{ DOWNLOAD AS PDF }
ABOUT AUTHORS
Jigar Vyas*, Juhi H. Patel, Riya N. Patel, Daxini kapil, Kumar vinodbhai
Sigma Institute of Pharmacy,
Ajwa-Nimeta road, Bakrol, Vadodara, Gujarat
ABSTRACT
Recently, incidences of breast cancer are increasing and it has become a leading cause of cancer related deaths of women globally. Though pharmaceutical and biotechnology industries are developing, and researchers are working to find a better treatment of breast cancer patients to improve the effectiveness of current therapy, the survival rate is very low. A lot of research is going on based on lipid particulate systems which has versatile applications for the treatment of life-threatening diseases such as breast cancer. Out of various lipid particulate systems, liposomes have been studied and explored widely as they have many applications such as improving solubility of antitumor drugs, reduces the side effects, long circulation in blood, possibility of passive and active targeting to tumor cells. Liposomal system is most popular because of its biocompatibility, capability to encapsulate hydrophilic as well as hydrophobic drugs, ease of surface modification and wide range of applications. Thus, a thorough research is ongoing for upgradation of liposomes as multipotential carriers and its effective therapeutic. Therefore, this review article includes the recent development and techniques of liposomal nano formulations drug delivery system. This review majorly focuses on the breast cancer and its therapy, current challenges in experimental and clinical studies in liposomal formulations.
Reference Id: PHARMATUTOR-ART-2661
|
PharmaTutor (Print-ISSN: 2394 - 6679; e-ISSN: 2347 - 7881) Volume 7, Issue 05 Received On: 26/03/2019; Accepted On: 29/03/2019; Published On: 01/05/2019 How to cite this article: Vyas, D., Patel, J., Patel, R. and Daxini, K.K. 2019. Receptor Targeting Liposomal Nano Formulations for Drug Delivery in Breast Cancer. PharmaTutor. 7, 5 (May 2019), 1-9 |
INTRODUCTION
Cancer as on today is one of the most difficult health related problem worldwide for men as well as women. Besides availability of understanding the molecular mechanisms of cancer, chemotherapeutic drugs and radiotherapy as well as surgical treatment options, current treatment strategies face many problems and a treatment does not turn out successful to eradicate cancer in patients. The major obstacles of current scenario for cancer therapy are, difficulty in early detection, off target effects of chemotherapy, relapse and multi drug resistance. Researchers are working on to develop nanoformulations drug delivery systems such as polymeric nanoparticles (Patel et al., 2018), targeted protein nanoparticles (Yewale et al., 2018), liposomes as well as gene therapy (P. Bhatt, Khatri, Kumar, Baradia, & Misra, 2015); (Patil et al., 2016) options are also being explored for selective targeting of drug, to reduces side effects and improve effectiveness of treatment in cancer therapy. Out of various forms of cancers, breast cancer is the leading cause of death among the women globally and a second most worldwide spread cancer. This cancer is day by day growing in the women and highly widespread which is reported by international agency for cancer research in the year of 2015. One woman out of eight has been diagnosed having breast cancer in united states in 2016. Therefore, in women death by the breast cancer is noted second foremost causes of death. The risk factors of the breast cancer are reproductive factors, lifestyle factors, family history, radiation exposure and changes in BCRA1, TP53 and BCRA2. The breast cancer treatments are radiotherapy, hormone therapy, chemotherapy, biological therapy and surgery (Hortobagyi, 1998). However, these treatments success on breast cancer depends on stages of cancer in patient (Elhazzat J, 2010); (Priyanka Bhatt & Narvekar, 2018). Moreover, chemotherapeutic is also one of the effective treatments but these treatments have several limitations because of its adverse effect on other healthy cells of body. Failures and ineffective results in chemotherapy mainly is because of multi drug resistance (Dhankhar et al., 2010). Additionally, chemotherapeutic agent’s nonselective cytotoxic activity on healthy cells, chemotherapeutics physicochemical property limitations in given dosage are other challenges in this treatment (J. W. Park, 2002). To overcome such problems and side effects, the novel liposomal nanodrug delivery system is one of the best suggested solution (Elhazzat J, 2010). These formulations are one of the most effective and efficient technique among others such as solid lipid nanoparticles, lipid nanoemulsions, nanostructured lipid carriers and nanosuspensions (Puri et al., 2009).
