancement of efficacy of potency of the drug and at the same time reduces its systemic toxic effects e.g. Antimonials encapsulated within niosomes are taken up by mononuclear cells resulting in localization of drug, increase in potency and hence decrease both in dose and toxicity.47
Table-3 Therapeutic Application of Niosomes-
|
Application |
Drugs Encapsulated in Niosomes |
Mechanism of Action |
|
Anticancer |
Doxorubicin HCl |
Destroy Daltons ascetic Lymphoma cell |
|
Methotrexate |
Increase AUC ,than the plain drug |
|
|
Bleomycin |
Less accumulation of drug |
|
|
Vincristine |
Higher tumoridal efficacy in s-180 |
|
|
Antiinfective Agent |
Sodium Stibogluconate |
Increase level of antimony for treatment of Visceral leshminiasis |
|
Antiinflammatory Agent |
Diclofenac |
Increases inflammatory Action than that of PlainDrug |
|
Nimesulide |
||
|
Flurbiprofen |
||
|
Opthalmic Drug Delivery |
Acetazolamide Gentamycin |
Reduce intraocular Pressure |
|
Transdermal Drug Delivery |
Keterolac with Span 60 |
Increases Bioavailability and Therapeutic effect |
|
Niosomal in Oral Drug Delivery |
Insulin prepared with Niosomes |
Increases gastro intestinal tract absorption |
|
Brain Targeted Delivery Sytem for the Vasoactive Instestinal peptide |
Radio labeled I 125VIP loaded glucose bearing niosomes |
Higher VIP brain uptake |
Table-4 Drug Used In Niosomal Delivery With Different Routes Of Administrarion
|
Routes of administration |
Examples of drug |
|
Intravenous route |
Doxorubicin, Methotrexate, Sodium stibogluconate, Iopromide, Vincristin, Diclofenac sodium, Flurbiprofen, Centchroman, Indomethacin, Colchicine, Rifampicin, Tretinoin, Transferrin and glucose ligand, Zidovudin, Insulin, Cisplatin, Amarogentin , 5-fluorouracil, Daunorubicin, Amphotericin B, Camptothecin, Adriamycin, Cytarabine hydrochloride |
|
Peroral route |
DNA vaccines, Proteins, Peptides, Ergot alkaloid, Ciprofloxacin, Norfloxacin,Insulin |
|
Transdermal route |
Flurbiprofen, Piroxicam, Estradiol, Levonorgestrol, Nimesulide, Dithranol, Ketoconazole, Enoxacin, Ketorolac |
|
Ocular route |
Timolol maleate, Cyclopentolate |
|
Nasal route |
Sumatriptan, Influenza viral vaccine |
|
Inhalation |
All trans retinoic acid |
MARKETED PRODUCT
- Lancome
Niosomal Preparation in the Market is Lancôme. Lancôme has come out with a variety of antiageing products which are based on noisome formulations.

Lancome antiageing formulation
- Meloxicam
Meloxicam is a non-steroidal anti-inflammatory agent (NSAIDs), has been widely used in thetreatment of rheumatoid arthritis, osteoarthritis. These vesicular structures acts as carriers for drugs and helps to overcome the barrier properties of the skin. The present study involves the preparation and characterization of meloxicam entrapped niosomes and finding the drug carrier qualities of the niosomes.48

Meloxicam product
Table-5 Agents Encapsulated In Niosomes And Results Obtained
|
Drug |
Result |
|
Antipyrine |
Niosomal drug extended the half life of the drug and small vesicles significantly altered the metabolic profiles of the drug |
|
Bovine Serum Albumin |
Niosomes elicit a stronger immune response and improved immunological selectivity. Show lower toxicity and more stability than conventional adjuvant. |
|
DGAVP |
Facilitated oral delivery of the peptide drug. Improved the stability of the peptide to gastric enzymes |
|
Diclofenac Sodium
|
Higher Anti-inflammatory activity of the niosomal drug when administered intraperitoneally and transdermally. |
|
Doxorubicin
|
Niosomal drug had a prolonged half life, reduced cardiotoxicity and enhanced cytotoxic activity. |
|
Estradiol |
In vitro transdermal delivery of niosomal drug resulted in higher diffusion rate of the drug through stratum corneum. |
|
Flurbiprofen & Piroxicam
|
Bio-availability improved both in case of oral and transdermal pre-aeration. Bioavailability improved in case of oral penetration only. |
|
Hemoglobin
|
Hemoglobin retained its oxygen carrying capacity when encapsulated in niosomes. |
|
Humectants Sunscreens & Tanning Agents |
Enhanced delivery of agents into the skin. Used as skin penetration enhancers in cosmetics. |
ADVANTAGES OF NIOSOMES
The application of vesicular systems in cosmetics and for therapeutic purpose may offer several advantages: -
1. The vesicle suspension is water–based vehicle. This offers high patient compliance in comparison with oily dosage forms.
