{ DOWNLOAD AS PDF }
ABOUT AUTHORS
SPANDANA. M*, VAISHNAVI. M, PRAVALIKA. P, SUSMITHA. S, AMATUL ALI SAMEERA
Dept. of Pharmacy Practice
Sree Dattha Institute of Pharmacy,
Sheriguda (V), Ibrahimpatnam (M), Ranga Reddy, Telangana, India
ABSTRACT
AIMS AND OBJECTIVES: Our aim is to assess the clinical usage of Nebulization and Antibiotic therapy in COPD.
METHOD: An Observational, Prospective, Cohort study was conducted at a tertiary care teaching hospital, Telangana State, India. A total of 102 patients from the In-patient department of Pulmonology in Gleneagles Global Hospital, who were prescribed Nebulizers and Antibiotics and those who fulfilled the exclusion and inclusion criteria were selected for the study which was conducted for 6 months. All information significant to the study was collected from the case records and discussion conducted with the In-patients and bystanders during ward rounds with the support of a physician which were analyzed by SPSS software. Moreover daily follow-ups were conducted to assemble data on amendment in therapy, add on therapy, and clinical improvement.
RESULTS: The Mean age was 66 years and Standard Deviation was 13 years of the population. 41% were smokers and 27% were alcoholics. The most commonly used Bronchodilator is Duolin with a percentage of 96%. The most commonly used antibiotic is Magnex Forte with a percentage of 41.1%. The most commonly used formulation is Orals with a percentage of 53%. 92% of individuals responded in a positive way after the Nebulization therapy with Oxygen therapy provided to them, as their SpO 2 levels with oxygen therapy changed to normal range.
CONCLUSION: Nebulization therapy had more clinical improvement when compared to Inhalational therapy. The addition of corticosteroids had decreased the patient stay in hospital. Antibiotics added to the treatment decreased the frequency of exacerbations. Antibiotics added to the treatment decreased the frequency of exacerbations. The patient should be educated about the rational use of the drugs and decrease the risk and exacerbations of COPD.
[adsense:336x280:8701650588]
Reference Id: PHARMATUTOR-ART-2628
|
PharmaTutor (Print-ISSN: 2394 - 6679; e-ISSN: 2347 - 7881) Volume 6, Issue 12 Received On: 15/09/2018; Accepted On: 05/11/2018; Published On: 01/12/2018 How to cite this article: Spandana, M., Vaishnavi, M., Pravalika, P., Susmitha, S. and Sameera, A.A. 2018. Assess the Clinical Usage of Nebulization Therapy and Antibiotics in In-Patients with Chronic Obstructive Pulmonary Disease: A Randomized Prospective Study at a Tertiary Care Teaching Hospital. PharmaTutor. 6, 12 (Dec. 2018), 10-40. DOI:https://doi.org/10.29161/PT.v6.i12.2018.10 |
INTRODUCTION
Chronic Obstructive Pulmonary Disease (COPD) is a major cause of chronic morbidity and mortality throughout the world. Many people suffer from this disease for years and die prematurely from it or its complications. COPD is the fourth leading cause of death in the world (Lozano R et al., 2012), and further increases in its prevalence and mortality can be predicted in the coming decades (Lopez AD et al., 2006). COPD is a major global health problem that increasingly constitutes a burden for the society and has large effects on health-care expenditure (Pauwels RA et al., 2001). The most recent definition of COPD according to GOLD is as follows: “Chronic obstructive pulmonary disease (COPD) is characterized by chronic airflow limitation and a range of pathological changes in the lung, some significant extra-pulmonary effects, and important comorbidities which may contribute to the severity of the disease in individual patients” (Mary Anne Koda-Kimble et al., 2005).
Chronic obstructive pulmonary disease (COPD) is now the fourth leading cause of death globally, and the World Health Organization (WHO) has predicted that it will become the third most common cause of death in the world by 2030 (World Health Organization., 2008). In developed countries, current information estimates a prevalence of 8% to 10% among adults 40 years of age and older, whereas in developing countries, prevalence varies significantly among countries and is difficult to quote (Diaz-Guzman E et al., 2014). It is estimated that more than 210 million people have the disease worldwide (Bousquet J et al., 2010). Concerning a large number of subjects, COPD generates important health and social costs. However, although COPD is one of the most common chronic diseases and has a high health and social impact, it is still poorly recognized among the general public and also clinicians. Consequently, there is a major and urgent need to better understand this complex disease (Barnes PJ et al., 2014).
India also has had the ignominy of experiencing the "highest loss in potentially productive years of life" worldwide in 2005 (Reddy KS et al., 2005). Crude estimates suggest there are 30 million COPD patients in India (Salvi S et al., 2012). India contributes a significant and growing percentage of COPD mortality which is estimated to be amongst the highest in the world, i.e. more than 64.7 estimated age standardized death rate per 100,000 amongst both sexes (Lopez AD et al., 2006).
Mortality from COPD is higher in males and increases with age in those over 45 yrs old. In the USA, within the framework of the National Health and Nutrition Examination Survey (NHANES I), 1,301 deaths were analysed in a cohort of 5,542 adults. The presence of COPD at various GOLD stages was associated with the following risk of death, as defined by hazard ratio (HR) (Lius S et al., 2007). Patients with COPD typically have comorbid conditions, such as lung cancer, cardiovascular disease (e.g. ischemic heart disease, depression, muscle wasting, reduced fat-free mass, osteopenia, and chronic infections). One explanation for this is that tobacco use drives disease in multiple organs. These disorders contribute to a high disease burden and early mortality in patients with COPD. Deaths in individuals with COPD are frequently attributed to a cause other than COPD (Mannino DM. AS Buist, 2007).
Active smoking is the main risk factor for COPD. The risk attributable to active smoking in COPD is thought to vary from 40% to 70% according to the country (Boutin-Forzano S et al., 2007). Exposure to pollution inside the home, in particular in developing countries, is an important risk for COPD (risk accounting for 35% of cases), due to exposure to smoke when cooking in particular for females (Postma DS et al., 1998). From an epidemiological point of view, males were classically more at risk of developing COPD in comparison with females because of their smoking habits. Progressively, however, and depending on the country, females who smoke as much as males seem to have an equivalent risk of developing COPD (Liu S et al., 2007; Mannino DM. AS Buist, 2007).
Exposure to biomass fuels like crop residues or woods or animal dung is also widely prevalent in India. More than one-half of the world's households uses biomass fuels (Shohaimi S et al., 2004). Biomass fuels are now considered a major cause of the causation of COPD (deKoning HW et al., 1985; Salvi S. Barnes PJ, 2010). In India, 70% of the homes use biomass fuel for cooking and heating purposes in poorly ventilated kitchens, and the amount of particulate matter pollution generated by the burning of biomass fuel is extremely high. 90% of rural households and 32% of urban households cook their meals on a biomass stove (Salvi S. Barnes PJ, 2009; Prasad R et al., 2012).
Because no curative therapy is available, management of severe exacerbations of COPD should be directed at relieving symptoms and restoring functional capacity (International Institute of Population Sciences., 2007). A major part of the GOLD report is devoted to the clinical Management of COPD and presents a management plan with four components:
• Assess and Monitor Disease
• Reduce Risk Factors
• Manage Stable COPD
• Manage Exacerbations (Lozano R et al., 2012).
These strategies include β2 agonists, the addition of anticholinergics, the intravenous administration of corticosteroids, antibiotic therapy when indicated, and the intravenous administration of methylxanthines such as aminophylline (Standards for the diagnosis and care of patients with COPD).
Hospitalization of patients with COPD may be necessary to provide antibiotic therapy, appropriate supportive care and monitoring of oxygen status. Oxygen supplementation via external devices or mechanical ventilation may be indicated to maintain oxygen delivery to vital tissues (Voelkel NF. Tuder R, 2000). Antibiotic therapy has been shown to have a small but important effect on clinical recovery and outcome in patients with acute exacerbations of chronic bronchitis and emphysema (Fein A. Fein AM, 2000). Therefore, antibiotic administration should be considered at the beginning of treatment for exacerbations of COPD. Oxygen status can be monitored clinically, as well as by pulse oximetry (Voelkel NF. Tuder R, 2000).