In recent technological development in targeted drug delivery system by the liposomes nano formulations are the most popular and common for breast cancer treatment (Koning & Storm, 2003). Also observing the last three decades the liposomes-based delivery techniques have been growing because of unique characteristics which includes low toxicity, lack of immune system activation, biocompatibility, bio-degradability and therapeutically active agents (Voinea & Simionescu, 2002). Moreover, it can incorporate hydrophilic as well as hydrophobic drugs as shown in figure1.
It has been noticed that liposomes have good therapeutic potential to carry payload and delivery it to the target sites. These tremendous potential leads researcher for clinical trials to developed the liposomal based nano formulation drug delivery for breast cancer therapy (Voinea & Simionescu, 2002). The first time liposomal based targeting drug delivery system was introduced as magic bullet in 1906 (Strebhardt & Ullrich, 2008). Henceforth, pharmacodynamic and kinetic profiles can be improved by sustain release of incorporated drugs to lower toxicity in comparison of free drug. There are several key points which reflects therapeutic potential of liposome such as intracellular delivery followed by triggered and organelle-specific targeting of therapeutic payloads to tumor cells byactively targeting (Sawant & Torchilin, 2010); (Torchilin, 2005). This review is exploring the recent development and employed strategies to enhance present liposomal techniques in breast cancer therapy.
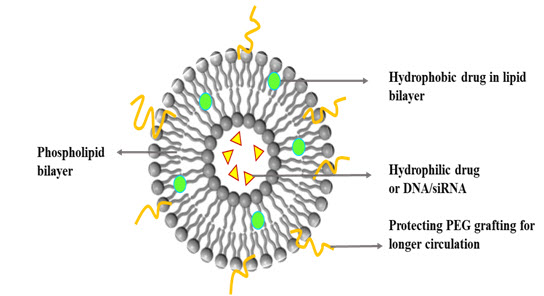
Figure 1. Hydrophilic and hydrophobic drugs encapsulated within a PEGylated liposome (Voinea & Simionescu, 2002).
Breast cancer current therapy and challenges
There are many types of diagnostics methods which are used to diagnose breast cancer. These imaging tests methods include pelvic examination by Magnetic Resonance Imaging, Computerized tomography scans, serum biomarkers detection of Cancer Antigen 125 and transvaginal ultrasound or ultrasonography etc. These technics of diagnostics is not reliable for first stage of breast cancer (Fishman et al., 2005). Hence, it is difficult to detect early stage cancer.
Currently for the treatment of breast cancer, chemotherapy and cyto-reductive surgery are mostly used. Taxol derivatives in combination with platinum compounds in standard chemotherapeutic for newly diagnosed breast cancer (Marchetti et al., 2010). In chemotherapy, risk of recurrence, poor quality patient life, drug resistance are the main barriers due to such problems chemotherapy treatment is clinically handicapped. Moreover, neurotoxicity, nephrotoxicity, ototoxicity and nausea are the adverse effects and can be vary depending on the cancer stage (Johnson, Ozols, & Hamilton, 1993).
Another challenge to breast cancer treatment by chemotherapy is Multi drug resistance. The drug efflux by P-gp is increased than drug in flux in tumor milieu. Detoxification, DNA starts to repair, modification in BCL2 and P53 expression are the recommended mechanism for multi drug resistance (Dinarvand et al., 2013); (Thomas & Coley, 2003); (Zhou et al., 2012). In more than 40% breast cancer it is observed that the P-gp over expression affects the drug concentration in tumor therefore the chemotherapy treatment is failed (Fishman et al., 2005). The drug delivery system is the important factor to overcome P-gp efflux effect by ion-pair formation and endocytosis. It has been observed that surface modification of liposomes with different peptides, antibodies and polymer may improve P-gp potentials (Seelig & Gerebtzoff, 2006).
