{ DOWNLOAD AS PDF }
ABOUT AUTHOR
S. Dhivya*, DR. A. N. Rajalakshmi
Department of Pharmaceutics,
Mother Theresa Post Graduate and Research Institute of Health Sciences,
Gorimedu, Puducherry, India
*dhivyaraji310@gmail.com
ABSTRACT: Design and development of herbal nanoparticles has become a frontier research in the nanoformulation arena. Curcumin, a hydrophobic polyphenol (diferuloyl methane) is a potent phytomolecule obtained from turmeric (Curcuma longa, Family-Zingiberaceae) has a wide range of biological activities in chronic diseases and has wide therapeutic efficacy. But the clinical application of curcumin was limited due to its poor water solubility, rapid metabolism and rapid elimination which ultimately results in poor bioavailability upon oral administration. Therefore introduction of nanotechnology provides a solution towards increased bioavailability of curcumin. In this review, an overview of curcumin nanoparticles is discussed.
[adsense:336x280:8701650588]
Reference Id: PHARMATUTOR-ART-2544
|
PharmaTutor (Print-ISSN: 2394 - 6679; e-ISSN: 2347 - 7881) Volume 5, Issue 12 Received On: 02/08/2017; Accepted On: 14/08/2017; Published On: 01/12/2017 How to cite this article: Dhivya S, Rajalakshmi AN;Curcumin Nano drug delivery systems: A Review on its type and therapeutic application; PharmaTutor; 2017; 5(12); 30-39 |
INTRODUCTION:
Curcumin (also known as curcumin-I) [1], is the principle curcuminoid found in Indian curry spice turmeric (Curcuma longa, Family-Zingiberaceae). The other curcuminoids present in turmeric are demethoxycurcumin (curcumin-II) [2], bisdemethoxycurcumin (curcumin-III) [3] and recently cyclocurcumin [4] is identified. Curcumin is a potent phytomolecule with a wide range of biological activities [5]. Curcumin is a principle nutraceutical molecules along with other curcuminoids and it has been used in the food and pharmaceuticals industries[6]. Its clinical applications are limited largely due to its poor solubility and rapid metabolism, which results in poor bioavailability. Curcumin is characterized with extremely low solubility in water (11ng/ml) and significant presystemic biotransformation, mainly via glucuronide and sulfate conjugation. The maximum oral dose of 8 gm/day of curcumin does not produce any toxic effects [7].
With the ongoing development of nanotechnology, the nanoparticles have its own importance in novel drug delivery system. The emergence of nanoscience, which is the creation and utilization of materials and tools on nanometer scale, has exerted a deep influence on numerous industries and particularly the pharmaceutical industry. The application of nanoparticle formulation will deliver new approaches for enhancement of solubility, stability, bioavailability and pharmacological activity and ability to avoid physical and chemical degradation. Therefore introduction of nanotechnology in curcumin provides a solution towards increased bioavailability and therapeutic efficacy.
Today, due to technological innovations nontoxic, biocompatible, inexpensive and biodegradable nanoparticles with various colloidal dimensions are being developed to enhance the penetration ability, reduce the frequency of doses, toxicity and to improve the therapeutic efficacy [8-10].
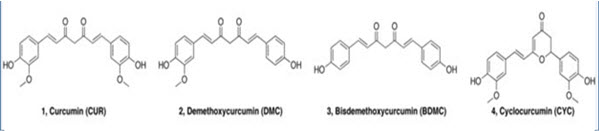
Chemical Structure of curcuminoids
Need of novel drug delivery system for curcumin
Curcumin is highly unstable in acidic pH of the stomach and degraded at alkaline pH before reaching to the blood and other constituents might be metabolized by the liver. Resulting, the optimum quantity of the curcumin may not reach the blood resulting in no/less therapeutic effect. Nanocarriers applying to curcumin will carry optimum amount of the drug to their site of action bypassing all the barriers such as acidic pH of stomach, liver metabolism and increase the prolonged circulation of the drug into the blood due to their small size. So curcumin was selected as feasible drug candidate for delivery through a nano delivery system because of the following properties
• To improve the solubility
• To enhance the bioavailability
• To reduce the dose.
• To target the site of action.
• To control the release of the drug
Types of Curcumin Nano drug delivery systems
• Liposomes
• Polymeric Nanoparticles
• Solid lipid nanoparticals
• Polymeric micelles
• Magnetic nanoparticals
• Nanogels
• Gold Nanoparticles
• Silver Nanoparticles
Liposomes: Liposomes are closed spherical vesicles consisting of a lipid bilayer that encapsulates an aqueous phase in which drugs can be entrapped [11]. With the advantages of high biocompatibility, easy preparation, chemical versatility, and simple modulation of their pharmacokinetic properties by changing the chemical composition of the bilayer components, they have been used to improve the therapeutic activity and safety of drugs for many years [12]. So, liposomes have found wide application in enhancing curcumin’s bioavailability and efficacy. In this regard, to enhance the solubility of curcumin, Rahman et al., prepared beta-cyclodextrin curcumin inclusion complexes that entrapped both native curcumin and the complexes separately into liposomes. All curcumin-containing formulations were effective in inhibiting cell proliferation in in vitro cell culture [13]. Chen et al., reported the effects of different liposomal formulations on curcumin stability in phosphate buffered saline, human blood, plasma, and culture medium. Liposomal curcumin showed a higher stability than free curcumin in phosphate buffered saline (PBS) [14].