Nebuliser therapy is frequently prescribed for management of chronic obstructive pulmonary disease (COPD) (Saint S et al., 1995; O’Driscoll R, 1991). About half of the patients who remain breathless despite high-dose bronchodilators delivered by pressurised metered dose inhalers (pMDIs) or dry powder inhalers (DPIs), derive benefits from domiciliary nebulizer therapy (Jarvis S et al., 2007). In any case, although long-acting bronchodilators have proved to be the most effective therapies so far available (Cazzola M. Matera MG, 2014), there are still fundamental questions regarding their use that require clarification to optimise utilisation of these drugs (Cazzola M. Page C, 2014). It is not know yet if it is appropriate to treat all COPD patients with long-acting bronchodilators, it is better to start with a β2-agonist or with an anti-muscarinic agent, it is useful to use a bronchodilator with a rapid onset of action, once or twice daily dosing is preferable (Barnes PJ, 2010). This can include the very young, the elderly, others who are simply unable to use an inhaler correctly, and those undergoing an exacerbation. Nebulizers have undoubted benefits including:
• They are able to provide a much larger dose of medication and can be used for not only bronchodilators and corticosteroids, but a wide range of antibiotics and other medications.
• They require less intensive training for elderly chronic obstructive pulmonary disease (COPD) patients and are easier to use than inhalers (O’Driscoll BR et al., 1992).
LITERATURE REVIEW
DEFINITION
Chronic obstructive pulmonary disease (COPD) is characterized by chronic airflow limitation and a range of pathological changes in the lung, some significant extra pulmonary effects, and important co-morbidities which may contribute to the severity of the disease in individual patients. Thus, COPD should be regarded as a pulmonary disease, but these significant co morbidities must be taken into account in a comprehensive diagnostic assessment of severity and in determining appropriate treatment.
Based on current knowledge, a working definition is: Chronic Obstructive Pulmonary Disease (COPD) is a preventable and treatable disease with some significant extra pulmonary effects that may contribute to the severity in individual patients. It’s pulmonary component is characterized by airflow limitation that is not fully reversible. The airflow limitation is usually progressive and associated with an abnormal inflammatory response of the lung to noxious particles or gases (Soriano JB et al., 2005).
EPIDEMIOLOGY
Chronic obstructive pulmonary disease is an extremely important cause of morbidity and mortality. For example, the prevalence of COPD more than doubled between 1990 and 2002, making it the fourth leading cause of death in the United States. Furthermore, it is projected to be the third leading cause of death by the year 2020. This increase in prevalence of COPD is felt to be related to the aging of the population and past smoking behavior, because COPD death rates and prevalence lag behind smoking rates by several decades. Because the smoking rates for women peaked later than rates for men, the increase in COPD prevalence in the United States is related primarily to an increase in COPD among women.
The economic impact of COPD is also significant, with estimated annual treatment costs exceeding $30 billion. This includes health care expenditures of $18 billion and indirect costs of $14 billion (e.g., lost earnings because of illness or early death).
A major cause of disability, COPD currently is considered the 11th leading cause of disability worldwide. Current projections suggest that by the year 2020, COPD will be the 5th leading cause of disability worldwide, behind only ischemic heart disease, major depression, traffic accidents, and cerebrovascular disease (Mary Anne Koda-Kimble et al., 2005).
COPD is a leading cause of morbidity and mortality worldwide and results in an economic and social burden that is both substantial and increasing. COPD prevalence, morbidity, and mortality vary across countries and across different groups within countries but, in general, are directly related to the prevalence of tobacco smoking although in many countries, air pollution resulting from the burning of wood and other biomass fuels has also been identified as a COPD risk factor (Tirimanna PR et al., 1996).
Morbidity: Morbidity measures traditionally include physician visits, emergency department visits, and hospitalizations.
Although COPD databases for these outcome parameters are less readily available and usually less reliable than mortality databases, the limited data available indicate that morbidity due to COPD increases with age and is greater in men than in women (Morbidity & Mortality: Chart book 1998; Soriano JR et al., 2000; Chapman KR, 2004). In these data sets, however, COPD in its early stages (Stage I: Mild COPD and Stage 2: Moderate COPD) is usually not recognized, diagnosed, or treated, and therefore may not be included as a diagnosis in a patient’s medical record.
Morbidity from COPD may be affected by other co-morbid chronic conditions (Schellevis FG et al., 1994) (e.g., musculoskeletal disease, diabetes mellitus) that are not directly related to COPD but nevertheless may have an impact on the patient’s health status, or may negatively interfere with COPD management. In patients with more advanced disease (Stage III: Severe COPD and Stage IV: Very Severe COPD), morbidity from COPD may be misattributed to another co morbid condition.
Another way of estimating the morbidity burden of disease is to calculate years of living with disability (YLD). The Global Burden of Disease Study estimates that COPD results in 1.68 YLD per 1,000 population, representing 1.8% of all YLDs, with a greater burden in men than in women (1.93% vs 1.42%) (Lopez AD et al., 2006; Murray CJL. Lopez AD, 1996; Murray CJ. Lopez AD 1997).
Prevalence: Existing COPD prevalence data show remarkable variation due to differences in survey methods, diagnostic criteria, and analytic approaches (Halbert RJ et al., 2006; Halbert RJ et al., 2003). Survey methods can include:
• Self-report of a doctor diagnosis of COPD or equivalent condition
• Spirometry with or without a bronchodilator
• Questionnaires that ask about the presence of respiratory symptoms
The lowest estimates of prevalence are usually those based on self-reporting of a doctor diagnosis of COPD or equivalent condition. For example, most national data show that less than 6% of the population has been told that they have COPD. This likely reflects the widespread under recognition and under diagnosis of COPD as well as the fact that those with Stage I: Mild COPD may have no symptoms, or else symptoms (such as chronic cough and sputum) that are not perceived by individuals or their health care providers as abnormal and possibly indicative of early COPD (van den Boom G et al., 1998).
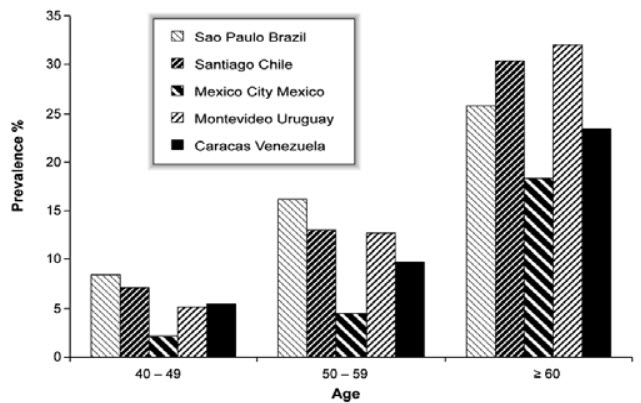
Prevalence = post bronchodilator FEV1/FVC < 0.70 (Stage I: Mild COPD and higher)
Figure 1: COPD Prevalence by Age in Five Latin American Cities
It is recognized that defining irreversible airflow obstruction as a post bronchodilator FEV1/FVC ratio less than 0.70 leads to the potential for significant misclassification, with under diagnosis (false negatives) in younger adults and over diagnosis (false positives) over age 50 years (Hnizdo E et al., 2006; Roberts SD et al., 2006; Celli BR et al 2003).
Many additional sources of variation can affect estimates of COPD prevalence, including sampling methods, response rates, quality control of spirometry, and whether spirometry is performed pre or post bronchodilator. Samples that are not population-based and poor response rates may give biased estimates of prevalence, with the direction of bias sometimes hard to determine (Sterk PJ, 2004).
The Latin American Project for the Investigation of Obstructive Lung Disease (PLATINO) examined the prevalence of post-bronchodilator airflow limitation (Stage I: Mild COPD and higher) among persons over age 40 in five major Latin American cities each in a different country-Brazil, Chile, Mexico, Uruguay, and Venezuela. In each country, the prevalence of Stage I: Mild COPD and higher increased steeply with age, with the highest prevalence among those over 60 years, ranging from a low of 18.4% in Mexico City, Mexico to a high of 32.1% in Mentevideo, Uruguay. In all cities/countries the prevalence was appreciably higher in men than in women (Menezes AM et al., 2005).