Liposome and their surface modification
Liposome is defined as lipid vesicle with at least one lipid bilayers. The lipid vesicle consists of aqueous core with one or more phospholipid bilayers. The phospholipid bilayers are consisting of hydrophobic drugs and aqueous compartment incorporates hydrophilic drugs. Therefore, these kinds of vehicles are used for delivering hydrophobic and hydrophilic drugs (Lao et al., 2013) (Kulkarni PR, 2011). The liposome size varies between 0.02 to 5.00 μm. These sizes varies accordingly preparation methods, surfactants and type of phospholipids used (Malam, Loizidou, & Seifalian, 2009). The liposomes have unique characteristics like biodegradability, nontoxicity and biocompatibility therefore it is used in many applications (Allen & Cullis, 2013).The modification in targeted drug delivery system is possible in the liposome hence off-target cytotoxicity can be reduced by targeting them to specific tumor site (Dua J, 2012); (Sharma et al., 2006); (Rivera, 2003). Based on the successful studies and clinical trials some liposomal products are available in the market. As discussed in the above section there are several strategies have been used to resolve the multi drug resistance through liposomal nano delivery system.
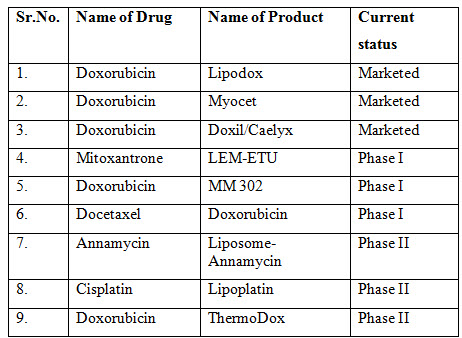
Liposomes surface modification is one of the appropriate drug delivery methods which can improve the ability of liposomal as drug delivery system. In surface modification polyethylene glycol is used as coating which can open new route in standard chemotherapy system (Priyanka Bhatt et al., 2018). Basically, pegylated liposomes improved the pharmacokinetic and pharmacodynamics parameters because of the long circulating system which increase the accumulation of drug in tumor cell. This is also known as stealth liposomes (A. A. Gabizon, 1994). This system has not only good stability but it can increase the blood circulation time by escaping from reticuloendothelial system (A. Gabizon et al., 1994). One of the study carried out on SN-38 included pegylated nanoliposomes and SN-38 included non-pegylated nanoliposomes and did the comparison. They found the pegylated nanoliposomes drug delivery system has significant results and less effects on the body organs like kidney, spleen and liver etc. compare to non-pegylated (A. A. Gabizon, Shmeeda, & Zalipsky, 2006); (Tandel, Bhatt, Jain, Shahiwala, & Misra, 2018). After clinical trials and experimental studies it has been found that the pegylated liposomes have less side effects and the higher safety. The doxorubicin loaded stealth liposomes compared to conventional liposomes are effective and lower cardiotoxic which can be more beneficial to metastatic breast cancer treatment (Atyabi et al., 2009); (J., 2003). The other formulations are under clinical study and development which are shown in the table 1. Also, some of marketed liposomal products are included in the table1.
Table 1. The current marketed products and status of clinical trial liposomes (J., 2003).
Active and passive liposomal targeting strategies
Recent research and development adopted different strategies for targeting liposome to the tumor site. Here in the following section active and passive targeting strategies of liposomes through has been discussed (Lalani, Bhatt, Rathi, & Misra, 2016).
Active liposomal targeting strategy
The development of the active targeted liposomes leads to reduction in the off-target effects of conventional chemotherapeutic drug. Surface of such liposomes are decorated with the ligands such as monoclonal antibodies and peptides as well as proteins to specifically target tumor cells (Rom et al., 2014); (Pathak Nandish 2019). It has been observed that the receptors like transferrin, vascular growth endothelial factor receptor, endothelial growth factor receptor and float receptors are overexpressed on the cancer cells (Vhora et al., 2014). The actively targeted liposomes are surface modified to specifically target these receptors to reduce nonspecific effects of chemotherapeutic drug (Priyanka Bhatt, Vhora, et al., 2016). Actively targeted liposomes function in the tumor microenvironment and later on liposomes are taken up by tumor cells (Vhora, Patil, Bhatt, & Misra, 2015). It is very important to prepare the liposomes to target specific protein on cancer cells as affinity of ligands for cell surface receptors, density of overexpressed receptor proteins and ligand properties play a very important role (Egusquiaguirre et al., 2012).
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT editor-in-chief@pharmatutor.org
Subscribe to Pharmatutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE
One such example of special drug delivery targeting ligand to target breast cancer cells is the folic acid. The folic acid-based targeting ligand decorated liposomes are specifically targeted to folate receptors which is overexpressed in breast cancer cells (Low, Henne, & Doorneweerd, 2008); (Kularatne & Low, 2010). Ligands such as antibodies, peptides and proteins play key role in the effectiveness of actively targeted liposomal formulation (Priyanka Bhatt et al., 2016). Antibodies are widely used as a ligands to target specific receptor protein and is grafted on surface of liposomes to develop immunoliposomes. Table 2 shows the current research and development in immunoliposomes based breast cancer treatments.