2. They possess an infrastructure consisting of hydrophilic, amphiphilic and lipophilic moieties together and as a result can accommodate drug molecules with a wide range of solubilities.
3. The characteristics of the vesicle formulation are variable and controllable. Altering vesicle composition, size, lamellarity, tapped volume, surface charge and concentration can control the vesicle characteristics.
4. The vesicles may act as a depot, releasing the drug in a controlled manner.
5. They are osmotically active and stable, as well as they increase the stability of entrapped drug.
6. Handling and storage of surfactants requires no special conditions.
7. They improve oral bioavailability of poorly absorbed drugs and enhance skin penetration of drugs.
8. They can be made to reach the site of action by oral, parenteral as well as topical routes.
9. The surfactants are biodegradable, biocompatible and non-immunogenic.
10. They improve the therapeutic performance of the drug molecules by delayed clearance from the circulation, protecting the drug from biological environment and restricting effects to target cells.
DISADVANTAGES OF NIOSOMES
1. Physical instability
2. Aggregation
3. Fusion
4. Leaking of entrapped drug
5. Hydrolysis of encapsulated drugs which limiting the shelf life of the dispersion.49
CONCLUSION
From the above article it is concluded that; Niosomes are one of the best targeted carriers. The self-assembly of non-ionic surfactants into vesicles was first reported in the 70s by researchers in the cosmetic industry. Niosomes are efficient carriers for controlled drug delivery, to entrap hydrophilic drugs in the larger interior aqueous layer, whereas, lipophilic drugs in the outer lipid bilayer. Since, the niosomes are biodegradable and non toxic and hence, a good carrier for targeting of therapeutic agents at the site of interest with reduced systemic toxicity. They represent a structure similar to liposome and hence they can represent alternative vesicular systems with respect to liposomes, due to the niosome ability to encapsulate different type of drugs within their multienvironmental structure. Various types of drug deliveries can be possible using niosomes like targeting, ophthalmic, topical, parentral, peroral vaccine etc. Drug efficacy depends on the drug loaded into the vehicle, temperature, drug solubility, pH, release characteristics, additives and most significantly, the vehicle morphology. In parentral applications higher concentration can readily be achieved with the use of harsh solvents. The surface area in contact affects the performance of the drug delivered at the location. More researches have to be made in this field to come out with all the potential in this novel drug delivery system.
REFERENCES
- Jose Juan Escobar-Chavez, “The Use of Sonophoresis in the Administration of Drugs Throughout the Skin” Journal Pharmaceutical Science, 2009; Page No. 88-115.
- M Bharkatiya, RK Nema, “Skin penetration enhancement techniques” Journal Young Pharmacist, 2009; Page No. 110-115.
- P. Loan Honeywell-Nguyen, Joke A. Bouwstra “Vesicles as a tool for transdermal and dermal delivery, Drug discovery today: Technologies, Drug delivery formulation and nanotechnology” 2005; Page No. 67-74..
- Hans Schreier and Joke Bouwstra, “Liposomes and niosomes as topical drug carriers: dermal and transdermal drug delivery” Journal Controlled Release, 1994; Page No. 1-15.
- Chengjiu Hu, David G. Rhodes, “Proniosomes: A Novel Drug Carrier Preparation”, International Journal of Pharmaceutics 1999; Page No. 23-35.
- Gilbert S, Christopher T. “Modern Pharmaceutics”, Fourth Edition, Marcel Dekker Inc.2002
- Andre Barel, Barel Paye Maib, Marc Paye Howard I. Maibach, “Handbook of Cosmetic Science and Technology”, Marcel Dekker Inc.2001; Page No. 776-780.
- V. P. Torchilin, Volkmar Weissig., “Liposomes: a practical approach” second edition, Oxford university press, 2003.