Mortality: The World Health Organization publishes mortality statistics for selected causes of death annually for all WHO regions, additional information is available from the WHO Evidence for Health Policy Department.
Despite the problems with the accuracy of the COPD mortality data, it is clear that COPD is one of the most important causes of death in most countries. The Global Burden of Disease Study has projected that COPD, which ranked sixth as the cause of death in 1990, will become the third leading cause of death worldwide by 2020 (Lopez AD et al., 2006; Murray CJL. Lopez AD, 1996; Murray CJ. L Lopez AD, 1997).
Trends in mortality rates over time provide further important information but, again, these statistics are greatly affected by terminology, awareness of the disease, and potential gender bias in its diagnosis (Jemal A et al., 2005).
ETIOLOGY
Smoking is the best-studied COPD risk factor, it is not the only one and there is consistent evidence from epidemiologic studies that nonsmokers may develop chronic airflow obstruction (Celli BR et al., 2005; Behrendt CE, 2005).
Genes: COPD is a polygenic disease. The genetic risk factors severe hereditary deficiency of alpha-1 antitrypsin, a major circulating inhibitor of serine proteases (Stoller JK et al., 2005). A significant familial risk of airflow obstruction has been observed in smoking siblings of patients with severe COPD. Suggesting that genetic factors could influence this susceptibility (McCloskey SC et al., 2001).
Inhalational Exposures: Each type of particle, depending on its size and composition, may contribute a different weight to the risk, and the total risk will depend on the integral of the inhaled exposures. Of the many inhalational exposures that may be encountered over a lifetime, only tobacco smoke, occupational dusts and chemicals are known to cause COPD on their own (US Surgeon General. The health consequences of smoking: chronic obstructive pulmonary disease 1984; Burrows B et al., 1977).
Tobacco Smoke: Cigarette smoking is by far the most commonly encountered risk factor for COPD. Cigarette smokers have a higher prevalence of respiratory symptoms and lung function abnormalities, a greater annual rate of decline in FEV1, and a greater COPD mortality rate than nonsmokers (Jindal SK et al., 2006).
Oxidative Stress: The lungs are continuously exposed to endogenous and exogenous oxidants. In addition, intracellular oxidants, such as those derived from mitochondrial electron transport, are involved in many cellular signaling pathways. Lung cells are protected against this oxidative challenge by well-developed enzymatic and non-enzymatic systems. When the balance between oxidants and antioxidants shifts oxidative stress occurs (MacNee W et al., 2005).
Gender: The role of gender in determining COPD risk remains unclear (US Surgeon General, 1984). In the past, most studies showed that COPD prevalence and mortality were greater among men than women. Studies from developed countries show that the prevalence of the disease is now almost equal in men and women, which probably reflects changing patterns of tobacco smoking. Some studies have suggested that women are more susceptible to the effects of tobacco smoke than men (XuX et al., 1994; Anthonisen NR et al., 1994).
PATHOPHYSIOLOGY
Alterations in tissue structure are caused by chronic inflammation that involves recruitment of inflammatory cells into the lung, as well as structural changes that result from repeated injury and tissue remodeling. The magnitude of structural changes and tissue dysfunction varies among patients and is often distinguished by two major phenotypes that include chronic bronchitis and emphysema. The clinical definition for chronic bronchitis is “the presence of chronic productive cough for 3 months in each of 2 consecutive years in a patient in whom other causes of chronic cough (e.g., asthma, congestive heart failure, gastroesophageal reflux) have been excluded.” Emphysema is described as “abnormal permanent enlargement of the airspaces distal to the terminal bronchioles, accompanied by destruction of their walls and without obvious fibrosis.”
Large (Central) and Small (Peripheral) Airways: The large airways, which include the trachea and first generations of the bronchi, are a major site of inflammation and mucus hypersecretion. This results from an increase in numbers (hyperplasia) and enlargement (hypertrophy) of the sub mucosal glands and mucus producing goblet cells within the surface epithelium. Overproduction of mucus in the large airways results in a chronic productive cough, as observed in chronic bronchitis, but this does not have a major impact on airflow limitation. Mucus hypersecretion coupled with impaired ciliary function, reduces mucociliary clearance, increases the accumulation of secretions, and enhances the risk of bacteria colonization. As a result, recurrent infections often occur.
The peripheral airway is composed of the smaller bronchi down to the terminal bronchioles, the smallest branches in the lung that are not involved in gas exchange. The extent of inflammation, fibrosis, and airway exudate in the peripheral airway correlates best with the reduction in airflow as measured by a decrease in FEV1 or the FEV1/FVC ratio. Chronic obstruction of the peripheral airway also results in air trapping on expiration, which contributes to hyperinflation. It is now known that hyperinflation can occur early in the course of COPD and is an important cause of exertional dyspnea.
Parenchymal Destruction: The terminal bronchioles lead directly to the alveolar ducts and sacs, the major site where gas exchange occurs. Emphysema is characterized by the destructive loss of alveolar walls and enlargement of the terminal airspaces resulting in a loss of gas-exchanging surface area. In advanced cases, large, balloon-shaped bullous lesions may develop. Rupture of these bullae can lead to collapse of lung segments (pneumothorax).
Significant Comorbid Illness: In the later stages of disease, chronic hypoxemia causes persistent vasoconstriction in the lung vascular bed, particularly the small pulmonary arteries. This can result in permanent structural alteration of the blood vessels causing intimal hyperplasia and smooth muscle hypertrophy. The loss of pulmonary capillaries in emphysema can also contribute to increased pulmonary vasculature pressures. The cumulative impact of vascular changes can result in progressive pulmonary hypertension and eventually, right-sided cardiac failure.
The clinical consequences include progressive dyspnea on exertion, chronic cough and sputum production, increased risk for respiratory infections.
Alpha-1 Antitrypsin Deficiency: The best characterized anti protease in the lung is α1antitrypsin. This serum glycoprotein is primarily produced in the liver and works by binding to and neutralizing proteases. Cigarette smoke can activate and attract inflammatory cells into the lung, there by promoting the release of proteases. Cigarette smoke can also inactivate endogenous protease inhibitors, including α1-antitrypsin, further supporting protease activity. This risk of tissue damage is greatly accentuated in patients with α1-antitrypsin deficiency (Mary Anne Koda-Kimble et al., 2005).
SIGNS AND SYMPTOMS
Dyspnea: Dyspnea, the hallmark symptom of COPD, is the reason most patients seek medical attention. Typical COPD patients describe their dyspnea as a sense of increased effort to breathe, heaviness, air hunger or gasping (Simon PM et al., 1990). However, the terms used to describe dyspnea vary both by individual and by culture (Elliott MW et al., 1991).
Cough: Chronic cough, often the first symptom of COPD to develop, is often discounted by the patient as an expected consequence of smoking and/or environmental exposures. Initially, the cough may be intermittent, but later is present every day, often throughout the day. The chronic cough in COPD may be unproductive. In some cases, significant airflow limitation may develop without the presence of a cough (Georgopoulas D. Anthonisen NR, 1991).
Sputum Production: COPD patients commonly raise small quantities of tenacious sputum after coughing bouts (Definition and classification of chronic bronchitis for clinical and epidemiological purposes, 1965). Sputum production is often difficult to evaluate because patients may swallow sputum rather than expectorate it. The presence of purulent sputum reflects an increase in inflammatory mediators, and its development may identify the onset of an exacerbation (Hill AT et al., 1999).
Wheezing and Chest Tightness: Wheezing and chest tightness are nonspecific symptoms that may vary between days, and over the course of a single day. These symptoms may be present in Stage I: Mild COPD or Stage III: Severe COPD and Stage IV: Very Severe COPD. Audible wheeze may arise at a laryngeal level and need not be accompanied by auscultatory abnormalities. Alternatively, widespread inspiratory or expiratory wheezes can be present on listening to the chest. An absence of wheezing or chest tightness does not exclude a diagnosis of COPD, nor does their presence confirm a diagnosis of asthma (Schols AM et al., 1993).
Inspection:
• Central cyanosis, or bluish discoloration of the mucosal membranes, may be present but is difficult to detect in artificial light and in many racial groups.