Table 2. Breast cancer treatment Immunoliposome based recent formulation development.

Another ligand is the peptides which can be used for the cancer treatment to develop targeted liposomes. The targeting ligands with peptides have significant results in to cells, nucleic acids and improve the injected drug effects. Moreover, such penetration of the peptides in to the cell are not selectively acting therefore these peptides ligands accompanied with antibodies which is delivered in to the tumor cell as cargo are effective (Govindarajan et al., 2012). Transmembrane receptors proteins on the surface of the cell and peptide concentration are two major factor which can affect efficacy of peptide targeted liposomal drug delivery system (Shroff & Kokkoli, 2012).
Passive liposomal targeting strategy
The passive targeting strategy also having advantage in cancer therapy. figure 2 indicates the passive targeting strategy as well as enhanced permeation retention effect.
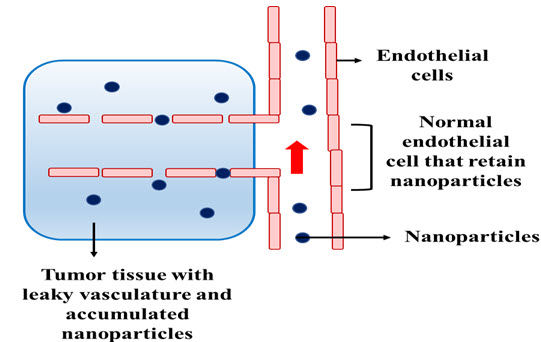
Figure 2. Passive Targeting strategy (E. K. Park, Lee, & Lee, 2005).
Maeda and Matsumura did first time research on polymer in the form of nanoparticles and shows the retention and permeation effects. Enhancement in permeation is due to anatomy of the capillaries of tumor tissues which allows permeation of macromolecules to the tissue as well as the poor lymphatic drainage of malignant tissue increases retention of them in a tumor thus nanoparticulate system deliver drugs 10 to 100 times higher than that through circulating systems. The tumor has leaky nature therefore it is spreads or in a contact with biomacromolecules, blood vessels and also entered in the interstitial area. Based on the cancer type the gap is 100 to 780 nm in-between endothelial cells lining which is tumor capillaries range (Haley & Frenkel, 2008).
The tumor microenvironment is key benefits in passive drug delivering. The chemotherapeutic agents entered in to the body by conjugating with the tumor material. The tumor environment are more active when polymer conjugated drug is delivered and this is known as tumor activated prodrug therapy (Mansour et al., 2003).
The local targeting passive strategy prevent the toxicity and improve the drug concentration at the tumor location. Nomura et al. did study on mitomycin c-dextran conjugate injection on the tumor site and as a result they reduced toxicity and improved anticancer drug’s concentration (Nomura et al., 1998). Similarly, Prabha and Labhasetwar investigated that improvement in antiproliferative effect in breast cancer by nanoparticles-mediated wild-type p53 (Prabha & Labhasetwar, 2004).
Triggered release liposome
The triggered released liposomal formulation to tumor tissue is one of the new developed drug delivery system. For successful and efficient result of the cancer treatment an immediate and direct drug released in to the tumor tissue and this direct delivered liposomal formulation is known as triggered released liposomal. Enzyme and pH alternation as an inner and ultrasound, local heating, light, magnetic field etc. as outer stimulants have been used to for different responsiveness of liposomal drug delivery systems with trigger release. Thermosensitive liposomes are temperature sensitive and are most effective in combination with mild hyperthermia. Such combination not only improve drug release but also enhance the blood flow, vessel pore size at tumor targeted tissues. The research and development in the triggered release liposomal formulation in the various combinations are ThermoDoxR, composed of lysolipid, Ultrasound triggered released drug, photo-triggered liposomes, Matrix metalloproteinase-9 triggered liposome etc. Therefore, such triggered release liposomal formulations are most effective in breast cancer.
