 ABOUT AUTHOR
ABOUT AUTHOR
Dr. R. S. Thakur
Former Secretary, Pharmacy Council of India.
Email : drramsthakur@gmail.com
Preamble
Ministry of Health and Family welfare, Government of India has circulated draft National Pharmacy Commission Bill, 2023 vide F. No. Z-28020/27/2023-AHS on 10th November, 2023 and invited comments of the general public/stakeholders within one month. In India there exists The National Medical Commission Act, 2019 (Act No. 30 of 2019); The National Dental Commission Act, 2023 (Act No. 21 of 2023); and The National Nursing and Midwifery Commission Act, 2023 (Act No. 26 of 2023). The proposed National Pharmacy Commission Bill, 2023 is fourth in the series that is aimed at setting up the National Pharmacy Commission and repeal the Pharmacy Act, 1948. The Pharmacy Act, 1948 was drafted on the basis of recommendations of The Drugs Enquiry Committee report, 1929-30.Thus it needed replacement by new law based on current circumstances and scenario of pharmacy practice prevailing in the advanced countries of the world to ensure world class pharmaceutical care. Thus the initiative from the Government of India is commendable and definitely better regulation and control of pharmacy education and profession of pharmacy is need of the hour. Sooner the proposed Bill is passed by the Parliament and receives assent of the President to become Act is better in the interest of the profession of Pharmacy and public at large.
Introduction
Pharmacists of India may recall that on 28th March, 2023 Government of India, Ministry of Health and Family Welfare had constituted an Expert Committee of 16 persons including the Chairman, vide Office Order F No.Z-28020/27/2023-AHS, FTS No. 8205489. The terms of Reference of the Expert committee were:-
(a) To review Pharmacy Education;
(b) To review the Pharmacy Act, 1948; and
(c) To make recommendations for restructuring of the Pharmacy Council of India.
Reference Id: PHARMATUTOR-ART-3010
The committee was requested to give its recommendations within one month of date of issue of the order.
On 28th April, 2023 the author published an article in Pharmatutor [1] highlighting the importance of the terms of reference of the Expert Committee and how their report can help the government in bring new legislation. The author minced no words to thank the Government for the bold step and suggested that now it is the turn of the enlightened pharmacists’ fraternity to put forth their suggestions on all the three terms of reference, before the expert committee along with justification and scheme of implementation. It was also emphasized that the suggestions should cover educational reforms that are imminent; practice scenario and enlargement of scope of practice to achieve global standards; and efficient monitoring system to assure quality of education and servicesboth.
Now the circulation of the draft Bill signifies Government’s honest and committed approach as well as another bold step towards better legislation for the regulation of profession of pharmacyin India. In order to support or criticize the draft it would be necessary to compare the existing Acts for Medical, Dental and Nursing and Midwifery [2-4] with the proposed Bill for Pharmacy. Unless the schemes of all the existing Acts are compared with the proposed Bill any deficiency or defect in the proposed Bill cannot be pin pointed for sure. In the above background this paper analyses the three existing Acts aforesaid along with the Bill to assess similarity and dissimilarity factor, so that remedial measures could be suggested.
Scheme of the Pharmacy Act, 1948
The Pharmacy Act, 1948 was enacted to meet three goals that are very well categorized in the Statement of Objects and Reasons of that Act. The Statement of objects and reasons as published in the Gazette of India, 1947, Pt. V, p-469 is reproduced below:
“ Statement of objects and Reasons – It is desirable that, as in most other countries, only persons who have attained a minimum standard of professional education should be permitted to practice the profession of pharmacy. It is accordingly proposed to establish a Central Council of Pharmacy, which will prescribe the minimum standards of education and approved courses of study and examinations for Pharmacists, and provincial Pharmacy Councils, which will be responsible for the maintenance of provincial register of qualified Pharmacists. It is further proposed to empower Provincial Governments to prohibit the dispensing of medicine on the prescription of a medical practitioner otherwise than by, or under the direct and personal supervision of, a registered Pharmacist.”