• Common chest wall abnormalities, which reflect the pulmonary hyperinflation seen in COPD, include relatively horizontal ribs, “barrel-shaped” chest, and protruding abdomen.
• Flattening of the hemi-diaphragms may be associated with paradoxical in-drawing of the lower rib cage on inspiration, and widening of the xiphosternal angle.
• Resting respiratory rate is often increased to more than 20 breaths per minute and breathing can be relatively shallow (Loveridge B et al., 1986).
• Patients commonly show pursed-lip breathing, which may serve to slow expiratory flow and permit more efficient lung emptying (Bianchi R et al., 2004).
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT editor-in-chief@pharmatutor.org
Subscribe to Pharmatutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE
DIAGNOSIS
Chest X-ray: An abnormal chest X-ray is seldom diagnostic in COPD unless obvious bullous disease is present, but it is valuable in excluding alternative diagnoses and establishing the presence of significant comorbidities. However, when there is doubt about the diagnosis of COPD, high resolution CT (HRCT) scanning might help in the differential diagnosis. In addition, if a surgical procedure such as lung volume reduction is contemplated, a chest CT scan is necessary since the distribution of emphysema is one of the most important determinants of surgical suitability (Pellegrino R et al., 2005).
Arterial blood gas measurement: In advanced COPD, measurement of arterial blood gases while the patient is breathing air is important. This test should be performed in stable patients with FEV1<50% predicted or with clinical signs suggestive of respiratory failure or right heart failure. The inspired oxygen concentration (FiO2-normally 21% at sea level) should be noted, a particularly important point if patient is using an O2-driven nebulizer. Changes in arterial blood gas tensions take time to occur, especially in severe disease. Thus, 20-30 minutes should pass before rechecking the gas tensions when the FiO2 has been changed.
Measurement of Airflow Limitation (Spirometry): Spirometry should be undertaken in all patients who may have COPD. Although spirometry does not fully capture the impact of COPD on a patient’s health, it remains the gold standard for diagnosing the disease and monitoring its progression.
Spirometry should measure the volume of air forcibly exhaled from the point of maximal inspiration (forced vital capacity, FVC) and the volume of air exhaled during the first second of this maneuver (forced expiratory volume in one second, FEV1), and the ratio of these two measurements (FEV1/FVC) should be calculated. Spirometry measurements are evaluated by comparison with reference values based on age, height, sex, and race (Fishman A et al., 2003).
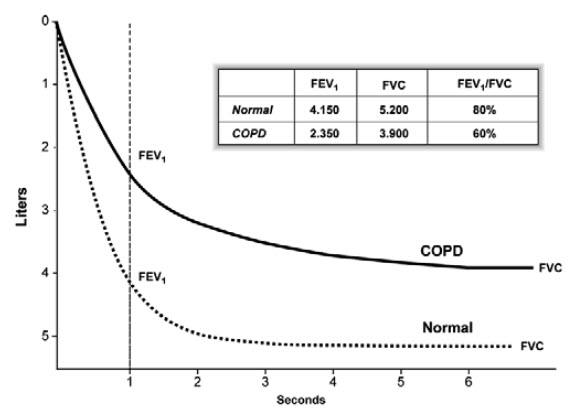
Figure 2: Normal Spirogram and Spirogram Typical of Patients with Mild to Moderate COPD
Alpha-1 antitrypsin deficiency screening: It may be valuable to identify coexisting alpha-1 antitrypsin deficiency. A serum concentration of alpha-1 antitrypsin below 15-20% of the normal value is highly suggestive of homozygous alpha-1 antitrypsin deficiency (Roberts CM et al., 1993).
ASSESSMENT
The goals of COPD assessment are to determine the severity of airflow limitation, its impact on the patient’s health status and the risk of future events (such as exacerbations, hospital admissions or death), guide therapy. To achieve these goals, COPD assessment must consider the following aspects of the disease separately:
• The presence and severity of the spirometric abnormality.
• Current nature and magnitude of the patient’s symptoms.
• Exacerbation history and future risk.
• Presence of comorbidities.
Classification of severity of airflow obstruction: The classification of airflow limitation severity in COPD is shown in following table. Spirometry should be performed after the administration of an adequate dose of at least one short-acting inhaled bronchodilator in order to minimize variability.
Table 1: Classification Of Airflow Limitation Severity In COPD

Assessment of symptoms: In the past, COPD was viewed as a disease largely characterized by breathlessness. A simple measure of breathlessness such as the Modified British Medical Research Council (mMRC) Questionnaire was considered adequate, as the mMRC relates well to other measures of health status and predicts future mortality risk.
Table 2: Modified MRC Dyspnea Scale
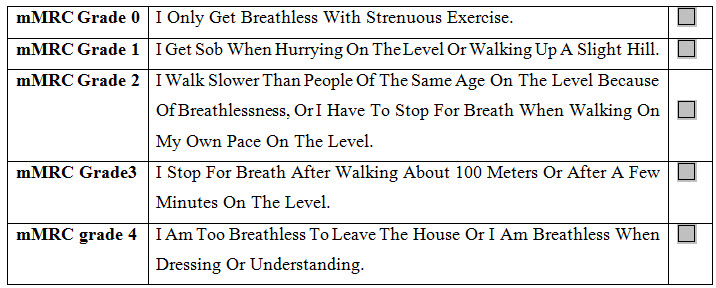
However, it is now recognized that COPD impacts patients beyond just dyspnea. For this reason, a comprehensive assessment of symptoms is recommended using measures such as the COPD Assessment Test (CAT) and the COPD Control Questionnaire (The CCQ) have been developed and are suitable.
TREAMENT
Pharmacologic therapy is used to prevent and control symptoms, reduce the frequency and severity of exacerbations, improve health status, and improve exercise tolerance.
No single pharmacologic class has been proven to provide superior benefit over others, although inhaled therapy is generally preferred. Medication selection is based on likely patient adherence, individual response, and side effects.
Sympathomimetics: β2-Selective sympathomimetics cause relaxation of bronchial smooth muscle and bronchodilation by stimulating the enzyme adenyl cyclase to increase the formation of cyclic adenosine monophosphate. They may also improve mucociliary clearance. The inhalation route is preferred to the oral and parenteral routes in terms of both efficacy and adverse effects.
Short-acting agents can be used for acute relief of symptoms or on a scheduled basis to prevent or reduce symptoms. The duration of action of short-acting β2-agonists is 4 to 6 hours. Long-acting inhaled β2-agonists that are dosed every 12 hours on a scheduled basis and provide bronchodilation throughout the dosing interval.
Anticholinergics: When given by inhalation, anticholinergic agents produce bronchodilation by competitively inhibiting cholinergic receptors in bronchial smooth muscle. This activity blocks acetylcholine, with the net effect being a reduction in cyclic guanosine monophosphate, which normally acts to constrict bronchial smooth muscle.
Ipratropium bromide has a slower onset of action than short-acting β2-agonists. But it is often prescribed as Ipratropium has a more prolonged bronchodilator effect than short-acting ß2-agonists. Its peak effect occurs in 1.5 to 2 hours and its duration is 4 to 6 hours.
Combination Anticholinergics and Sympathomimetics: The combination of an inhaled anticholinergic and β2-agonist is often used, especially as the disease progresses and symptoms worsen over time. Combination of both short- and long acting β2-agonists with ipratropium has been shown to provide added symptomatic relief and improvements in pulmonary function. A combination product containing albuterol and ipratropium (Combivent) is available as an MDI (metered-dose inhaler) for chronic maintenance therapy of COPD.
Methylxanthines: Theophylline and aminophylline may produce bronchodilation by inhibition of phosphodiesterase, thereby increasing cyclic adenosine monophosphate levels and inhibition of release of mediators from mast cells and leukocytes. Chronic theophylline use in COPD has been shown to produce improvements in lung function, including vital capacity and FEV1. Subjectively, theophylline has been shown to reduce dyspnea, increase exercise tolerance, and improve respiratory drive. Methylxanthines are no longer considered first-line therapy for COPD, because of theophylline’s risk for drug interactions and the interpatient variability in dosage requirements. Theophylline may be considered in patients who are intolerant or unable to use an inhaled bronchodilator. As with other bronchodilators in COPD, parameters other than objective measurements such as FEV1 should be monitored to assess efficacy. The role of theophylline in COPD is as maintenance therapy in non acutely ill patients. Therapy can be initiated at 200 mg twice daily and titrated upward every 3 to 5 days to the target dose.