Polymeric Nanoparticles: Due to the small size and excellent biocompatibility, polymeric nanoparticles can circulate in the bloodstream for a longer time; thus, specific therapy can be achieved [15]. The widely researched synthetic polymers include chitosan [16,17], poly(D,L-lactideco-glycolide) (PLGA)[18-20], and PEG [21] for the cucrcumin nanoparticle formation. Moreover, polymers can be combined to form copolymers, which could be a promising drug carrier for the site targeting and sustained action.
Solid Lipid Nanoparticles (SLNs): SLNs are made of natural or synthetic lipid or lipoid, such as lecithin and triglycerides, which are solid at human physiological temperature [22]. SLNs offer unique properties such as smaller size, larger surface area, interaction of phases at the interfaces, and these are attractive for their ability to improve performance of neutraceuticals, pharmaceuticals and other materials. Solid lipid nanoparticles possess a solid lipid core matrix that can solubilize Lipophilic molecules. The lipid core is stabilized by surfactants (emulsifiers). For pharmaceutical applications, all formulation excipients must have Generally Recognized as Safe (GRAS) status to achieve and maintain a solid lipid particle upon administration, the lipid nanoparticles melting point must exceed body temperature (37 °C) [23]. Kakkar et al., prepared curcumin-loaded solid lipid nanoparticles (C-SLNs) for the improvement of its oral bioavailability [24]. Dadhaniya et al., examined the adverse effects of a new solid lipid curcumin particle in rats [25].
Polymer Micelles: Micelles are lipid molecules that arrange themselves in a spherical form in aqueous solutions with a very narrow range from 10 to 100 nm in size, which makes them more stable towards dilution in biological fluids [26]. The functional properties of micelles are based on amphiphilic block copolymers, which come together to form a nanosized core/shell structure in aqueous media. Polymeric micelles can serve as transporters of water-insoluble drugs such as curcumin, which can augment the drug’s efficiency by targeting definite cells or organs; therefore, fewer drugs accumulate in healthy tissues and their toxicity reduces, and occasionally higher doses can be administered [27]. In this regard, to overcome the poor water solubility of curcumin, Liu et al., prepared curcumin loaded biodegradable self-assembled polymeric micelles for sustained release[28]. In addition, the preparation of curcumin-loaded micelles based on amphiphilic Pluronic/ polycaprolactone block copolymer was investigated by Raveendran et al., which proved to be efficient in enhancing curcumin’s aqueous solubility [29]. Thus the micellar system is efficient for solubilization, stabilization, and controlled delivery of curcumin.
Magnetic Nanoparticles: Magnetic drug targeting, in which a drug is conjugating with a magnetic material under the action of the external magnetic field. Drug-loaded magnetic nanoparticles can accumulate in target tissue areas under the action of the external magnetic field; the drug then releases from the particles in a controllable way [30]. Yallapu et al., [31] introduced magnetic drug carriers with a pluronic polymer (F127) shell for controlled delivery of curcumin. A nanosized magnetofluorescent water-dispersible Fe3O4-curcumin conjugate with chitosan or oleic acid asits outer shell and entrapped curcumin was designed by Tran et al., [32]. The Fe3O4-curcumin conjugate exhibited a high-loading cellular uptake that was distinctly observed by magnetic and fluorescent methods and was also shown to be a good candidate for a dual (optical and magnetic) imaging probe.
Nanogels: Nanogels are cross-linked three-dimensional polymer chain networks which are created through covalent linkages and can be customized to gel networks with biocompatible and degradable properties. Nanogels demonstrate excellent potential for systemic drug delivery that should have a few common features including a smaller particle size (10–200 nm), biodegradability and/or biocompatibility, prolonged half-life, high stability, higher amount of drug loading and/or entrapment, and molecules protection from immune system. Goncalves et al., [33] applied a self-assembled dextrin nanogel as curcumin delivery system by using dynamic light scattering and fluorescence measurements. Various nanogel properties can be attained by altering the chemical functional groups, cross-linking density, and surface-active and stimuli-responsive elements. Wu et al., [34] designed a class of water-dispersible hybrid nanogels for intracellular delivery of hydrophobic curcumin.
Gold nanoparticles: With the optical and electrochemical uniqueness, gold nanoparticles have proven to be a potent apparatus in nanomedicinal requests [35]. Moreover, the stability of AuNPs and their capability to combine with biomolecules are their other outstanding properties. AuNPs are studied broadly as imperative drug delivery vectors due to some of their characteristic aspects, such as low cytotoxicity, tunable surface features, and stability in in vivo conditions, and can be easily synthesized and functionalized. Rajesh et al., [36] used polyvinyl pyrrolidone (PVP) as a proven drug carrier to curcumin conjugation with AuNPs to enhance solubility of curcumin. In a study by Singh et al., [37] curcumin was bound on the surface of AuNPs in order to increase the bioavailability of it. Manju and Sreenivasan [38] also formulated a simple method for the fabrication of water-soluble curcumin conjugated AuNPs to target various cancer cell lines. AuNPs also cause targeting and sustained release of curcumin and an excellent antioxidant activity.