It is amply clear from the above that the Act is aimed at -
(i) permitting only such persons who have attained a minimum standard of professional education to practice the profession. The minimum standard were prescribed in The Education Regulations framed under Sec.10 of the Act as back as in 1953 and amended since then in 1972, 1981, 1991 and 2020 [5] and implemented under Sec.11 of the Act across the country. To meet this objective constitutionof a Central Council of Pharmacy is provided;
(ii) establishing provincial Pharmacy Councils, which will be responsible for the maintenance of provincial register of qualified Pharmacists; and
(iii) empowering Provincial Governments to prohibit the dispensing of medicine on the prescription of a medical practitioner otherwise than by or under the direct and personal supervision of a registered Pharmacist.
Unfortunately there has been complete failure on all the three fronts as illustrated below:-
(i) Although as back as in 1932 USA opted for 4 years degree course as only qualification to register as pharmacist in USA, India could not simulate that model and adopt a replica of that course and even in 2023 a two years academic and 3 months practical training diploma course after 10+2 (Science academic stream) with Physics, Chemistry and Biology or Mathematics is minimum qualification for registration as pharmacist under this Act;
(ii) Although the statement of Objects and Reasons prescribe for register of qualified pharmacists the Act was repeatedly amended to pave way for registration of unqualified persons en masse. Such dilutions in qualification for registration were done by inserting new Section 32A [6] (Special provisions for registration ofcertain persons) in 1959, and insertion of new Section 32B [7] (Special provisions for registration of displaced persons, repatriates and other persons) in 1976. These provisions were badly misused by many state Pharmacy Councils to register unqualified persons as pharmacist. Moreover, more than sixty seven thousand unqualified persons were registered by West Bengal Pharmacy Council alone during 1976 and 1978. Other State Pharmacy Councils also registered several thousand persons fraudulently. The Government of West Bengaleven constituted a Commission of Enquiry against West Bengal Pharmacy council u/s. 45(5) under the Chairmanship of Hon’ble Mr. Justice Deepak Kumar Sen of Calcutta High Court. The fraud played by West Bengal Pharmacy Council was proved before the Commission by the author through documentary evidences. At that point of time the member of the Commission appointed by the West Bengal Pharmacy Council resigned in calculated manner (Of course on legal advice of their advocate) and that Council did not appoint his substitute to make the Commission of Enquiry defunct due to vacancy; and
(iii) The failure on the third front is non-implementation of Section 42 of the Act by State Governments and State Pharmacy Councils by not implementing Section 26A in absence of any Rules made under clause (ff) of sub-section (2) of Section 46 of the Act by State Governments except Kerala Government. In pursuance of Section 46(2)(ff) of Pharmacy Act, Kerala Government made rules by inserting Chapter XXI in Kerala State Pharmacy Council Rules, 2012 prescribing qualification, powers and duties of Inspectors [8] spread over rule 157 to 159.
After very prolonged litigation vide W. P. No. 44688 of 2015 (GM-RES-PIL) Sri Ashok SwamyHeroorvs The state of Karnataka and others in the Hon’ble High Court of Karnataka, Government of Karnataka made Rules in this context vide notification No. HFW 40 IMM 2016 dated 06-06-2022
In the State of Uttar Pradesh Vijay Singh and 15 others filed W.P. No. 15356(S.S.) of 2017 in the Hon’ble High Court of Judicature at Allahabad, Lucknow Bench, Lucknow for appointment of Inspectors under Section 26A of Pharmacy Act. The matter is still pending.
The above description of the total failure of Pharmacy Act, 1948 to meet any of its set objectives definitely justifies its demolition without any further delay and hence draft Bill is a welcome step by all means.