- Azmin, M.N., Florence, A.T., Handajani-vila, R.M., Stuart, J.F., Vanlerberghe, G., and Wittaker, J.S. (1985). “The effect of nonionic surfactant vesicle entrapment on the absorption and distribution”, Journal Pharm. Pharmacol. 37: Page No. 237-242. Goodman and Gilman’s, Alfred Goodman Gilman, (1990), “Route of administration and therapeutic uses., The Pharmacological Basis of Therapeutics”, Pergamom press, New york, eighth edition, Vol. 1, Page No. 225.
- Malhotra M, Jain NK, “Niosomes as drug carriers”, 1994; Page No. 81-86.
- Vora B, Khopade AJ, Jain NK, “Proniosome based transdermal delivery of levonorgestrel for effective contraception”, Journal Controlled Release, 1998; Page No. 149-165.
- Weindl G, Schaller M, Schafer-korting M, Korting HC, “Hyaluronic acid in the treatment and prevention of skin diseases,, Molecular, biological, pharmaceutical and clinical aspect”, Skin Pharmacol Physiol. 2004; Page No. 207-213.
- Sayo T, Sakais I, “Synergistic effect of N-acetyl glucosamine and retinoids on hydaluronan production in human keration cytes”, Skin Pharmacol Physiol. 2004; Page No. 77-83.
- Ghersetich I, Lotti T, Campanile G, Grappone C, Dini G, “Hyaluronic acid in cutaneous aging” International Journal of Dermatology 1994; Page No. 119-122
- Imokawa G., “Analysis of carbohydrate properties essential for melanogenesis in tyrosinase of cultured malignant melanoma cells by differential carbohydrate processing inhibition”, Journal Invest Dermatology 1990; Page No. 39-49.
- Imokawa G., “Analysis of initial melanogenesis including tyrosinase transfer and melanosome differentiation through interrupted melanization by glutathione” Journal Invest Dermatology. 1989; Page No. 101-107.
- Imokawa G, Mishima Y., “Importance of glycolproteins in the initiation of melanogenesisi an electron microscopie study of B-16 melanoma cells after release from inhibition of glycosylation”, Journal Invest Dermatology, 1986; Page No. 319-325.
- Imokawa G, Mishima Y., “Analysis of tyrosinases as asparagines linked oligosaccharides by concanvalin a lectin chromatography: appearance of new segment of tyrosinase in melanoma cells following interrupted melanogenesis induced by glycosylation inhibitors”, Journal Invest Dermatology, 1985; Page No. 165-168.
- Imokawa G, Mishima Y., “Functional analysis of tyrosinase isoanzymes of cultured malignant melanoma cells during the recovery period following interrupted melanogenosis induced by glycosylation inhibitors”, Journal Invest Dermatology 1984; Page No. 196-201.
- Bissett Dl, Robinson L, Li J, Miyamoto K., “Topical N-acetyl glucosamine reduces the appearance of hyper pigmented spots on human facial skin” Journal Am Acad Dermatol. 2006; AB 54.
- Kanwischer M, Kim SY, Kim JS, Bian S, Kwon KA, Kim DD., “Evaluation of the physicochemical stability and skin permeation of glucosamine sulfate”, Drug Dev Ind Pharm. 2005; Page No. 91-97.
- Theresa M.A., Drugs published by Adis international Ltd., 1998; Page No. 747-756.
- Breimer D.D. and Speiser R., “Topics in pharmaceutical Sciences”, 5 Elsevi Science Publishers, New York, USA, 1985; Page No. 291
- Buckton G., Harwood, “Interfacial phenomena in Drug Delivery and Targeting” Academic Publishers, Switzerland. 1995; Page No. 154-155.
- Don A., Van H., Joke A.B. and Hans E., “Non ionic surfactant vesicles containing estradiol for topical application”, Centre for drug research. 1997; Page No. 330-339.
- Handjani-Vila R.M., Ribier A., Rondot B. and Vanlerberghe G., “Dispersions of lamellar phases of non-ionic lipids in cosmetic products”, International Journal of cosmetic science, 1979; Page No. 303-314.
- Harisha Atluri, Banmeet S. Anand, Jignesh Patel, Ashim K. Mitra, “Mechanism of a model dipeptide transport across blood-ocular barriers following systemic administration”, 2004; Page No. 815-822.