Silver Nanoparticles: Silver is usually utilized as an incredibly efficient material for antimicrobial utility [39]. Silver nanoparticles are identified for their brilliant optoelectronic properties originated from surface plasmon resonance. They have shown excellent antimicrobial activity compared to other available silver antimicrobial agents. Sodium carboxylmethyl cellulose silver nanocomposite films were attempted for antibacterial applications, so, to improve their applicability, novel film-silver nanoparticle curcumin complexes have been developed [40]. In addition, silver nanoparticles could protect cells against HIV1 infection and help with the wound healing process and also have essential function as an anti-inflammation, an antiviral, and an anticancer agent [41]. So, the combination of silver nanoparticles and curcumin, besides prolonged therapeutic outcomes and sustained release, has several other useful effects such as anti-inflammatory, anti-infection, anticancer, and wound healing.
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT editor-in-chief@pharmatutor.org
Subscribe to Pharmatutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE
Therapeutic Applications of Curcumin:
1. Anticancer activity:
Cancer is the most common threatful disease diagnosed throughout the world. Conventional treatments like chemotherapy, radiation therapy and surgery cause adverse side effects. Hence, it is essential to develop safer and alternative method of treatment to cure this malignant disease. Currently, herbal drugs are used to discover novel drugs. It is believed that they contain various life-saving pharmacological compounds which are non-toxic and can be used to treat various types of cancer. Thus, curcumin is a herbal product which is used for the treatment of diverse variety of cancer such as pancreatic, oral, breast, prostate, skin, ovary, etc. These effects are mediated by regulating multiple important cellular signalling pathways. Recently, curcumin nanoformulations with enhanced bioavailability, solubility and specific tumour cell targetting were used as novel therapy for the treatment of cancer.
* Breast cancer:
Breast cancer is the most common and frequently diagnosed cancer affecting women worldwide. Somasundaram et al., [42] reported a significant inhibition of tumor regression in a xenograft mouse model of human breast cancer. Lvov et al., [43] used gelatin layer coated nanoparticles to deliver polyphenols effectively to breast cancer sites. The combination of curcumin-encapsulated nanoparticles with electroporation technique in MCF-7 human breast cancer cells depicted better anticancer activity [44].
* Ovarian cancer:
Ovarian cancer comprises different types of cancer depending on the cells from which they form. The major difficulty in treating advanced ovarian cancer is chemoradiotherapy resistance. But the curcumin nanoparticles (conjugated with Monoclonal antibody) enhances the site specificity and sensitivity of the chemoradiotherapy resistance of ovarian cancer cells (Yallapu et al.)., [45] . Upon pre-treatment of test subjects with curcumin, it dramatically inhibits proliferation and clonogenic potential of cisplatin resistant cells (A2780CP) in the presence of low levels of cisplatin or radiation. Curcumin has been found to completely inhibit the effect of C-reactive protein (CRP) which has a tendency to damage the vascular endothelial cells [46].
* Pancreatic cancer
Pancreatic cancer is one of the most common cancers, and the fourth leading cause of cancer-related mortality. Bisht et al., synthesised curcumin-loaded polymeric nanoparticles using the co-polymers Nisopropylacrylamide, N-vinyl-2-pyrrolidone and poly (ethylene glycol) monoacrylate. It acts as a potential agent to inhibit the tumour growth in xenograft models of human pancreatic cancer. The therapeutic effectiveness of nanocurcumin was confirmed by cell viability and clonogenic assays [47]. Curcumin-loaded magnetic nanoparticles significantly stopped the growth of human pancreatic cancer cells (HPAF-II and Panc-1) in xenograft mouse model. This formulation showed higher stability with increased bioavailability and biodistribution when compared with normal curcumin [48]. Nanoparticle-Encapsulated Curcumin (NanoCurc) Blocks Tumor Growth and Metastases by binding to NF-κB hence curing the DNA-binding capacity of cellular components [49].
* Prostate cancer:
Prostate cancer is a disease which develops in the prostate gland of the male reproductive system. Gradually, it may spread to other parts of the body like bones and lymph nodes [50]. Curcumin-loaded poly (lactic-co-glycolic acid) (PLGA) nanoparticles prepared by Yallapu et al., [51] demonstrated the anticancer activity of curcumin nanoparticles against prostate cancer. The in vitro studies of curcumin-loaded PLGA nanospheres in prostate cancer cell lines showed sustained delivery of curcumin for a prolonged period of time and increased rate of intracellular uptake of nanospheres [52].
* Cervical cancer:
Cervical cancer is one of the most common and deadly cancers among women worldwide and is associated with persistent Human Papillomavirus (HPV) infection. Nano-CUR effectively inhibits cell growth, induces apoptosis, and arrests the cell cycle in cervical cancer cell lines. The in vitro antitumor activity of curcumin in HPV associated cells has been established [53]. Curcumin modulates the in vitro expression and function of P-gp in multidrug-resistant human KB-V1 cells [54,55]. The effect of curcumin in HPV-associated cells was found to involve the down-regulation of viral oncogenes, NF-jB and AP-1 [54,56].