Methodology
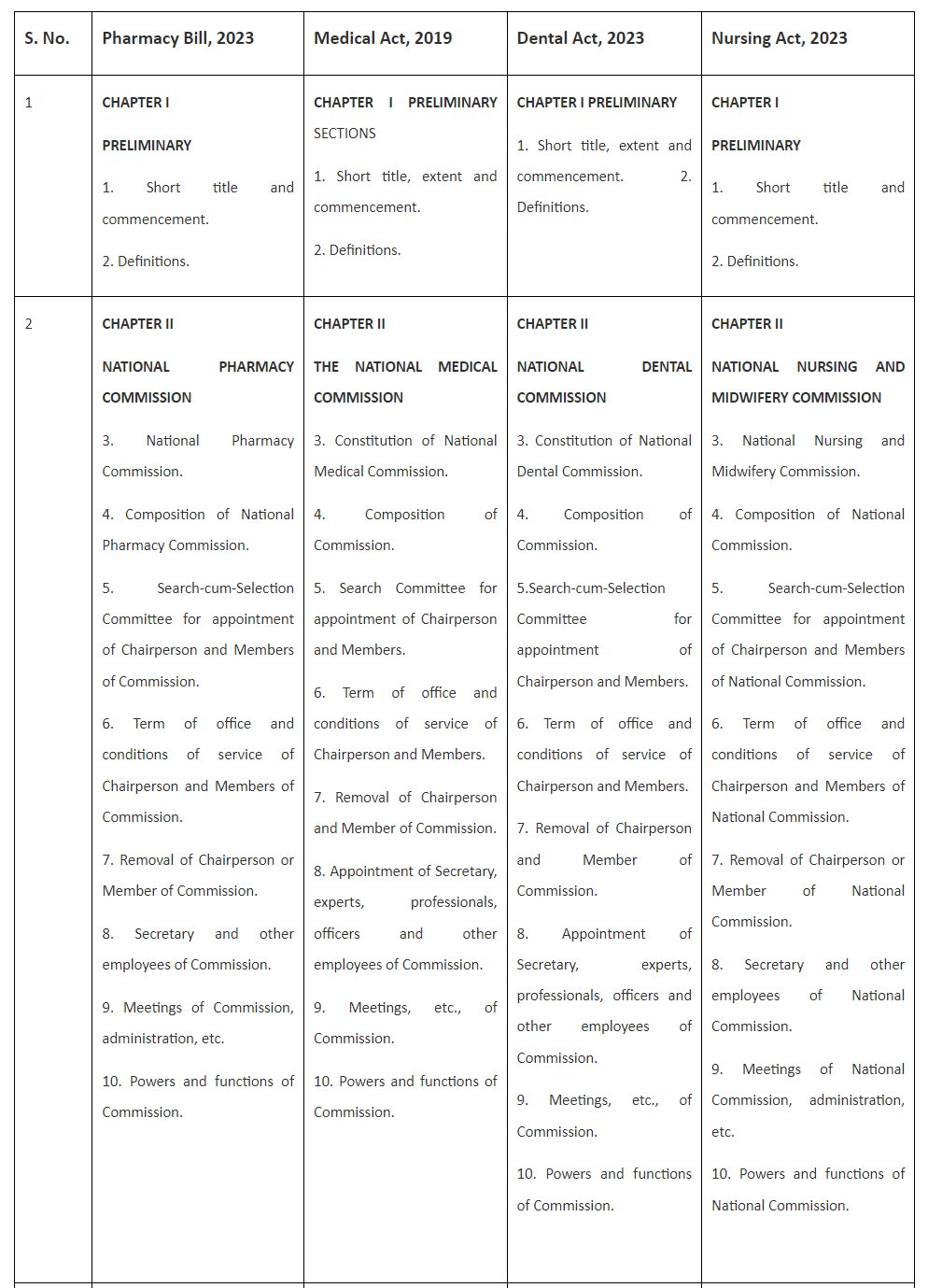
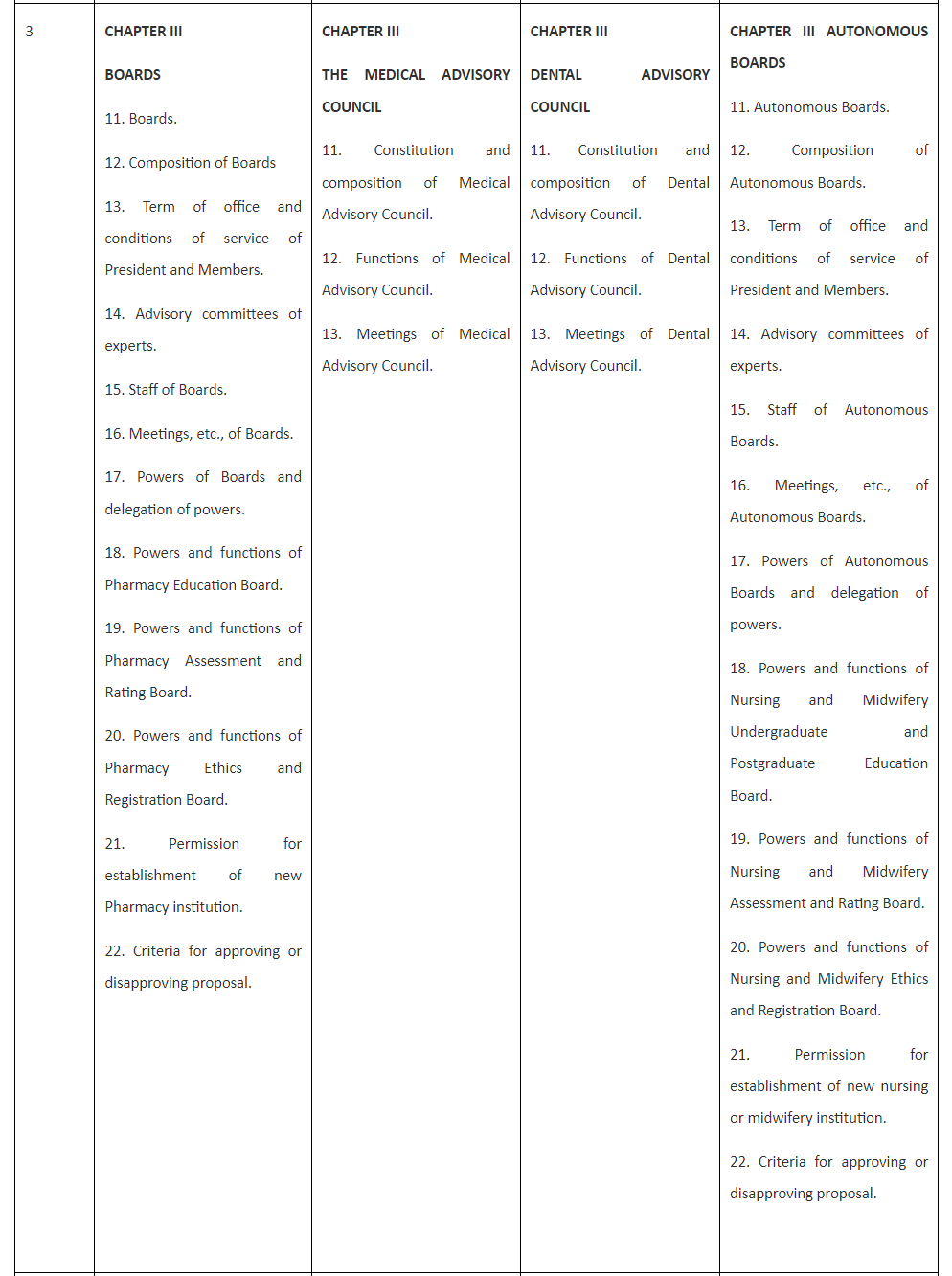
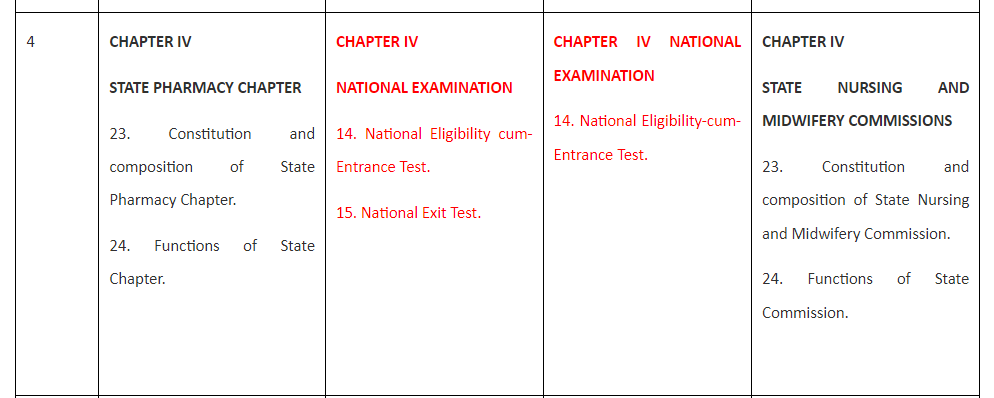