In passive drug delivery system, the drug is inserted in inactive form means in a passive way and is directly comes in contact with the highly active tumor tissues. The main function and work of nanoparticles is to locate disease sites and its specific tissues for that they follow the enhanced permeation system and enhanced retention system. The diameter size of the nano particulate liposomes should be less than 100 nm which can help to improve efficiency of delivery system. Moreover, such surface treated pegylated liposome helps to protect surface against plasma protein adsorption. The coating of poloxamines, like polyethylene glycol and polysaccharides can be used as hydrophilic polymer coating of liposomes (Moghimi & Hunter, 2000); (E. K. Park, Lee, & Lee, 2005).
CONCLUSION
The breast cancer pathophysiology progressions and development encourage therapeutic delivery systems development for effective and promising therapy for breast cancer. Since many years ago liposomes were developed and till today research is going on for improvement in drug delivery system applications. Studies and experimental investigation concluded that the liposomal based nano formulation drug delivery system have enhanced cytotoxicity and is potential treatment option of breast cancer therapy. Moreover, it also reduces the side effects of traditional non-specific chemotherapeutic agents. The pegylated liposomes are useful to elongate pharmacokinetic, ameliorate efficacy and stability of the liposomes. These surface modification decreases the off-target toxicities in comparison to free drug. Breast cancer treatment can be improved by developing actively targeted liposomes and surface modification of liposomes by targeting ligands such as peptides, folic acid, antibodies, carbohydrates and modifying the liposomal structure. Basically, actively targeting liposomes are made for decrease the off-targeting effects in the breast cancer using small-molecule ligands. In a passive targeting drug delivery system through leaky capillary nature take benefits and drug can easily cross the vessel wall and reaches to tumor tissues. Also, in tumor vasculature lymphatic drainage is very less and it can accommodate the drug loaded nano liposomal formulation by retention effect.
Several liposomal nano formulations are under the clinical trial and currently on different stages for breast cancer treatment. As seen in this review the research is going on very selectivity, individually and specificity for treatment of breast cancer. These novel approaches are promoted by young researchers and they are promising and presenting the effective results.
REFERENCES
1. Allen, T. M., & Cullis, P. R. (2013); Liposomal drug delivery systems: from concept to clinical applications; Adv Drug Deliv Rev; 65(1);36-48.
2. Atyabi, F., Farkhondehfai, A., Esmaeili, F., & Dinarvand, R. (2009); Preparation of pegylated nano-liposomal formulation containing SN-38: In vitro characterization and in vivo biodistribution in mice.; Acta Pharm; 59(2);133-144.
3. Barrajon-Catalan, E., Menendez-Gutierrez, M. P., Falco, A., Carrato, A., Saceda, M., & Micol, V. (2010); Selective death of human breast cancer cells by lytic immunoliposomes: Correlation with their HER2 expression level; Cancer Lett;290(2);192-203.
4. Bhatt P., Khatri N., Kumar M., Baradia D., & Misra A. (2015); Microbeads mediated oral plasmid DNA delivery using polymethacrylate vectors: an effectual groundwork for colorectal cancer; Drug Delivery; 22(6); 849-861.
5. Bhatt P., Lalani R., Mashru R., & Misra A. (2016); Abstract 2065: Anti-FSHR antibody Fab’ fragment conjugated immunoliposomes loaded with cyclodextrin-paclitaxel complex for improved in vitro efficacy on ovarian cancer cells; Cancer Research; 76(14 Supplement): 2065.
6. Bhatt P., Lalani R., Vhora I., Patil S., Amrutiya J., Misra A., & Mashru R. (2018); Liposomes encapsulating native and cyclodextrin enclosed paclitaxel: Enhanced loading efficiency and its pharmacokinetic evaluation; International journal of pharmaceutics; 536(1); 95-107.
7. Bhatt P., Narvekar P. (2018); Challenges and Strategies for Drug Transport across the Blood Brain Barrier; ARC Journal of Neuroscience; 3(3);17-21.
8. Bhatt P., Vhora I., Patil, S., Amrutiya J., Bhattacharya C., Misra A., & Mashru, R. (2016); Role of antibodies in diagnosis and treatment of ovarian cancer: Basic approach and clinical status; Journal of Controlled Release; 226; 148-167.
9. Dhankhar, R., Vyas, S. P., Jain, A. K., Arora, S., Rath, G., & Goyal, A. K. (2010); Advances in novel drug delivery strategies for breast cancer therapy; Artif Cells Blood Substit Immobil Biotechnol; 38(5);230-249.
