Researchers have designed the first ‘living medicine’ to treat lung infections. The treatment targets Pseudomonas aeruginosa, a type of bacteria which is naturally resistant to many types of antibiotics and is a common source of infections in hospitals.
The treatment involves using a modified version of the bacterium Mycoplasma pneumoniae, removing its ability to cause disease and repurposing it to attack P. aeruginosa instead. The modified bacterium is used in combination with low doses of antibiotics that would otherwise not work on their own.
Researchers tested the efficacy of the treatment in mice, finding that it significantly reduced lung infections. The ‘living medicine’ doubled mouse survival rate compared to not using any treatment. Administering a single, high dose of the treatment showed no signs of toxicity in the lungs. Once the treatment had finished its course, the innate immune system cleared the modified bacteria in a period of four days.
The findings are published in the journal Nature Biotechnology and are supported by the “la Caixa” Foundation through the CaixaResearch Health call. The study was led by researchers at the Centre for Genomic Regulation (CRG) and Pulmobiotics in collaboration with the Institut d'Investigacions Biomèdiques August Pi i Sunyer (IDIBAPS), Hospital Clinic de Barcelona and the Institute of Agrobiotechnology (IdAB), a joint research institute of Spain’s CSIC and the government of Navarre.
P. aeruginosa infections are difficult to treat because the bacteria lives in communities that form biofilms. Biofilms can attach themselves to various surfaces in the body, forming impenetrable structures that escape the reach of antibiotics.
P. aeruginosa biofilms can grow on the surface of endotracheal tubes used by critically-ill patients who require mechanical ventilators to breathe. This causes ventilator-associated pneumonia (VAP), a condition which affects one in four (9-27%) patients who require intubation. The incidence exceeds 50% for patients intubated because of severe Covid-19. VAP can extend the duration in intensive care unit for up to thirteen days and kills up to one in eight patients (9-13%).
The authors of the study engineered M. pneumoniae to dissolve biofilms by equipping it with the ability to produce various molecules including pyocins, toxins naturally produced by bacteria to kill or inhibit the growth Pseudomonas bacterial strains. To test its efficacy, they collected P. aeruginosa biofilms from the endotracheal tubes of patients in intensive care units. They found the treatment penetrated the barrier and successfully dissolved the biofilms.
“We have developed a battering ram that lays siege to antibiotic-resistant bacteria. The treatment punches holes in their cell walls, providing crucial entry points for antibiotics to invade and clear infections at their source. We believe this is a promising new strategy to address the leading cause of mortality in hospitals,” says Dr. María Lluch, Chief Scientific Officer at Pulmobiotics, co-corresponding author of the study and principal investigator at the International University of Catalonia.
With the aim of using the ‘living medicine’ to treat VAP, the researchers will carry out further tests before reaching the clinical trial phase. The treatment is expected to be administered using a nebulizer, a device that turns liquid medicine into a mist which is then inhaled through a mouthpiece or a mask.
M. pneumoniae is one of the smallest known species of bacteria. Dr. Luis Serrano, Director of the CRG, first had the idea to modify the bacteria and use it as a ‘living medicine’ two decades ago. Dr. Serrano is a specialist in synthetic biology, a field that involves repurposing organisms and engineering them to have new, useful abilities. With just 684 genes and no cell wall, the relative simplicity of M. pneumoniae makes it ideal for engineering biology for specific applications.
One of the advantages of using M. pneumoniae to treat respiratory diseases is that it is naturally adapted to lung tissue. After administering the modified bacterium, it travels straight to the source of a respiratory infection, where it sets up shop like a temporary factory and produces a variety of therapeutic molecules.
By showing that M. pneumoniae can tackle infections in the lung, the study opens the door for researchers creating new strains of the bacteria to tackle other types of respiratory diseases such as lung cancer or asthma. “The bacterium can be modified with a variety of different payloads – whether these are cytokines, nanobodies or defensins. The aim is to diversify the modified bacterium’s arsenal and unlock its full potential in treating a variety of complex diseases,” says ICREA Research Professor Dr. Luis Serrano.
