Breast cancer research takes a significant stride forward as Professor Semin Lee and his research team from the Department of Biomedical Engineering at UNIST, in collaboration with Professor Ji-Yeon Kim and Professor Young-Hyuck Im from the Division of Hematology-Oncology at Samsung Medical Center in Seoul, delves into the exploration of triple negative apocrine carcinoma. This rare breast cancer subtype has garnered attention due to its unique genetic characteristics and improved prognosis when compared to other forms of triple-negative breast cancer (TNBC).
Triple negative apocrine carcinoma accounts for only 1-4% of all breast cancers. While it falls under the category of TNBC, which is characterized by the absence of hormone receptors and epidermal growth factor receptors, this particular subtype exhibits a more favorable prognosis than other TNBC subtypes. However, due to limited analysis studies on triple negative apocrine carcinoma, treatment criteria have remained ambiguous.
In their study titled ‘Multiple Omics Study of Triple-Negative Apocrine Carcinoma,’ the research team employed advanced molecular biological methods to conduct multi-omics analyses—integrating genetic information and RNA molecules—to unravel novel insights into this rare form of breast cancer.
The findings revealed distinct genomic characteristics that impact the prognosis for patients with triple negative apocrine carcinoma. The researchers identified four to five unique subtypes within breast cancer based on gene expression profiles. Notably, patients with triple-negative apocrine carcinoma exhibited similarities to Luminal A—a subtype associated with better prognostic outcomes. The study confirmed an impressive five-year disease-free survival rate for these patients at 92.2%, significantly higher than those diagnosed with other types of TNBC (59.1%).
“This research has the potential to guide treatment decisions regarding adjuvant chemotherapy after surgery and predict patient survival outcomes,” explained Sabin Park (Department of Biomedical Engineering, UNIST), first author of the study, while emphasizing the potential impact of these findings.
Published in the journal, Experimental & Molecular Medicine on July 3, this study received support from the Korea Research Foundation’s Cancer Control Research Center by Intercellular Signal Communication—a program under the Ministry of Science and ICT.
As breast cancer continues to be a significant global health concern affecting millions of lives, studies like these play a crucial role in advancing personalized treatment approaches based on specific genetic mutations. The groundbreaking insights gained from Professor Lee’s team provide hope for improved clinical management and better prognostic outcomes for patients diagnosed with triple negative apocrine carcinoma.
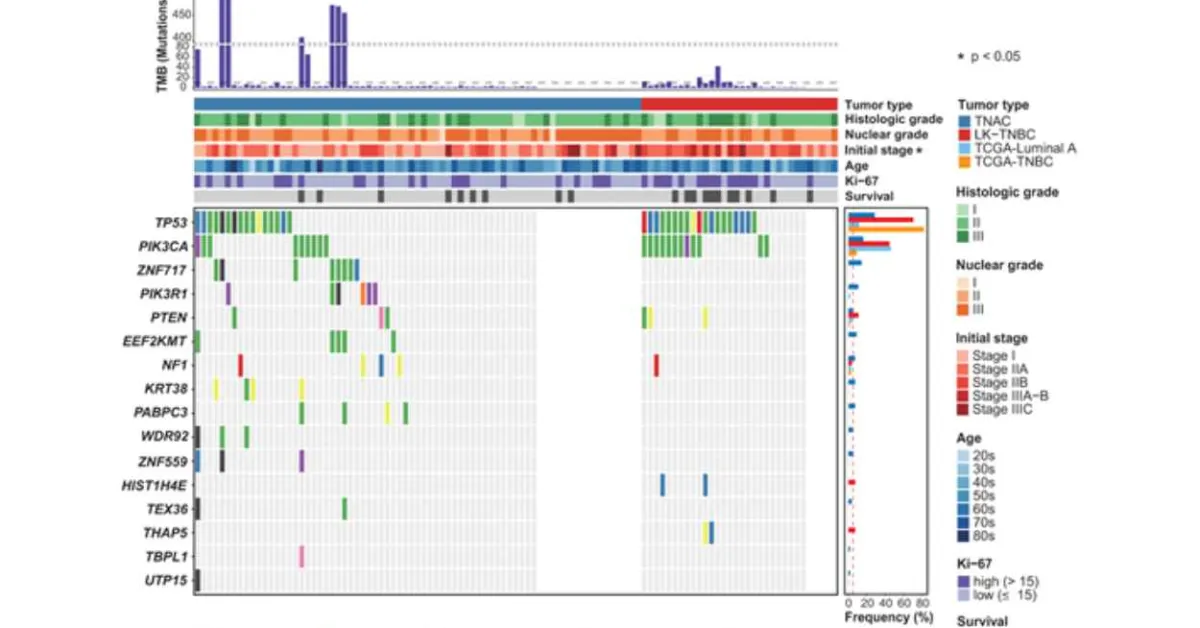
Figure 1. Somatic Point Mutations in TNAC and LK-TNBC. The top 16 significantly mutated genes as determined by the dNdScv algorithm (p < 0.001, TNAC: 13 genes, LK-TNBC: 5 genes) in TNAC and LK-TNBC were sorted by their mutation frequency. The right bar plot shows the mutation frequencies of the 16 genes in our cohorts and TCGA cohorts. The red dotted vertical line represents a 5% mutation frequency. The top bar plot indicates tumor mutational burden (TMB), which represents the number of mutations per megabase (Mb) for each sample. The samples with TMB values greater than 10 were considered hypermutated (black dotted horizontal line). The clinical and pathological characteristics were also annotated. The significant differences between TNAC and LK-TNBC according to baseline characteristics were calculated by Fisher’s exact test.