10. Dinarvand, R., Varshochian, R., Kamalinia, G., Goodarzi, N., & Atyabi, F. (2013); Recent approaches to overcoming multiple drug resistance in breast cancer using modified liposomes; Clin. Lipidol; 8(4);391–394
11. Drummond, D. C., Noble, C. O., Guo, Z., Hayes, M. E., Connolly-Ingram, C., Gabriel, B. S., Kirpotin, D. B. (2010); Development of a highly stable and targetable nanoliposomal formulation of topotecan; J Control Release; 141(1); 13-21
12. Dua J, R. A., Bhandari A. . (2012); Liposome: methods of preparation and applications; Int. J. Pharm. Stud. Res.; 3(2);14–20
13. Egusquiaguirre, S. P., Igartua, M., Hernandez, R. M., & Pedraz, J. L. (2012); Nanoparticle delivery systems for cancer therapy: advances in clinical and preclinical research; Clin Transl Oncol; 14(2); 83-93
14. Elhazzat J, E.-S. M. (2010); Advances in targeted breast cancer therapy. . Curr. Breast Cancer Rep; 2(3); 146–151
15. Fishman, D. A., Cohen, L., Blank, S. V., Shulman, L., Singh, D., Bozorgi, K., . . . Schwartz, P. E. (2005) ; The role of ultrasound evaluation in the detection of early-stage epithelial ovarian cancer; Am J Obstet Gynecol; 192(4); 1214-1221
16. Gabizon, A., Catane, R., Uziely, B., Kaufman, B., Safra, T., Cohen, R., . . . Barenholz, Y. (1994); Prolonged circulation time and enhanced accumulation in malignant exudates of doxorubicin encapsulated in polyethylene-glycol coated liposomes; Cancer Res, 54(4); 987-992.
17. Gabizon, A. A. (1994); Liposomal anthracyclines. Hematol Oncol Clin North Am; 8(2); 431-450.
18. Gabizon, A. A., Shmeeda, H., & Zalipsky, S. (2006); Pros and cons of the liposome platform in cancer drug targeting; J Liposome Res, 16(3);175-183.
19. Gao, J., Liu, W., Xia, Y., Li, W., Sun, J., Chen, H., Guo, Y. (2011); The promotion of siRNA delivery to breast cancer overexpressing epidermal growth factor receptor through anti-EGFR antibody conjugation by immunoliposomes; Biomaterials; 32(13); 3459-3470.
20. Gao, J., Zhong, W., He, J., Li, H., Zhang, H., Zhou, G., . . . Zhong, Y. (2009); Tumor-targeted PE38KDEL delivery via PEGylated anti-HER2 immunoliposomes. International journal of pharmaceutics; 374(1-2); 145-152.
21. Gomes-da-Silva, L. C., Santos, A. O., Bimbo, L. M., Moura, V., Ramalho, J. S., Pedroso de Lima, M. C., Moreira, J. N. (2012); Toward a siRNA-containing nanoparticle targeted to breast cancer cells and the tumor microenvironment. International journal of pharmaceutics; 434(1-2); 9-19.
22. Govindarajan, S., Sivakumar, J., Garimidi, P., Rangaraj, N., Kumar, J. M., Rao, N. M., & Gopal, V. (2012); Targeting human epidermal growth factor receptor 2 by a cell-penetrating peptide-affibody bioconjugate; Biomaterials; 33(8); 2570-2582.
23. Haley, B., & Frenkel, E. (2008); Nanoparticles for drug delivery in cancer treatment. Urol Oncol; 26(1); 57-64.
24. Hortobagyi, G. N. (1998); Treatment of breast cancer; N Engl J Med; 339(14); 974-984.
25. J., O. S. (2003); Liposomal anthracyclines for breast cancer: overview; Oncologist; 8(Suppl. 2 ); 1–2
26. Johnson, S. W., Ozols, R. F., & Hamilton, T. C. (1993); Mechanisms of drug resistance in ovarian cancer; Cancer; 71(2 Suppl); 644-649.
27. Kikumori, T., Kobayashi, T., Sawaki, M., & Imai, T. (2009); Anti-cancer effect of hyperthermia on breast cancer by magnetite nanoparticle-loaded anti-HER2 immunoliposomes; Breast Cancer Res Treat; 113(3); 435-441.