In addition to designing the ‘living medicine’, Dr. Serrano’s research team are also using their expertise in synthetic biology to design new proteins that can be delivered by M. pneumoniae. The team are using these proteins to target inflammation caused by P. aeruginosa infections.
Though inflammation is the body's natural response to an infection, excessive or prolonged inflammation can damage lung tissue. The inflammatory response is orchestrated by the immune system, which release mediator proteins such as cytokines. One type of cytokine – IL-10 – has well-known anti-inflammatory properties and is of growing therapeutic interest.
Research published in the journal Molecular Systems Biology by Dr. Serrano’s research group used protein-design softwares ModelX and FoldX to engineer new versions of IL-10 purposefully optimised to treat inflammation. The cytokines were designed to be created more efficiently and to have higher affinity, meaning less cytokines are needed to have the same effect.
The researchers engineered strains of M. pneumoniae that expressed the new cytokines and tested its efficacy in the lungs of mice with acute P. aeruginosa infections. They found that engineered versions of IL-10 were significantly more effective at reducing inflammation compared to the wild type IL-10 cytokine.
According to Dr. Ariadna Montero Blay, co-corresponding author of the study in Molecular Systems Biology, “live biotherapeutics such as M. pneumoniae provide ideal vehicles to help overcome the traditional limitations of cytokines and unlock their huge potential in treating a variety of human diseases. Engineering cytokines as therapeutic molecules was critical to tackle inflammation. Other lung diseases such as asthma or pulmonary fibrosis could also stand to benefit from this approach.”
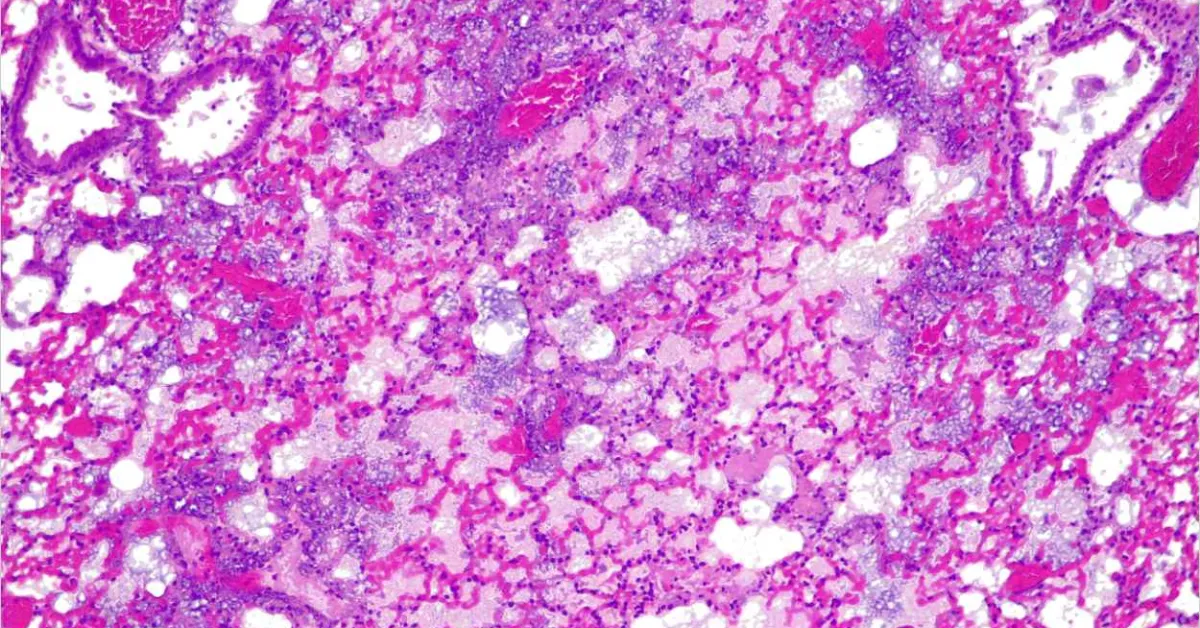
A cross-section of a mouse lung infected with Pseudonomas aeruginosa, resulting in severe pneumoniae. This is characterised by massive infiltration of inflammatory cells into the alveolar septa, resulting in loss of air in the alveoli. Credit: Rocco Mazzolini/CRG