- Pranav sharma, Kamal dua, Sharma VK, Sara U.V.S, Yadav V.P, Samad A, Sunil Prajapati, “Ophathalmic drug delivery system”, The Indian Pharmacist 2008, VII, Page No. 76.
- Baillie AJ., “The preparation and properties of niosomes-nonionic surfactant vesicles” Journal Pharm. Pharmacol, 1985; Page No. 863-868
- Hanan M. El-Laithy, Omar Shoukry, Laila G. Mahran, “Novel sugar esters proniosomes for transdermal delivery of vinpocetine: Preclinical and clinical studies”, European Journal of Pharmaceutics and Biopharmaceutics, 2011; Page No. 43-55.
- Bhavana Vora, Ajay J. Khopade, N.K. Jain, “Proniosome based transdermal delivery of levonorgesterol for effective contraception”, Journal of Controlled Release, 1998; Page No. 149-165
- Navin K. Dahiya, “Preparation and characterization technique in niosomal vesicular systems-A review”, Journal of Pharmaceutical and Biomedical Science, 2011; Page No. 1-8.
- Buckton G., Harwood, “Interfacial phenomena in Drug Delivery and Targeting”, Academic Publishers, Switzerland. 1995; Page No. 154-155.
- Rogerson A., Cummings J., Willmott N. and Florence A.T., “The distribution of doxorubicin in mice following administration in niosomes”, J Pharm Pharmacol. 1988; Page No. 337-342.
- Baillie A.J., Coombs G.H. and Dolan T.F., “Non-ionic surfactant vesicles, niosomes, as delivery system for the anti-leishmanial drug”, J. Pharm. Pharmacol. 1986; Page No. 502-505.
- Khandare J.N., Madhavi G. and Tamhankar B.M., “Niosomes as novel drug delivery system”, The Eastern Pharmacist, 1994; Page No. 61-64.
- Raja Naresh R.A., Chandrashekhar G., Pillai G.K. and Udupa N., “Antiinflammatory activity of Niosome” Indian Journal of Pharmacology, 1994; Page No. 46-48.
- Chauhan S. and Luorence M.J., “The preparation of polyoxyethylene containing non-ionic surfactant. Vesicles”, J. Pharm. Pharmacol. 1989; Page No. 6.
- Blazek-Walsh A.I. and Rhodes D.G. Pharm. Res. SEM imaging predicts quality of niosomes from maltodextrin-based proniosomes. 2001; Page No. 656-661.
- Chandraprakash K.S., Udupa N., Umadevi P. and Pillai G.K., “Pharmacokinetic evaluation of surfactant vesicles containing methotrexate in tumor bearing mice”, International Journal Pharmaceutics, 1990; R1-R3:
- ManosroiA, Wongtrakul P,M J,Sakai H, Sugawara F, Yuasa M, “Characterization of vesicles prepared with various non-ionic surfactants mixed with cholesterol” 2003; Page No. 129-38.
- P. Arunothayanun, T. Sooksawate, and A. T. Florence “Extrusion of niosomes from capillaries: approaches to a pulsed delivery device”, Journal of Control Release, 1999; Page No. 391-397.
- Chauhan S, Luorence MJ., “The preparation of polyoxyethylene containing non-ionic surfactant vesicles”, J. Pharm. Pharmacol. 1989; Page No. 4-6.
- Hu C. and Rhodes D.G., “Proniosomes: a novel drug carrier preparation”, International Journal of Pharmaceutics, 1999; Page No. 23-35.
- Goopi N. Devaraj., Prakash, S. R., Ravi Devaraj, Apte, S.S., B. Ramesh Rao., D.Rambhav., “Release studies on Niosomes containing fatty alcohols as bilayer stabilizers instead of cholesterol”, Journal of colloids and interface science, 2002; Page No. 251,360-365.
- G.H. Buckton, “Interfacial phenomena in Drug Delivery and Targeting”, Academic Publishers Switzerland, 1995; Page No. 154-155
- N.K. Jain, “Controlled & Novel Drug Delivery”, First edition, published by CBS Publishers & Distributors, New Delhi; 2004; Page No. 292
- Gregoriadis G., “Targeting of drugs: implications in medicine”, Lancet. 1981; Page No. 241-246.
- Suzuki K. and Sokan K., “The Application of Liposomes to Cosmetics” Cosmetic and Toiletries. 1990; Page No. 65-78.