28. Koning, G. A., & Storm, G. (2003); Targeted drug delivery systems for the intracellular delivery of macromolecular drugs; Drug Discov Today; 8(11); 482-483.
29. Kularatne, S. A., & Low, P. S. (2010); Targeting of nanoparticles: folate receptor ; Methods Mol Biol; 624; 249-265.
30. Kulkarni PR, Y. J., Vaidya KA. . (2011); Liposomes: a novel drug delivery system; Int. J. Curr. Pharm. Res; 3(2); 10–18
31. Kullberg, M., McCarthy, R., & Anchordoquy, T. J. (2014); Gene delivery to Her-2+ breast cancer cells using a two-component delivery system to achieve specificity. ; Nanomedicine;10(6); 1253-1262.
32. Lalani R. A., Bhatt P., Rathi M., & Misra A. (2016); Abstract 2063: Improved sensitivity and in vitro efficacy of RGD grafted PEGylated gemcitabine liposomes in RRM1 siRNA pretreated cancer cells; Cancer Research; 76(14 Supplement); 2063.
33. Lao, J., Madani, J., Puertolas, T., Alvarez, M., Hernandez, A., Pazo-Cid, R., . . . Anton Torres, A. (2013); Liposomal Doxorubicin in the treatment of breast cancer patients: a review; J Drug Deliv; 2013,; 456409.
34. Low, P. S., Henne, W. A., & Doorneweerd, D. D. (2008). Discovery and development of folic-acid-based receptor targeting for imaging and therapy of cancer and inflammatory diseases; Acc Chem Res; 41(1); 120-129.
35. Malam, Y., Loizidou, M., & Seifalian, A. M. (2009); Liposomes and nanoparticles: nanosized vehicles for drug delivery in cancer; Trends Pharmacol Sci; 30(11); 592-599.
36. Mansour, A. M., Drevs, J., Esser, N., Hamada, F. M., Badary, O. A., Unger, C., Kratz, F. (2003); A new approach for the treatment of malignant melanoma: enhanced antitumor efficacy of an albumin-binding doxorubicin prodrug that is cleaved by matrix metalloproteinase 2; Cancer Res; 63(14); 4062-4066.
37. Marchetti, C., Pisano, C., Facchini, G., Bruni, G. S., Magazzino, F. P., Losito, S., & Pignata, S. (2010); First-line treatment of advanced ovarian cancer: current research and perspectives. Expert Rev Anticancer Ther; 10(1); 47-60.
38. Moghimi, S. M., & Hunter, A. C. (2000); Poloxamers and poloxamines in nanoparticle engineering and experimental medicine; Trends Biotechnol; 18(10); 412-420.
39. Nomura, T., Saikawa, A., Morita, S., Sakaeda Kakutani, T., Yamashita, F., Honda, K., . . . Hashida, M. (1998); Pharmacokinetic characteristics and therapeutic effects of mitomycin C-dextran conjugates after intratumoural injection; J Control Release; 52(3), 239-252.
40. Park, E. K., Lee, S. B., & Lee, Y. M. (2005) ; Preparation and characterization of methoxy poly(ethylene glycol)/poly(epsilon-caprolactone) amphiphilic block copolymeric nanospheres for tumor-specific folate-mediated targeting of anticancer drugs; Biomaterials; 26(9); 1053-1061.
41. Park, J. W. (2002); Liposome-based drug delivery in breast cancer treatment; Breast Cancer Res; 4(3); 95-99.
42. Patel J., Amrutiya J., Bhatt P., Javia A., Jain M., & Misra, A. (2018); Targeted delivery of monoclonal antibody conjugated docetaxel loaded PLGA nanoparticles into EGFR overexpressed lung tumour cells; Journal of Microencapsulation; 35(2); 204-217.
43. Pathak Nandish , P. P. (2019); Applications of liposome in cancer drug delivery and treatment: A review Asian Journal of Pharmaceutical Research and Development; 7(1); 62-65.
44. Pathak N., P. Pathak. (2019); Nanoparticles and Target Drug Delivery for Cancer Treatment: A Comprehensive Review; International Journal of Drug Regulatory Affairs; 7(1); 53-58.
45. Patil S., Bhatt P., Lalani R., Amrutiya J., Vhora I., Kolte A., & Misra A. (2016); Low molecular weight chitosan–protamine conjugate for siRNA delivery with enhanced stability and transfection efficiency; RSC Advances; 6(112); 110951-110963.