2. Antimicrobial activity
Micro-organisms play a major role in causing numerous infections to humans. Traditionally, turmeric has been used as an antimicrobial agent. Curcumin nanoparticles were used as they are known to possess superior antimicrobial activity than the normal curcumin. Bhawana et al., reported the antibacterial and antifungal activities of nanocurcumin prepared by wet-milling technique. The nanocurcumin was more water dispersible in the absence of any surfactants and highly active against Staphylococcus aureus, Bacillus subtilis, Escherichia coli, Pseudomonas aeruginosa, Penicillium notatum and Aspergillus niger. The nanocurcumin formulation was more reactive against Gram-positive bacteria than the Gram-negative bacteria [57]. In another study, curcumin-encapsulated nanoparticles inhibited the growth of methicillin-resistant S.aureus and P. aeruginosa and enhanced the wound-healing activity in an in vivo murine wound model [58]. Similarly, the in vitro studies of curcumin-loaded chitosan tripolyphosphate nanoparticles on mouse skin suppressed the growth of S. aureus and P. aeruginosa [59].
3. Anti-HIV activity
Human immunodeficiency virus (HIV) attacks the immune system by destroying CD4+ T cells. The progressive failure of the immunity finally leads to acquired immunodeficiency syndrome (AIDS). The CD4+ T cells are a type of white blood cells that protect the body from infections. The antiretroviral drug suppresses the virus, but the complete elimination was not yet achieved. Hence, it is necessary to find an alternative therapy to treat this fatal condition. Curcumin containing apotransferrin nanoparticles exhibit effective inhibition of HIV-1 replication in vitro. The nanocurcumin down regulates gag gene expression as a result inhibits the synthesis of proviral genes. However the cytotoxicity of the cell is less and the nanoparticle effectivity is dependent on the cellular up take mediated by transferrin receptor [60].
4. Antimalarial activity
Malaria is caused by parasites and carried by female Anopheles mosquitoes. The in vivo studies ofcurcumin-loaded hydrogel nanoparticles reported by Dandekar et al., [61] showed antimalarial activity. The toxicity studies proved the oral safety and cytotoxic effects of the nanoformulations. Curcumin-loaded chitosan nanoparticles cured the mice infected with Plasmodium yoelii by blocking the hemozoin synthesis [62].
5. Anti-inflammatory activity
In ancient Indian medicine, turmeric has been used as an anti-inflammatory agent. Rocha et al., [63] compared the anti-inflammatory activity of normal curcumin and nanocurcumin in rat. The inhibitory effect shown by nanocurcumin at dose 50 mg/kg was similar to that of normal curcumin at dose 400 mg/kg which proved the improved anti-inflammatory activity of nanocurcumin. Curcumin-encapsulated exosomes were studied for their potency in lipopolysaccharide-induced septic shock mouse model. In that experiment, curcumin delivered by exosome demonstrated more stability, target specificity and they were found in high concentrations in blood [64].
6. Alzheimer’s disease
Alzheimer’s disease (AD) is a progressive neurodegenerative disorder that occurs all over the world. It is a common type of dementia associated with memory loss and gradual death of brain cells. AD is characterized by the presence of extracellular deposition of aggregated amyloid-ß (Aß) peptide and intraneuronal accumulation of hyper phosphorylated Tau protein and activation of caspase pathway [65]. Curcumin suppressed oxidative tissue damage and reduced amyloid-ß deposit in mice. Nps-Cur could be a promising drug delivery strategy to protect neurons against oxidative damage in Alzheimer’s disease [66]. Nanocurcumin treated mice showed better cue memory in the contextual fear conditioning test and greater working memory in the radial arm maze test [67]. PLGA-coated curcumin nanoparticles in conjugation with Tet-1 peptide possess anti-amyloid and antioxidant property and it can be used as a potential drug for treating AD [68]. Thus, employing nanocurcumin proved to be a better therapeutic approach for the treatment of AD.
7. Parkinson’s disease
Parkinson’s disease is a disorder that affects the central nervous system. It is caused by the abnormal accumulation of aggregated a-synuclein (aS) which happens due to genetic mutations and exposure to neurotoxins and intracellular reactive oxygen species (ROS) that affects the mitochondria that leads to mitochondrial disfunction. It is found that curcumin can mitigate aS-induced cytotoxicity provided with the advantage that curcumin can cross the blood barrier system in neuron degeneration . In parkinson’s disease curcumin reduced ROS levels that has been generated by oligomeric a-synuclein [69,70]
8. Chronic obstructive pulmonary disease
COPD is caused by bacterial and viral infections and also due to smoking. Reactive oxygen species play an important role in causing inflammation through stress kinases and redox sensitive transcription factors such as nuclear factor (NF)-κB and activator protein. Activation of (NF)-κB increases acetylation and inhibits deacetylation activity which leads to inflammatory gene expression and attenuated glucocorticoid sensitivity. The polyphenols present in curcumin play a role in controlling the activation of NF-κB and thus it can be used in lung epithelial cells to control the expression of inflammatory gene [71].
9. Heart failure
Heart failure is generally caused by the increase in thickness of cardiac muscle (hypertrophy). Hypertrophy is aided with diastolic, systolic dysfunction of the heart and activates hypertrophy-responsive transcriptional factors. The activation of transcriptional factors is mediated through acetylation by histone deacetylase and intrinsic histone acetyl transferase (HAT), p300 [72,73]. Curcumin, a natural therapeutic agent for heart diseases possess a HAT inhibitory activity. Shimatsu et al [74] proved the effect of curcumin in two different heart failure models in vivo: one model was hypertensive heart disease in salt-sensitive Dahl (DS) rats, and the other model was MI in rats. The result shows that inhibited activity of HAT by curcumin prevented the heart failure in both models. NF-κB factor is also involved in cardiomyocyte hypertrophy. So curcumin which has already been known to inhibit NF-κB can also be used in preventing myocarditis.