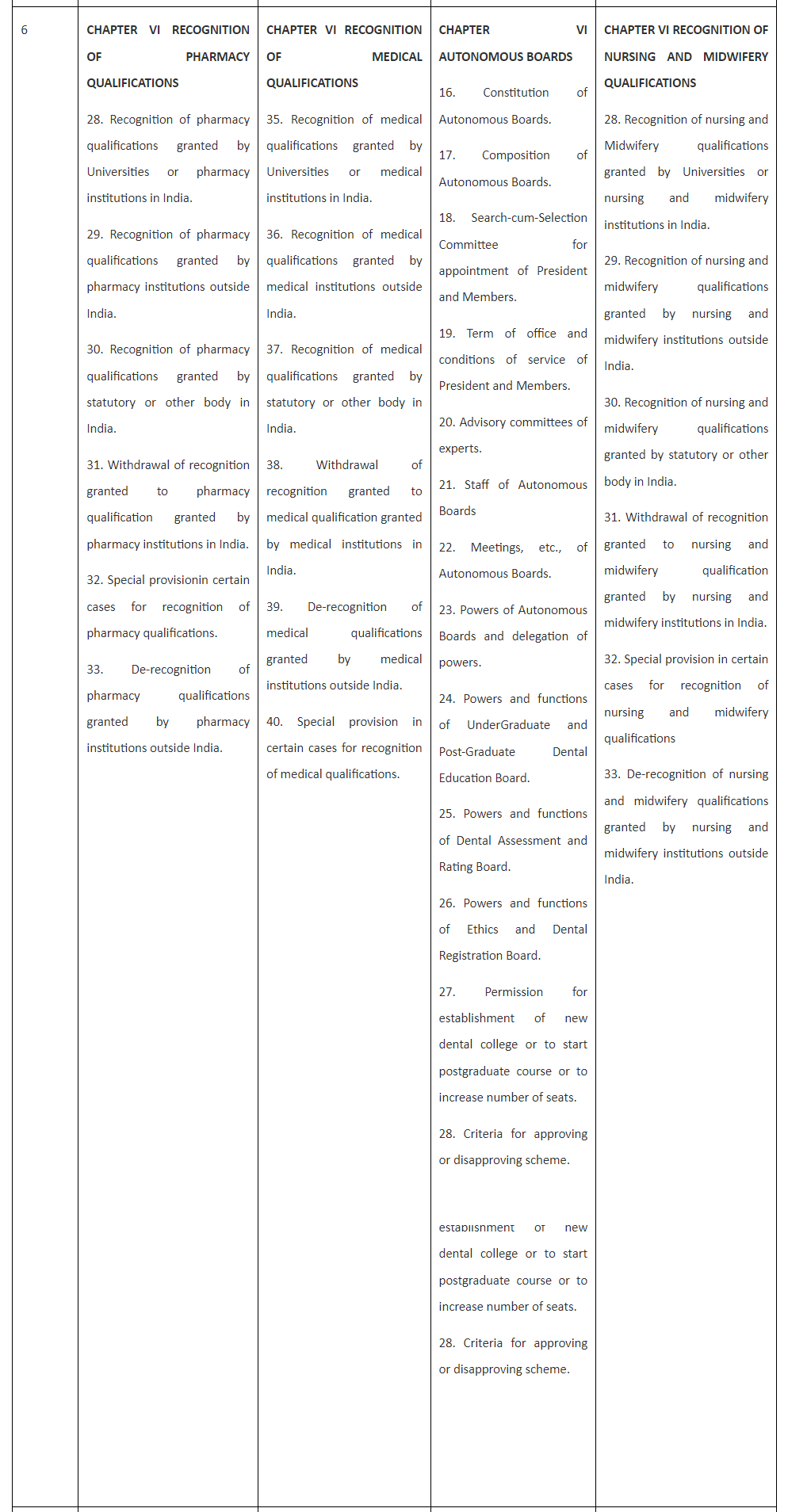
Comparison of Existing three Acts with proposed Bill for Pharmacy is tabulated and presented in Table 1 for finding similarity and dissimilarity among the legislations recently enacted.