46. Prabha, S., & Labhasetwar, V. (2004); Nanoparticle-mediated wild-type p53 gene delivery results in sustained antiproliferative activity in breast cancer cells; Mol Pharm, 1(3); 211-219.
47. Puri, A., Loomis, K., Smith, B., Lee, J. H., Yavlovich, A., Heldman, E., & Blumenthal, R. (2009). Lipid-based nanoparticles as pharmaceutical drug carriers: from concepts to clinic; Crit Rev Ther Drug Carrier Syst; 26(6); 523-580.
48. Rivera, E. (2003); Liposomal anthracyclines in metastatic breast cancer: clinical update; Oncologist; 8 (Suppl 2); 3-9.
49. Rom, J., Bechstein, S., Domschke, C., Golatta, M., Mayer, C., Heil, J., . . . Schneeweiss, A. (2014); Efficacy and toxicity profile of pegylated liposomal doxorubicin (Caelyx) in patients with advanced breast cancer; Anticancer Drugs, 25(2); 219-224.
50. Sawant, R. R., & Torchilin, V. P. (2010). Liposomes as ‘smart’ pharmaceutical nanocarriers. Soft Matter; 6(17); 4026-4044.
51. Seelig, A., & Gerebtzoff, G. (2006); Enhancement of drug absorption by noncharged detergents through membrane and P-glycoprotein binding. Expert Opin Drug Metab Toxicol, 2(5); 733-752.
52. Sharma, G., Anabousi, S., Ehrhardt, C., & Ravi Kumar, M. N. (2006); Liposomes as targeted drug delivery systems in the treatment of breast cancer; J Drug Target, 14(5); 301-310.
53. Suthar S. M., Rathva B. A. (2019; Development of liposomal formulation: from formulation to sterilization; World Journal of Pharmaceutical Research; 8(3);1561-1571.
54. Shroff, K., & Kokkoli, E. (2012); PEGylated liposomal doxorubicin targeted to alpha5beta1-expressing MDA-MB-231 breast cancer cells; Langmuir; 28(10); 4729-4736.
55. Strebhardt, K., & Ullrich, A. (2008); Paul Ehrlich's magic bullet concept: 100 years of progress. Nat Rev Cancer, 8(6); 473-480
56. Tandel H., Bhatt P., Jain K., Shahiwala A., & Misra A. (2018); In-Vitro and In-Vivo Tools in Emerging Drug Delivery Scenario: Challenges and Updates. In: Misra ASA, editor. In-vitro and in-vivo tools in drug delivery research for optimum clinical outcomes. Boca Raton: CRC Press;19-42.
57. Thomas, H., & Coley, H. M. (2003); Overcoming multidrug resistance in cancer: an update on the clinical strategy of inhibiting p-glycoprotein; Cancer Control; 10(2);159-165.
58. Torchilin, V. P. (2005); Recent advances with liposomes as pharmaceutical carriers; Nat Rev Drug Discov; 4(2); 145-160.
59. Vhora I., Patil S., Bhatt P., Gandhi R., Baradia D., & Misra A. (2014); Receptor-targeted drug delivery: current perspective and challenges; Ther Delivery; 5(9); 1007-1024.
60. Vhora I., Patil S., Bhatt P., & Misra A. (2015); Protein- and Peptide-drug conjugates: an emerging drug delivery technology; Adv Protein Chemistry Structural Biology; 98; 1-55.
61. Voinea, M., & Simionescu, M. (2002); Designing of 'intelligent' liposomes for efficient delivery of drugs; J Cell Mol Med; 6(4); 465-474.
62. Yewale C., Baradia D., Patil S., Bhatt P., Amrutiya J., Gandhi R.,Misra A. (2018); Docetaxel loaded immunonanoparticles delivery in EGFR overexpressed breast carcinoma cells; Journal of Drug Delivery Science and Technology; 45; 334-345.
63. Zhou, T., Duan, J., Wang, Y., Chen, X., Zhou, G., Wang, R.,Xu, F. (2012). Fluoxetine synergys with anticancer drugs to overcome multidrug resistance in breast cancer cells. ; Tumour biology : the journal of the International Society for Oncodevelopmental Biology and Medicine; 33(5);1299-1306.
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT editor-in-chief@pharmatutor.org
Subscribe to Pharmatutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE