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT editor-in-chief@pharmatutor.org
Subscribe to Pharmatutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE
CONCLUSION: Nanotechnology is an innovative idea that can be used to overcome the problems associated with curcumin solubility, stability and bioavailability. In this review, the types and therapeutic applications of curcumin nanoparticles are discussed. Development of nanodosage forms of curcumin includes liposomes, polymeric nanoparticles, solid lipid nanoparticles, polymeric micelles, magnetic nanoparticles, nanogels, gold nanoparticles, silver nanoparticles, etc. These nanoparticles act as a potential agent against various diseases like cancer, microbial infection, HIV, malaria, inflammatory diseases, alzheimer, parkinson, pulmonary diseases and heart diseases. Therefore, the promise of nanotechnology-based medicine may become a reality with sufficient efforts and further researches. Human trials need to be conducted to establish curcumin's effectiveness in clinical applications.
REFERENCES
1. Masuda T, Isobe J, Jitoe A, and Nakatani N, (1992): Antioxidative curcuminoids from rhizomes of Curcuma xanthorrhiza. Phytochemistry. , 31: 3645-7.
2. Sreejayan S, and Roa M. N, (1994): Curcuminoids as potent inhibitor of lipid peroxidation. J. Pharm. Pharmacol, 46: 1013-6.
3. Unnikrishnan, M. K. and Roa, M. N. (1995): Inhibition of nitrite induce oxidation of hemoglobin by curcuminoids. Pharmazie. , 50: 490-2.
4. Kiuchi F, Goto Y, Sugimoto N, Akao N, Kondo K, Tsuda Y: Nematocidal activity of turmeric: synergistic action of curcuminoids. Chem Pharm Bull (Tokyo) 1993; 41:1640–3.
5. Sharma K, Agrawal SS, Gupta M. Development and validation of uv spectrophotometric method for the estimation of curcumin in bulk drug and pharmaceutical dosage forms. Int J drug dev res, 2012; 4(2): 375-380.
6. Sabita N, Saldanha and Trygve O, Tollefsbol: The Role of Nutraceuticals Chemoprevention and Chemotherapy and Their Clinical Outcomes. Journal of Oncology. , vol. 2012, 23 pages.
7. Lao C. D, Ruffin M. T, Normolle D, Heath D. D, Murray S. I, Bailey J. M, Boggs M. E, Crowell J, Rock C. L, Brenner D. E: Dose escalation of a curcuminoid formulation. BMC Complement. Altern. Med. 2006, 6, 10.
8. Letizia, Da Sacco, Andrea, Masotti: Chitin and Chitosan as Multipurpose Natural Polymers for Groundwater Arsenic Removal and As2O3 Delivery in Tumor Therapy. Mar. Drugs. , 2010, 8: 1518-1525.
9. Anwunobi1 A. P, and Emeje M. O: Recent Applications of Natural Polymers in Nanodrug Delivery. J Nanomedic Nanotechnol. , 2011. , S4: 1-6.
10. Paul, D. R. and Robeson, L. M: Polymer nanotechnology: Nanocomposites. Polymer. , 2008, 49: 3187– 3204.
11. Malam Y, Loizidou M, and Seifalian A. M: Liposomes and nanoparticles: nanosized vehicles for drug delivery in cancer, Trends in Pharmacological Sciences, 2009, vol. 30, no. 11, pp. 592– 599.
12. Li L, Braiteh FS, and Kurzrock R: Liposome-encapsulated curcumin: in vitro and in vivo effects on proliferation, apoptosis, signaling, and angiogenesis, Cancer, 2005, vol. 104, no. 6, 1322–1331.
13. Rahman S, Cao S, Steadman KJ, Wei M, Parekh HS: Native and beta-cyclodextrin-enclosed curcumin: entrapment within liposomes and their in vitro cytotoxicity in lung and colon cancer, Drug Delivery, 2012, vol. 19, no. 7, pp. 346–353.
14. Chen Y, Wu Q, Zhang Z, Yuan L, Liu X, Zhou L: Preparation of curcumin-loaded liposomes and evaluation of their skin permeation and pharmacodynamics. , Molecules, 2012; vol. 17, no. 5, 5972–5987.
15. Li DC, Zhong XK, Zeng ZP: Application of targeted drug delivery system in Chinese medicine. , 2009, Journal of Controlled Release, vol. 138, no. 2, pp. 103–112.
16. Anitha A, Maya S, Deepa N: Efficient water soluble O-carboxy methyl chitosan nanocarrier for the delivery of curcumin to cancer cells, Carbohydrate Polymers, 2011, vol. 83, no. 2, pp. 452–461.
17. Das RK, Kasoju N, and Bora U: Encapsulation of curcumin in alginate-chitosan-pluronic composite nanoparticles for delivery to cancer cells. , Nanomedicine, 2010, vol. 6, no. 1, pp. 153–160.
18. Yallapu MM, Gupta BK, Jaggi M, Chauhan SC: Fabrication of curcumin encapsulated PLGA nanoparticles for improved therapeutic effects in metastatic cancer cells. , Journal of Colloid and Interface Sci, 2010, vol. 351, no. 1, pp. 19–29.
19. Anand P, Nair HB, Sung B: Design of curcumin loaded PLGA nanoparticles formulation with enhanced cellular uptake, and increased bioactivity in vitro and superior bioavailability in vivo, Biochemical Pharmacology, 2010, vol. 79, no. 3, pp. 330–338.
20. Shaikh J, Ankola DD, Beniwal V, Singh D, Kumar MNVR: Nanoparticle encapsulation improves oral bioavailability of curcumin by at least 9-fold when compared to curcumin administered with piperine as absorption enhancer, European Journal of Pharm Sci, 2009, vol. 37, no. 3-4, pp. 223–230.
21. Song Z, Feng R, Sun M: Curcumin-loaded PLGA-PEG-PLGA triblock copolymeric micelles: Preparation, pharmacokinetics and distribution in vivo, J. of Colloid and Interface Sci, 2011, vol. 354, no. 1, pp. 116-123.
22. Pardeike J, Hommoss A and Muller RH: Lipid nanoparticles (SLN, NLC) in cosmetic and pharmaceutical dermal products, International Journal of Pharmaceutics, 2009, vol. 366, no. 12, pp. 170–184.
23. Michael D. Triplett, PhD thesis: Enabling Solid Lipid Nanoparticle Drug Delivery Technology By Investigating Improved Production Techniques, 2004.
24. Kakkar V, Singh S, Singla D, Kaur IP: Exploring solid lipid nanoparticles to enhance the oral bioavailability of curcumin. , Molecular Nutrition and Food Research, 2011, vol. 55, no. 3, pp. 495–503.
25. Dadhaniya P, Patel C, Muchhara J: Safety assessment of a solid lipid curcumin particle preparation: acute and subchronic toxicity studies, Food and Chemical Toxicology, 2011, vol. 49, no. 8, pp. 1834–1842.
26. Aqil, Munagala R, Jeyabalan J, Vadhanam MV: Bioavailability of phytochemicals and its enhancement by drug delivery systems, Cancer Letters, 2013, vol. 334, no. 1, pp. 133–141 .
27. Jones MC and Leroux JC: Polymeric micelles- a new generation of colloidal drug carriers, European J. Of Pharmaceutics and Biopharmaceutics, 1999, vol. 48, no. 2, pp. 101–111.
28. Liu L, Sun L, Wu Q: Curcumin loaded polymeric micelles inhibit breast tumor growth and spontaneous pulmonary metastasis, International Journal of Pharmaceutics, 2013, vol. 443, no. 1-2, pp. 175–182.
29. Raveendran R, Bhuvaneshwar G, and Sharma CP: In vitro cytotoxicity and cellular uptake of curcumin-loaded fluronic/ Polycaprolactone micelles in colorectal adenocarcinoma cells, Journal of Biomaterials Applications, 2013, vol. 27, no. 7, pp. 811– 827.
30. Yang Y, Jiang JS, Du B, Gan ZF, Qian M, Zhang P: Preparation and properties of a novel drug delivery system with both magnetic and biomolecular targeting. , Journal of Materials Science, 2009, vol. 20, no. 1, pp. 301–307.
31. Yallapu MM, Othman SF, Curtis ET, BGupta BK, Jaggi M, Chauhan SC: Multi-functional magnetic nanoparticles for magnetic resonance imaging and cancer therapy, Biomaterials, 2011, vol. 32, no. 7, pp. 1890–1905.
32. Tran LD, Hoang NMT, Mai TT: Nanosized magnetofluorescent Fe3O4-curcumin conjugate for multimodal monitoring and drug targeting, Colloids and Surfaces, 2010, vol. 371, no. 1–3, pp. 104–112.
33. Goncalves C, Pereira P, Schellenberg P, Coutinho P, Gama F: Self-assembled dextrin nanogel as curcumin delivery System, Journal of Biomaterials and Nanobiotechnology, 2012, vol. 3, no. 2, pp. 178–184.
34. Wu W, Shen J, Banerjee P, Zhou S: Water-dispersible multifunctional hybrid nanogels for combined curcumin and photothermal therapy, Biomaterials, 2011 vol. 32, no. 2, pp. 598–609.
35. Omidfar K, Khorsand F, Darziani Azizi M: New analytical applications of gold nanoparticles as label in antibody based sensors, Biosensors and Bioelectronics, 2013, vol. 43, pp. 336–347.
36. Rajesh J, Rajasekaran M, Rajagopal G, Athappan P: Analytical methods to determine the comparative DNA binding studies of curcumin-Cu(II) complexes, Spectrochimica Acta: Molecular and Biomolecular Spectroscopy, 2012, vol. 97, pp. 223–230.
37. Singh DK, Jagannathan R, Khandelwal P, Abraham PM, Poddar P: In situ synthesis and surface functionalization of gold nanoparticles with curcumin and their antioxidant properties: An experimental and density functional theory investigation, Nanoscale, 2013, vol. 5, no. 5, pp. 1882–1893.
38. Manju S and Sreenivasan K: Gold nanoparticles generated and stabilized by water soluble curcumin-polymer conjugate: Blood compatibility evaluation and targeted drug delivery onto cancer cells, Journal of Colloid and Interface Science, 2012, vol. 368, no. 1, pp. 144–151.
39. Sweet MJ and Singleton I: Silver nanoparticles: A microbial Perspective, Advances in Applied Microbiology, 2011, vol. 77, pp. 115–133.
40. Varaprasad K, MuraliMohan Y, Vimala K, Mohana Raju K: Synthesis and characterization of hydrogel-silver nanoparticle-curcumin composites for wound dressing and antibacterial application, Journal of Applied Polymer Science, 2011, vol. 121, no. 2, pp. 784–796.
41. Zhou H, Wu X, Xu W, Yang J, Yang Q: Fluorescence enhancement of the silver nanoparticales-curcumin-cetyl trimethyl ammonium bromide-nucleic acids system and its analytical application, Journal of Fluorescence, 2010, vol. 20, no. 4, pp. 843–850.
42. Somasundaram S, Edmund NA, Moore DT, Small GW, Shi YY, Orlowski RZ: Dietary curcumin inhibits chemotherapy induced apoptosis in models of human breast cancer, Cancer Res, 2002, vol. 62, 3868–3875.
43. Lvov, Shutava TG, Akundi SS, Vangala P, Steffan JJ, Bigelow RL, Cardelli JA, O’Neal DP: Layer-by-Layer-Coated Gelatin Nanoparticles as a Vehicle for Delivery of Natural Polyphenols. American Chemical Society (ACS Nano), 2009 ; 3(7); 1877-1885.
44. Lin W, Cooper C, Camarillo I: The effectiveness of electroporation based nanocurcumin and curcumin treatments on human breast cancer cells. In Proceedings of ESA annual meeting on electrostatics. , 2014 ,17-19; University of Notre Dame, Notre Dame, Indiana, USA: Electrostatics Society of America; p. 1-7.
45. Yallapu MM, Maher DM, Sundram V, Bell MC, Jaggi M, Chauhan SC: Curcumin induces chemo/radio-sensitization in ovarian cancer cells and curcumin nanoparticles inhibit ovarian cancer cell growth.
J Ovarian Res, 2010; 3: 11.
46. Ganta S, Amiji M: Co-administration of Paclitaxel and Curcumin in nanoemulsion formulation to overcome Multidrug resistance in tumour cells. , Mol Pharm, 2009; 6(3): 928-939.
47. Bisht S, Mizuma M, Feldmann G, Ottenhof N, Hong SM, Pramanik D, Chenna V, Karikari C, Sharma R, Goggins MG, Rudek MA, Ravi R, Maitra A, Maitra A: Systemic administration of polymeric nanoparticle-encapsulated curcumin (NanoCurc) blocks tumor growth and metastases in preclinical models of pancreatic cancer.
Mol Cancer Ther, 2010; 9(8): 2255-2264.
48. Yallapu MM, Ebeling MC, Khan S, Sundram V, Chauhan N, Gupta BK, Puumala SE, Jaggi M, Chauhan SC: Novel curcumin-loaded magnetic nanoparticles for pancreatic cancer treatment. ,
Mol Cancer Ther, 2013; 12(8): 1471-1480.
49. Amarnath Maitra, Anirban Maitra, Collins Karikari, Dipankar Pramanik, Georg Feldmann, Masamichi Mizuma, Michael G. Goggins, Michelle A. Rudek, Systemic Administration of polymeric nanoparticle-encapsulated curcumin(Nanocurc) blocks tumour growth and metastases in Preclinical Models of Pancreatic Cancer. Molecular Cancer Therapeutics, 2010; 9:2255-2264.
50. Ruddon RW. Cancer biology, 4th ed. Oxford University Press, Oxford, 2007, pp. 223.
51. Yallapu MM, Khan S, Maher DM, Ebeling MC, Sundram V, Chauhan N, Ganju A, Balakrishna S, Gupta BK, Zafar N, Jaggi M, Chauhan SC: Anti-cancer activity of curcumin loaded nanoparticles in prostate cancer. , Biomaterials, 2014; 35(30): 8635-8648.
52. Mukerjee A, Vishwanatha JK: Formulation, characterization and evaluation of curcumin-loaded PLGA nanospheres for cancer therapy. , Anticancer Res, 2009; 29(10): 3867-3876.
53. Roy M, Chakraborty S, Siddiqi M, Bhattacharya RK: Induction of apoptosis in tumor cells by natural phenolic compounds, Asian Pac. J. Cancer Prev. 3 (2002) 61–67.
54. Aggarwal B, Bhatt ID, Ichikawa H, Ahn KS, Sethi G, Sandur SK, Sundaram C, Seeram N, Shishodia S: Curcumin – biological and medicinal properties and Turmeric the Genus Curcuma, CRC Press, NY, 2007, pp. 297–368.
55. Chearwae W, Anuchapreeda S, Nandigama K, Ambudkar SV, Limtrakul P, Biochemical mechanism of modulation of human P-glycoprotein (ABCB1) by curcumin I, II, and III purified from turmeric powder, Biochem. Pharmacol. 68 (2004) 2043–2052.
56. Divya CS, Pillai MR: Antitumor action of curcumin in human papillomavirus associated cells involves down regulation of viral oncogenes, prevention of NFkB and AP-1 translocation, and modulation of apoptosis, Mol. Carcinog. 45 (2006) 320–332.
57. Bhawana B, Basniwal RK, Buttar HS, Jain VK, Jain N: Curcumin nanoparticles: Preparation, characterization and antimicrobial study, Agricultural and Food Chemistry, 2011, vol. 59, no. 5, pp. 2056–2061.
58. Krausz AE, Adler BL, Cabral V, Navati M, Doerner J, Charafeddine RA, Chandra D, Liang H, Gunther L, Clendaniel A, Harper S, Friedman JM, Nosanchuk JD, Friedman AJ: Curcumin-encapsulated nanoparticles as innovative antimicrobial and wound healing agent. ,
Nanomedicine, 2015; 11(1): 195206.
59. Mirnejad R, Jahromi MAM, Al-Musawi S, Pirestani M, Ramandi MF, Ahmadi K, Rajayi H, Hassan ZM, Kamali M: Curcumin-loaded Chitosan Tripolyphosphate Nanoparticles as a safe, natural and effective antibiotic inhibits the infection of S. aureus and S. aeruginosa in vivo: Iran J Biotech, 2014; 12(3): e1012.
60. Golla Kishore, Raju C. Reddy, R. K. Chaitanya and Upendhar Gandapu: Curcumin Loaded Apotransferrin Nanoparticles Provide Efficient Cellular Uptake and Effectively Inhibit HIV-1 Replication in vitro, 2011.
61. Dandekar PP, Jain R, Patil S, Dhumal R, Tiwari D, Sharma S, Vanage G, Patravale V: Curcumin-loaded hydrogel nanoparticles: application in anti-malarial therapy and toxicological evaluation. , J Pharm Sci. , 2010; 99(12): 4992-5010.
62. Akhtar F, Rizvi MM, Kar SK: Oral delivery of curcumin bound to chitosan nanoparticles cured Plasmodium yoelii infected mice. ,
Biotechnol Adv, 2012; 30(1): 310-320.
63. Rocha BA, Gonçalves OH, Leimann FV, Rebecca ESW, Silva-Buzanello RA, Filho LC, Araújo PHH, Cuman RKN, Bersani-Amado CA: Curcumin encapsulated in poly-L-lactic acid improves its anti-inflammatory efficacy in vivo. , Adv Med Plant Res, 2014; 2(4): 62-73.
64. Sun D, Zhuang X, Xiang X, Liu Y, Zhang S: A novel nanoparticle drug delivery system: the anti-inflammatory activity of curcumin is enhanced when encapsulated in exosomes. , Mol Ther. , 2010, 18: 1606–1614
65. Luan-Feng Pan, Ping Zhou, Teng Jiang , Xiu-Ling Zhi , Yue-Hong Zhang: Inhibitory effect of curcumin on the Al(III)-induced Aβ42 aggregation and neurotoxicity in vitro. Biochimica et Biophysica Acta (BBA) – Molecular Basis of Disease. 2012; 1822(8): 1207-1215.
66. Cheng KK, Yeung CF, Ho SW, Chow SF, Chow AH, Baum L: Highly Stabilized Curcumin Nanoparticles Tested in an in Vitro Blood–Brain Barrier Model and in Alzheimer’s Disease Tg2576 Mice. , AAPS J, 2013; 15(2): 324-336.
67. Mathew A, Fukuda T, Nagaoka Y, Hasumura T, Morimoto H, Yoshida Y, Maekawa T, Venugopal K, Kumar DS: Curcumin loaded PLGA Nanoparticles conjugated with Tet-1 peptide for potential use in Alzheimer’s Disease. , PLoS One, 2012; 7(3): e32616.
68. Tiwari SK, Agarwal S, Seth B, Yadav A, Nair S, Bhatnagar P, Karmakar M, Kumari M, Chauhan LK, Patel DK, Srivastava V, Singh D, Gupta SK, Tripathi A, Chaturvedi RK, Gupta KC: Curcumin-Loaded Nanoparticles Potently Induce Adult Neurogenesis and Reverse Cognitive Deficits in Alzheimer’s Disease Model via Canonical Wnt/beta-Catenin Pathway. , ACS Nano, 2014; 8(1): 76-103.
69. Ballerini P, Bau C, D’Alimonte I, Jiang SC, Pettifer KM, Rathbone MP and Werstiuk ES: MPP+ induced cytotoxicity in neuroblastoma cells: Antagonism and reversal by guanosine. Purinerg Signalling, 2007, 3(4): 399-409.
70. Chen J, Chen P. X, Cui Y, Feng J. Q, Sun S. N, Tang X. Q, Tang E. H, Yu H. M, Zhi J. L. Curcumin protects PC12 cells against 1-methyl-4 phenylpyridinium ion-induced apoptosis by Bcl-2-mitochondria-ROSiNOS pathway. Apoptosis: An International Journal on Programmed cell Death, 2006, 11(6): 943-953.
71. Adcock IM and Rahman: Oxidative stress and redox regulation of lung inflammation in COPD. European Respiratory Journal, 2006,28, 219-242.
72. Backs J and Olson EN: Control of cardiac growth by histone
acetylation/deacetylation, Circulation Research,2006; 98: 15-24.
73. McKinsey TA and Olson EN: Cardiac histone acetylation-therapeutic opportunities abound, Trends in Genetics, 2004; 20(4): 206-213.
74. Akira Shimatsu, Atsushi Nagasawa, , Hiromichi Wada, Koji Hasegawa, Masashi Komeda, Masatoshi Fujita, Tatsuya Morimoto, , Teruhisa Kawamura, Tomohide Takaya, Toru Kita and Yoichi Sunagawa. The dietary compound curcumin inhibits p300 histone acetyltransferase activity and prevents heart failure in rats. The Journal of clinical Investigation, 2008, 118(3): 868–878.
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT editor-in-chief@pharmatutor.org
Subscribe to Pharmatutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE