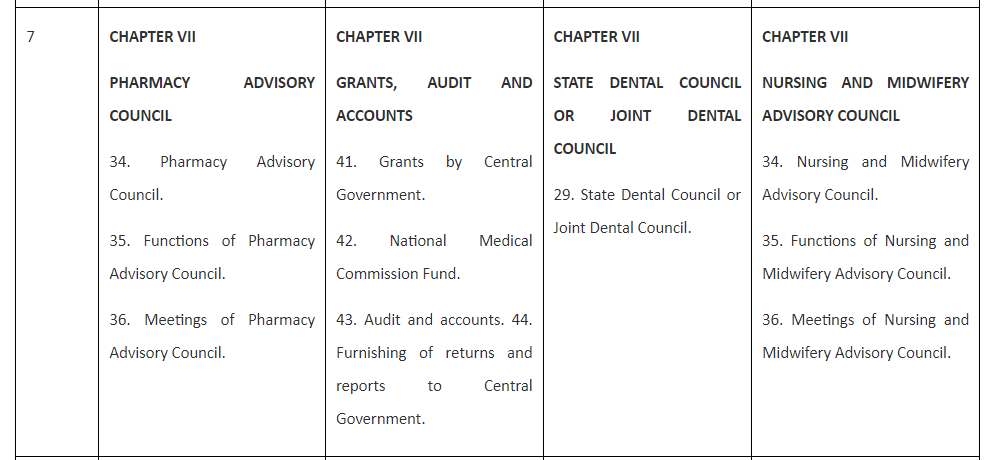
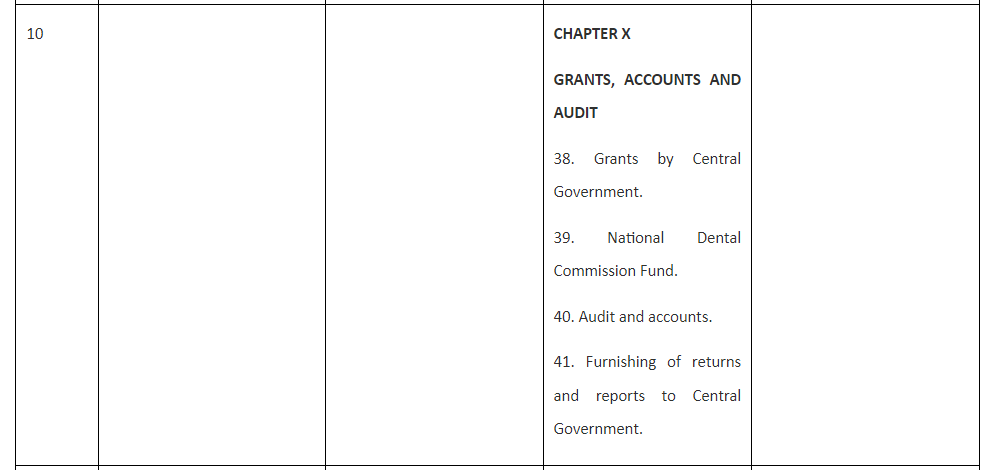
Table 1. Comparison of the draft Bill with Acts for Medical, Dental and Nursing and Midwifery.





Results
Table 1 reveals that the Bill for Pharmacy is spread over nine Chapters and 57 Sections and so is the National Nursing and Midwifery Commission Act, 2023 (26 of 2023). Chapter I and Chapter II of all the three Acts and the Bill are completely similar containing Sections 1 and 2 in the former and Sections 3 to 10 in the later.
Table 1 also reveals that Chapter III to Chapter IX of both Pharmacy Bill and Nursing and Midwifery Act are completely similar and replica of each other as they contain similar sections 11 to 23 in Chapter III; Sections 23 to 24 in Chapter IV, Sections 25 to 27 in Chapter V,Sections 28 to 33 in Chapter VI,Sections 34 to 36 in Chapter VII,Sections37 to 40 in Chapter VIII,and Sections 41 to 57 in Chapter IX.
Unlike the Bill for Pharmacy and the Act of Nursing and Midwifery, National Medical Commission Act, 2019 is having only eight Chapters but 61 Sections and The Schedule while the National Dental Commission Act, 2023 has eleven Chapters, 59 Sections and The Schedule. Thus National Medical Commission Act, 2019 has four more sections than Pharmacy Bill and the Nursing and Midwifery Act while National Dental Commission Act, 2023 has two more Sections than Pharmacy Bill and the Nursing and Midwifery Act. These dissimilarities are presented and explained hereunder:
The additional Sections in Medical and Dental Acts represent Section 14 National Eligibility cum-Entrance Test and Section 15 National Exit Test. Moreover, Section 50 and 51 of the National Medical Commission Act, 2019 represent Section 50 Joint sittings of Commission, Central Councils of Homoeopathy and Indian medicine to enhance interface between their respective systems of medicine and Section 51 State Government to promote primary healthcare in rural areas.
Discussion
The comparative analysis of the existing legislations and the Bill reveals complete similarity in all legislations except that NEET and National ExitTest are not there in the National Nursing and Midwifery Commission Act, 2023 as also in the National Pharmacy Commission Bill, 2023.
Analysis of the National Medical Commission Act, 2019 further reveal that there are two additional Sections as 50 and 51 in that Act which relate to joint sitting and coordination with Central councils of AYUSH for better health care and promotion of rural healthcare.
In view of the exhaustive and critical review of the documents referred to hereinabove it appears that the proposed Bill has been meticulously drafted to bring all the four National Commissions on par so far as functional aspects are concerned. Hopefully the new body will utilize its provisions to take profession of pharmacy to new heights and strive to ensure world class pharmacy education and pharmaceutical care in this country.
Conclusion
The demise of Pharmacy Act, 1948 is not only imminent but inevitable too. To cope with the fast advancements in pharmaceutical technology and pharmaceutical care it is essential that the outdated Pharmacy Act, 1948 be abandoned lock stock and barrel because it failed to protect the interests of qualified pharmacists and the patients as well.
References
[1]. R. S. Thakur. Expert Committee on Pharmacy Education, Law and Regulator.Pharmatutor.28thApril, 2023. https://www.pharmatutor.org/articles/ expert-committee-on-pharmacy-education-law-and-regulator
[2].The National Medical Commission Act, 2019 (Act No. 30 of 2019) [8th August, 2019.] Available at : India Code https://www.indiacode.nic.in>A2019_30 Accessed on: 25.11.2023
[3]. The Gazette of India Extraordinary Part II – Section 1, No. 24, New Delhi dated August 11, 2023, p 1-29.
[4]. The Gazette of India Extraordinary Part II – Section 1, No. 29, New Delhi dated August 14, 2023, p 1-29.
[5]. The Gazette of India Extraordinary Part III – Section 4, No. 435, New Delhi dated October 16, 2020, p 1-22.
[6]. The Gazette of India Extraordinary Part II – Section 1, No. 21, New Delhi dated August 28, 1959, p 198-199.
[7]. The Pharmacy (Amendment) Act, 1976 (70 of 1976). w.e.f. 1-9-1976.In the Pharmacy Act, 1948 (8 of 1948), Government of India, Ministry of law, Justice and Company Affairs.Printed by the Manager, Government of India Press, Shimla. 1978. Page 15.
[8]. Kerala Gazette Extraordinary, Vol. I.VII; No. 1663: 6th August 2012, Notification G. O. (P) No. 237/2012 dated 26th July, 2012. Page 41-42.