Corticosteroids: The anti-inflammatory mechanisms whereby corticosteroids exert their beneficial effect in COPD include reduction in capillary permeability to decrease mucus, inhibition of release of proteolytic enzymes from leukocytes, and inhibition of prostaglandins. The clinical benefits of systemic corticosteroid therapy in the chronic management of COPD are often not evident, and there is a high risk of toxicity. Appropriate situations to consider corticosteroids in COPD include:
i. Short term systemic use for acute exacerbations.
ii. Inhalation therapy for chronic stable COPD.
Consensus guidelines indicate that inhaled corticosteroid therapy should be considered for symptomatic patients with stage III or IV disease (FEV1 less than 50%) who experience repeated exacerbations despite bronchodilator therapy. Combination therapy with salmeterol plus fluticasone or formoterol plus budesonide is associated with greater improvements in FEV1, health status, and exacerbation frequency than either agent alone (Barbara G. Wells et al., 2009).
CHRONIC OBSTRUCTIVE PULMONARY DISEASE EXACERBATION
An exacerbation of COPD is defined as an event in the natural course of the disease characterized by a change in the patient’s baseline dyspnea, cough and/or sputum that is beyond normal day-to-day variations is acute in onset, and may warrant a change in regular medication in a patient with underlying COPD (Rodriguez-Roisin, 2000; Burge S. Wedzicha JA, 2003).
Controlled oxygen therapy: Oxygen therapy is the cornerstone of hospital treatment of COPD exacerbations. Supplemental oxygen should be titrated to improve the patient’s hypoxemia. Adequate levels of oxygenation (PaO2 > 8.0 kPa, 60 mm Hg, or SaO2 > 90%) are easy to achieve in uncomplicated exacerbations, but CO2 retention can occur insidiously with little change in symptoms. Once oxygen is started, arterial blood gases should be checked 30-60 minutes later to ensure satisfactory oxygenation without CO2 retention or acidosis (Celli BR. MacNee W, 2004).
Bronchodilator therapy: Short-acting inhaled β2-agonists are usually the preferred bronchodilators for treatment of exacerbations of COPD. If a prompt response to these drugs does not occur, the addition of an anticholinergic is recommended. Methylxanthines (theophylline or aminohylline) is currently considered second-line intravenous therapy, used when there is inadequate or insufficient response to short-acting bronchodilators (Celli BR. MacNee W, 2004; National Institute for Clinical Excellence 2004; Barbera JA et al., 1992).
Glucocorticosteroids: Oral or intravenous glucocorticosteroids are recommended as an addition to other therapies in the hospital management of exacerbations of COPD. 30 to 40 mg of oral prednisolone daily for 7-10 days is effective and safe. Prolonged treatment does not result in greater efficacy and increases the risk of side effects (e.g., hyperglycemia, muscle atrophy) (Davies L et al., 1999; Niewoehner DE et al., 1999).
Antibiotics: Randomized placebo-controlled studies of antibiotic treatment in exacerbations of COPD have demonstrated a small beneficial effect of antibiotics on lung function (Saint S et al., 1995) and a randomized controlled trial has provided evidence for a significant beneficial effect of antibiotics in COPD patients who presented with an increase in all three of the following cardinal symptoms: dyspnea, sputum volume, and sputum purulence. There was also some benefit in those patients with an increase in only two of these cardinal symptoms (Anthonisen NR et al., 1987).
Ventilatory support: The primary objectives of mechanical ventilatory support in patients with COPD exacerbations are to decrease mortality and morbidity and to relieve symptoms. Ventilatory support includes both noninvasive intermittent ventilation using either negative or positive pressure devices, and invasive (conventional) mechanical ventilation by oro-tracheal tube or tracheostomy (Clinical indications for noninvasive positive pressure ventilation in chronic respiratory failure due to restrictive lung disease, 1999; Lightowler JV et al., 2003).
OTHER PHARMACOLOGIC CONSIDERATIONS
A number of other treatments have been explored over the years. Among these therapies, there is either insufficient evidence to warrant recommending their use, or they have been proven to not be beneficial in the management of COPD.
Suppressive Antimicrobial Agents: Because COPD patients often are colonized with bacteria and experience recurrent exacerbations of their condition, a common practice employed in the past has been the use of low-dose antimicrobial therapy as preventative or prophylaxis against these acute exacerbations.
Expectorants and Mucolytics: Adequate water intake generally is acceptable to maintain hydration and assist in the removal of airway sections. Beyond this, the regular use of mucolytics or expectorants for COPD patients has no proven benefit. This includes the use of saturated solutions of potassium iodide, ammonium chloride, acetylcysteine, and guaifenesin.
Narcotics: Systemic (oral and parenteral) opioids, especially morphine, can relieve dyspnea in patients with endstage COPD. Nebulized therapy is sometimes used in clinical practice although data about clinical benefit are lacking.
NON PHARMACOLOGICAL TREATMENT
Patients with COPD should receive education about their disease, treatment plans, and strategies to slow progression and prevent complications.
Smoking Cessation: A primary component of COPD management is avoidance of or reduced exposure to risk factors. Exposure to environmental tobacco smoke is a major risk factor, and smoking cessation is the most effective strategy to reduce the risk of developing COPD. As confirmed by the Lung Health Study, smoking cessation is the only intervention proven at this time to affect long-term decline in FEV1 and slow the progression of COPD. In this 5-year prospective trial, smokers with early COPD were randomly assigned to one of three groups: smoking-cessation intervention plus inhaled ipratropium three times a day, smoking-cessation intervention alone, or no intervention. During an 11-year follow up, the rate of decline in FEV among subjects who continued to smoke was more than twice the rate in sustained quitters. Smokers who underwent smoking cessation intervention had fewer respiratory symptoms and a smaller annual decline in FEV1 compared with smokers who had no intervention. Every clinician has a responsibility to assist smokers in smoking cessation efforts. In 2004, a report from the Surgeon General on the health consequences of smoking broadened the scope of the detrimental effects of cigarette smoking, indicating that “Smoking harms nearly every organ of the body, causing many diseases and reducing the health of smokers in general.” Advice should be given to smokers even if they have no symptoms of smoking-related disease or if they are receiving care for reasons unrelated to smoking. Brief interventions (3 minutes) of counseling are proven effective. However, it must be recognized that the patient must be ready to stop smoking because there are several stages of decision making. The usual duration of therapy is 8 to 12 weeks, although some individuals may require longer courses of treatment.
Pulmonary Rehabilitation: Exercise training is beneficial in the treatment of COPD to improve exercise tolerance and to reduce symptoms of dyspnea and fatigue. Pulmonary rehabilitation programs are an integral component in the management of COPD and should include exercise training along with smoking cessation, breathing exercises, optimal medical treatment and health education. High-intensity training (70% maximal workload) is possible even in advanced COPD patients, and the level of intensity improves peripheral muscle and ventilatory function. Studies have demonstrated that pulmonary rehabilitation with exercise three to seven times per week can produce long-term improvement in activities of daily living, quality of life, exercise tolerance, and dyspnea in patients with moderate to severe COPD.
Immunizations: Vaccines can be considered as pharmacologic agents. However, their role is described here in reducing risk factors for COPD exacerbations. Because influenza is a common complication in COPD that can lead to exacerbations and respiratory failure, an annual vaccination with the inactivated intramuscular influenza vaccine is recommended. Immunization against influenza can reduce serious illness and death by 50% in COPD patients.
The polyvalent pneumococcal vaccine, administered one time, is widely recommended for people from 2 to 64 years of age who have chronic lung disease and for all people older than age 65 years. Thus COPD patients at any age are candidates for vaccination. Although evidence for the benefit of the pneumococcal vaccine in COPD is not strong. Currently, administering the vaccine remains the standard of practice and is recommended by the Centers for Disease Control and Prevention and the American Lung Association.
Long-Term Oxygen Therapy: The use of supplemental oxygen therapy increases survival in COPD patients with chronic hypoxemia. Although long-term oxygen has been used for many years in patients with advanced COPD.
Oxygen-conservation devices are available that allow oxygen to flow only during inspiration, making the supply last longer. These may be particularly useful to prolong the oxygen supply for mobile patients using portable cylinders. However, the devices are bulky and subject to failure.
Long-term oxygen therapy provides even more benefit in terms of survival after 5 years of use, and it improves the quality of life of these patients. Before patients are considered for long-term oxygen therapy, they should be stabilized in the outpatient setting, and pharmacotherapy should be optimized. Once this is accomplished, long term oxygen therapy should be instituted if either of two conditions exists:
i. a resting PaO2 of less than 55 mm Hg,
ii. evidence of right-sided heart failure, polycythemia, or impaired neuropsychiatric function with a PaO2 of less than 60 mm Hg.
The most practical means of administering long-term oxygen is with the nasal cannula, at 1 to 2 L/min which provides 24% to 28% oxygen. The goal is to raise the PaO2 above 60 mm Hg. There are three different ways to deliver oxygen, including
• in liquid reservoirs,
• compressed into a cylinder, and
• via an oxygen concentrator.
Adjunctive Therapies: In addition to supplemental oxygen, adjunctive therapies to consider as part of a pulmonary rehabilitation program are psychoeducational care and nutritional support. Psychoeducational care (such as relaxation) has been associated with improvement in the functioning and well-being of adults with COPD.
SURGICAL INTERVENTION
Various surgical options have been employed in the management of COPD. These include bullectomy, lung volume reduction surgery (LVRS) and lung transplantation.
Bullectomy has been performed for many years and may be useful when large bullae (>1 cm) are noted on computed tomographic scan. The presence of bullae may contribute to complaints of dyspnea and their removal can improve lung function and reduce symptoms, although there is no evidence of a mortality benefit. Because of the prevalence of COPD, it is the most frequent indication for lung transplantation.
Recent trials have evaluated the effect of bilateral LVRS for management of severe COPD. Short-term trials comparing the effects of pulmonary rehabilitation plus LVRS with pulmonary rehabilitation alone reported that the combination of treatments resulted in greater improvements in lung function, gas exchange, and quality of life at 3 months. The National Emphysema Treatment Trial, a prospective, randomized trial evaluating the long-term effects of LVRS plus pulmonary rehabilitation compared with pulmonary rehabilitation alone, followed 1,218 patients for 3 years. The primary end points for the study were mortality and maximal exercise capacity 2 years after randomization. Secondary end points included pulmonary function, distance walked in 6 minutes, and quality-of life measurements. At an interim analysis, patients with an FEV1 of less than 20% of predicted or a carbon monoxide diffusing capacity of less than 20% of predicted were noted to be at high risk of death after surgery and subsequently were excluded from the study. Results of the study showed no mortality benefit with LVRS compared with pulmonary rehabilitation alone. Patients undergoing surgery had improved exercise capacity, lung function, and quality of life at 2 years, but these patients also had a higher risk of short-term morbidity and mortality associated with the surgery. A subgroup analysis of the study noted that patients with predominately upper-lobe emphysema and low exercise capacity undergoing surgery had lower mortality rates at 2 years compared with patients treated with medical therapy alone. Because of the costs and risks associated with LVRS (Robert L. Talbert et al., 2008).
CURRENT RESEARCH STATUS ON COPD
Maryam Mahmoodan et al (2017) carried out a study on Drug Utilization Evaluation in Chronic Obstructive Pulmonary Disease Patients. Chronic obstructive pulmonary disease (COPD) requires extensive treatment with multiple drug therapy. Due to its increasing prevalence and complications, it is rapidly becoming one of the world’s most serious health issues. It is necessary to treat the disease at the earliest with proper monitoring and evaluation of the therapy to prevent further progression and associated complications. Drug utilization evaluation is mainly used to understand the drug use pattern; use of irrational drugs; intervention to improve drug use and continuous quality improvement. Hence the present study was undertaken to understand the prescription pattern of patients diagnosed with COPD in a teaching hospital of South India. A total of 100 patients diagnosed with COPD was included in the study. The severity level of COPD was analyzed using Modified Medical Research Council Questionnaire (mMRC) scale and the medications prescribed for treating were analyzed using GOLD treatment guidelines. Most of the patients were admitted with Grade-2 of modified Medical Research Council (mMRC) dyspnoea scale. Corticosteroids (22%) were the most prescribed class of drugs in which inhaled corticosteroids (budesonide and fluticasone) were preferred. The most prescribed combination therapy was salbutamol with ipratropium bromide. The majority of patients was treated with multiple drug therapy, in which combination of corticosteroids, β2-agonists, methylxanthines and anticholinergics along with antibiotics were used. Independent of socioeconomic status, poor health literacy is associated with greater COPD severity (Maryam Mohmoodan et al., 2017).
Dr. R. Kothai et al (2017) carried out a study on Analysis of Prescribing Pattern of COPD Patients in a Tertiary Care Hospital Salem. Chronic obstructive pulmonary disease (COPD), a common preventable and treatable disease, is one of the leading causes of morbidity and mortality in the industrialized and the developing countries. An attempt was made to assess the current prescribing pattern with the WHO prescribing indicators to see whether the prescription pattern followed WHO guidelines in a tertiary care hospital in Salem. Retrospective analysis of 150 patients admitted in the General Medicine department over a period of six months from November 2015 to April 2016 in Vinayaka Mission’s Kirupananda Variyar Medical College And Hospitals, Salem, Tamil Nadu were selected for analyzing the drug use pattern using WHO prescribing indicators and drug-drug interactions in the prescriptions. The prescriptions were analyzed using descriptive statistics and results were expressed in percentage. A total of 1015 drugs were prescribed and the average number of drugs per prescription was found to be 7.69±2.24. Most of the drugs were prescribed by brand name (63.34%). The percentage of drugs prescribed as per EDL-WHO 2015 was 22.36%. The most commonly prescribed COPD drug was Deriphylline [123(20.5%)]. The prescribing patterns were not in accordance with WHO guidelines so it is necessary to make doctors aware about the use of drugs, importance of prescribing drugs with generic names, safety of prescribing drugs from EDL and patient’s point of view (Dr. R. Kothai et al., 2017).
Aswathy Unni et al (2015) carried out a study On Drug Utilization Pattern In Chronic Obstructive Pulmonary Disease In patients At A Tertiary Care Hospital. Drug utilization studies provide useful insights into the current prescribing practices. In this retrospective study, all patient data relevant to the study were obtained by examination of patient’s medical records and hospital information system. A total of 237 patients with acute exacerbation were evaluated. The population predominantly consisted of males and most of the patients were in the age group of 61 to 70 years. Hypertension was the most common co-morbidity. Candida albicans and Pseudomonas aeruginosa were the most common microorganisms isolated from sputum samples. Majority of the patients were on multidrug therapy. During hospital stay, the most commonly prescribed drugs were ipratropium and levosalbutamol. The prescribing trend observed at our hospital appears to be in concordance with the current guidelines for the management of COPD patients (Aswathy Unni et al., 2015).
SK Veettil et al (2014) carried out a prospective study to recognize The Drug Utilization Pattern for the Treatment of Acute Exacerbation of COPD for 7-Days Under Non-Experimental Settings. The information related to the study was collected from the case records and discussed with the inpatients during ward rounds, with the support of physician. The results indicates that only those above 40 years of age were admitted into the hospital and the majority of patients were of the male sex. The percentage of drugs used were as follows: Salbutamol accounts for about 74% use, Parenteral steroid is 78%, all of them received hydrocorticosteroid, steroid inhalers-25%, Anticholinergics-77.5% and Antibiotics were used in 86.7% patients. The study suggested that despite the use of drugs according to the availability and physician's preference, it was found in the analysis that majority were in accordance with Global Initiative for Chronic Obstructive Lung Disease criteria recommendations (Sajesh Kalkandi Vettil et al., 2014).
Anderw Woods J et al (2014) carried out a study to review The Use of Corticosteroid in the Treatment of Acute Exacerbations of COPD. All reference citations from identified publications were reviewed for possible inclusion. All identified randomized, placebo-controlled trials, meta-analyses, and systematic reviews evaluating the efficacy of systemic corticosteroids in the treatment of AECOPD were reviewed and summarized. The result indicates that are in comparison. The study suggests that the Systemic corticosteroids are efficacious in the treatment of AECOPD and considered a standard of care for patients experiencing an AECOPD. Therefore, systemic corticosteroids should be administered to all patients experiencing AECOPD severe enough to seek emergent medical care. The lowest effective dose and shortest duration of therapy should be considered (Woods JA et al., 2014).
WW Wu et al (2014) carried out a study to know The Effect of Oxygen-Driven Nebulization (ODN) at Different Oxygen Flows on Heart Rate, Respiratory Rate, SpO2, SaO2, PaO2, PaCO2 and pH of Acute Exacerbation of Chronic Obstructive Pulmonary Disease (AECOPD) Patients. In this study a total of 9 AECOPD patients were randomly divided into 3 groups, numbered A, B and C and treated with ODN. Oxygen flow of groups A, B and C was 4-5, 6-7 and 8-9 L/min, respectively. Heart rate, respiratory rate, SpO2, SaO2, PaO2, PaCO2 and pH were recorded before ODN and 30 minutes after ODN. Statistical differences of data before or after ODN were analyzed by analysis of variance and F-test, whereas data before and after ODN were tested by paired t test. The results indicate that there was no significant difference of heart rate, respiratory rate, SpO2, PaO2, PaCO2, SaO2 and pH among 3 groups before ODN or after ODN. The heart rate was increased in all groups after ODN. But significant increase was only present in groups A and C but not in group B. SaO2 was significantly increased in group C after ODN but no statistical difference was observed between before and after ODN in groups A and B. There was no significant change in respiratory rate, SpO2, PaO2, PaCO2, SaO2 and pH between before and after ODN in all groups (Wu WW et al., 2014).
Dhand R et al (2012) carried out a study To Evaluate the Role of Nebulized Therapy in the Management of COPD. Current guidelines recommend inhalation therapy as the preferred route of drug administration for treating chronic obstructive pulmonary disease (COPD). Previous systematic reviews in COPD patients found similar clinical outcomes for drugs delivered by handheld inhalers - pressurized metered-dose inhalers (pMDIs), dry powder inhalers (DPIs) - and nebulizers, provided the devices were used correctly. However, in routine clinical practice critical errors in using handheld inhalers are highly prevalent and frequently result in inadequate symptom relief. In comparison with pMDIs and DPIs, effective drug delivery with conventional pneumatic nebulizers requires less intensive patient training. Moreover, by design, newer nebulizers are more portable and more efficient than traditional jet nebulizers. The study show despite several known drawbacks associated with nebulized therapy, we recommend that maintenance therapy with nebulizers should be employed in elderly patients, those with severe disease and frequent exacerbations, and those with physical and/or cognitive limitations. Likewise, financial concerns and individual preferences that lead to better compliance may favor nebulized therapy over other inhalers. For some patients, using both nebulizers and pMDI/DPI may provide the best combination of efficacy and convenience (Dhand R et al., 2012).
K Richard et al (2011) carried out a prospective parallel group, Placebo-Controlled Design Study. Participants were randomly selected in a 1:1 ratio to receive azithromycin, at a dose of 250 mg orally, or an identical-appearing placebo, once daily. The participants should be atleast of 40 years of age. Among subjects at increased risk for acute exacerbations of COPD who received azithromycin-250 mg once daily, for 1 year, the frequency of acute exacerbations was decreased. The results indicates that among 1577 subjects who were screened, 1142 (72%) were randomly selected to receive azithromycin at a dose of 250mg daily (570 participants) or placebo (572 participants) for 1 year and the rate of 1-year follow-up was 89% in the azithromycin group and 90% in the placebo group. The frequency of exacerbations was 1.48 exacerbations per patient-year in the azithromycin group, as compared with 1.83 per patient-year in the placebo group (P=0.01), and the hazard ratio for having an acute exacerbation of COPD per patient-year in the azithromycin group was 0.73 (95% CI, 0.63 to 0.84; P<0.001). The study shows that among selected subjects with COPD azithromycin taken daily for 1 year, when added to usual treatment, decreased the frequency of exacerbations and improved quality of life but caused hearing decrements in a small percentage of subjects (Albert RK et al., 2011).
Paolo Montuschi (2006) carried out a study on Pharmacological Treatment Of Chronic Obstructive Pulmonary Disease. None of the drugs currently available for chronic obstructive pulmonary disease (COPD) are able to reduce the progressive decline in lung function which is the hallmark of this disease. Smoking cessation is the only intervention that has proved effective. The current pharmacological treatment of COPD is symptomatic and is mainly based on bronchodilators, such as selective β2-adrenergic agonists (short and long-acting), anticholinergics, theophylline, or a combination of these drugs. Glucocorticoids are not generally recommended for patients with stable mild to moderate COPD due to their lack of efficacy, side effects, and high costs. New pharmacological strategies for COPD need to be developed because the current treatment is inadequate (Paolo Montuschi et al., 2006).
P Moayyedi et al (1995) carried out a study to known any benefit in adding ipratropium bromide to salbutamol in acute exacerbations of COPD and Combination therapy with the two drugs was compared with salbutamol alone in the treatment of acute exacerbations of COPD during a hospital admission Seventy patients admitted to hospital with an acute exacerbation of COPD were randomly allocated to receive either nebulised salbutamol 5 mg and ipratropium bromide 500 mg, or nebulised salbutamol 5 mg alone (all four times a day) on admission. All other treatment was prescribed at the discretion of the attending physician. Length of stay in hospital and spirometeric values on days 1, 3, 7, 14, and discharge were assessed. The results indicates that there was no difference between the two groups in the mean (SD) length of stay (salbutamol 10-5 (4.7) days, salbutamol + ipratropium bromide 11*8 (4.4) days; 95% CI -1-02 to 3.62). There was no difference in spirometric values on days 1, 3, 7, 14 or discharge between the two groups. The subjective improvement was similar with both treatments. The study show that the routine addition of nebulised ipratropium bromide to salbutamol appears to be of no benefit in the treatment of acute exacerbations of COPD (P Moayyedi et al., 1995).
AIMS AND OBJECTIVES
The present study aims to document the evaluation of clinical use of nebulization therapy and antibiotics in in-patients with chronic obstructive pulmonary disease.
1. To assess the clinical usage of nebulization therapy.
2. To assess the clinical usage of antibiotic therapy.
3. To assess the social history of COPD patients.
4. To assess Changes in Oxygen saturation before and after nebulization.
5. To assess organism causing COPD by Culture sensitivity.
METHODOLOGY
PLAN OF WORK
A study was carried out to evaluate the clinical use of nebulization therapy and antibiotics in in-patients with COPD. All the nebulizers and antibiotics containing prescription were monitored to know the frequency extent of nebulizer and antibiotic use and also the conditions in which it was prescribed. Rationality of prescriptions was evaluated by using the WHO core drug prescribing indicators, that is, (a) average number of drugs per encounter, (b) percentage of encounters with an antibiotic, (c) percentage of encounters with an injection, (d) percentage of encounters with nebulizer. Literatures which support the study were collected and were reviewed for study on clinical use of nebulizer and antibiotic therapy in Chronic Obstructive Pulmonary Disease. A standard data entry format for collecting patient’s details was designed and during the ward rounds the entire patient data with special reference to the nebulizers and antibiotics prescribe were recorded in the format.
STUDY SITE
In-Patient of Pulmonology Department at Gleneagles Global Hospitals, Bairamalguda, LB Nagar, Hyderabad.
The study was conducted in the Pulmonology Department, Gleneagles Global Hospitals. It is a 300 bedded multispecialty hospital which has intensive care units with 50 beds, General wards with 80 beds and twin sharing rooms with 96 beds and medical sharing with 74 beds providing its health care services with around 40 physicians, 8 surgeons and around 30 Duty medical officers and 1 clinical pharmacist located in Bairamalguda, LB Nagar, Hyderabad. It has rendered its services in the entire state. It is a massive building with expertise in various fields in hospital services.
STUDY DESIGN
A hospital based prospective observational study was carried out on 102 in-patients in general medicine wards.
The data was collected which contains patients demographics (age, sex), date of admission, date of discharge, history of present illness, past medical history, diagnosis, name of the drugs, dosage regimen (form, dose, route, frequency and duration). The knowledge assessment questionnaire form contains about 21 questions to assess the knowledge of patient towards the medications used.
SAMPLE SIZE
A total of 102 in-patients from the in-patient of Pulmonolgy department in Gleneagles Global Hospital, who were prescribed nebulizers and antibiotics and those who fulfilled the exclusion and inclusion criteria were selected for the study.
STUDY PERIOD
The study was carried out for a period of six months.
STUDY CRITERIA
Inclusion Criteria:
• Patients aged between 30 years to 80 years.
• Patients of either sex with a prescription of mentioned drugs.
• Patients with and without co-morbidities.
• Smoking status.
• Alcoholic status.
Exclusion Criteria:
• Outpatients.
• Pregnant.
STUDY APPROVAL
The study was approved by Ethical Committee of Sree Dattha Institute of Pharmacy. Permission for collection of data and to accompany physician in the Pulmonology Department was taken from the Head of the Pulmonology Department before starting the study. The authors were permitted to utilize the hospital facilities and make a follow up with prescriptions in the department.
SOURCES OF DATA
Patient Profile Form:
• Patient demographic details (age, sex, alcoholic status, smoking status),
• Chief complaints,
• History of present illness,
• Past medical and medication history,
• Family history,
• Final diagnosis,
• Name of the drugs,
• Dosage regimen (form, dose, route, frequency and duration),
• Date on which pharmacotherapy was initiated,
• Laboratory data.
Knowledge Assessment Questionnaire Contains The Following Questions:
• Do you cough?
• Does your cough produce phlegm?
• What kind of mucus is produced?
• What is the colour of the mucus?
• Does your cough disturb your sleep?
• Do you have shortness of breath?
• Do you have shortness of breath when hurrying or walking up a straight hill?
• Are you a smoker?
• Do you smoke now?
• How many cigarettes a day do you smoke?
• When did you quit smoking?
• Do you smoke even though you are sick in bed for most of the time?
• Do you have wheezing?
• If you have wheeze, is it worse in the morning?
• Do you feel shortness of breath when you wheeze?
• Do you have chest problem?
• How would you describe your chest condition?
• Do you consume alcohol?
• When did you Quit Alcohol?
• How frequently do you consume Alcohol?
• Have you ever felt you should stop consuming alcohol?
DETERMINATION OF PRESCRIBING PATTERNS:
A suitable data collection form was designed and used.
Data was collected with respect to:
• Demographic details: name, age, sex,
• Condition and reasons for hospital admission were recorded,
• Drug data: Brand and generic name of the drugs prescribed, dose, route of administration, frequency and their follow up for few days was recorded.
STATISTICAL SOFTWARE:
• SPSS Software.
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT editor-in-chief@pharmatutor.org
Subscribe to Pharmatutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE
RESULTS
According to the plan of work, Rationality of prescriptions are evaluated by using the WHO core drug prescribing indicators, which resulted as:
a. Average number of drugs per encounter: 11 drugs
b. Percentage of encounters with an antibiotic: 25.2%
c. Percentage of encounters with an injection: 30.3%
d. Percentage of encounters with nebulizer: 17.7%
Population Description: A total of 102 patients with presumed/confirmed COPD. The Mean Age of males was 67 years and females was 61 years and the standard deviation of males was 13 years and female was 15 years. Of the population, 84% were males and 16% were females, 41% were smokers and 59% were non-smokers. The main demographic and clinical characteristics, co-morbidities have been described later.
Table 3: Distribution Of Subjects Based Upon Age In COPD (%)
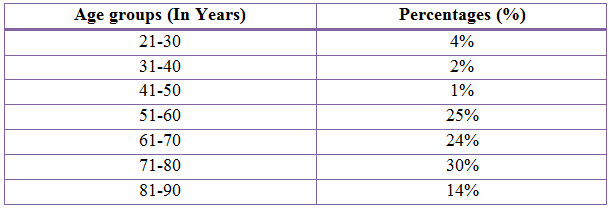
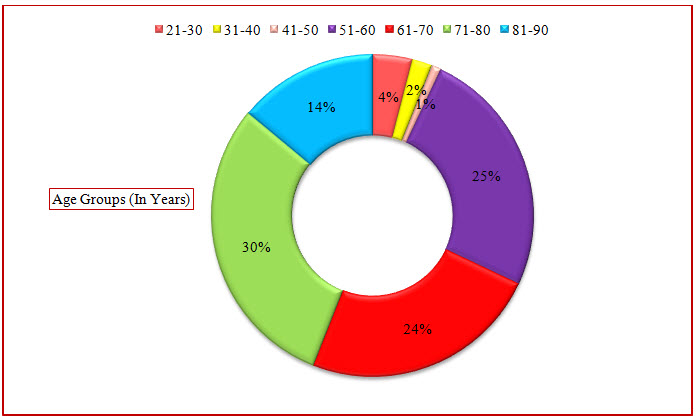
Figure 3: Doughnut Presentation Of Distribution Of Subjects Based Upon Age In COPD(%)
Table 4: Statistical Presentation of the age group data by Column Statistics
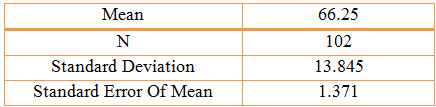
Table 5: Distribution Of Subjects Based Upon Gender In COPD (%)

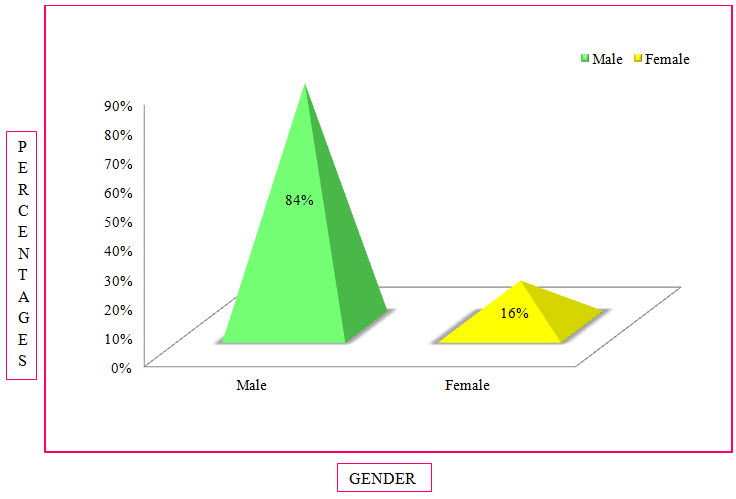
Figure 4: Column Presentation Of Distribution Of Subjects Based On Gender In COPD(%)
Among The Study Population, 86 Individuals Were Found To Be Males And 16 Individuals Were Females.
Table 6: Distribution Of Subjects Based Upon Social History In COPD (%)

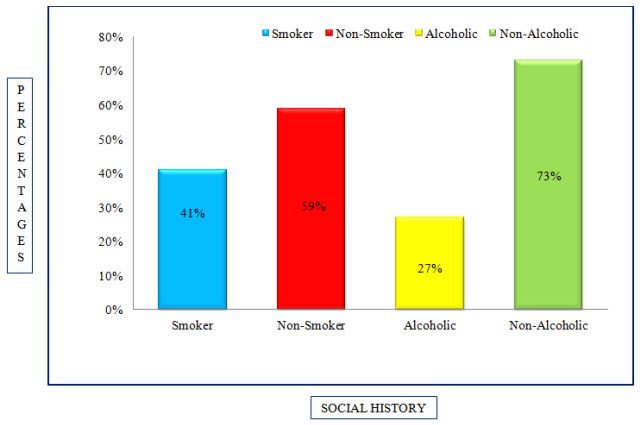
Figure 5: Column Presentation Of Distribution Of Subjects Based Upon Social History In COPD (%)
Among the study population, 28 individuals were alcoholics, 74 were non-alcoholics and 42 of individuals were smokers, 60 were non-smokers.
Table 7: Distribution Of Subjects Based Upon Culture Sensitivity Test In COPD (%)

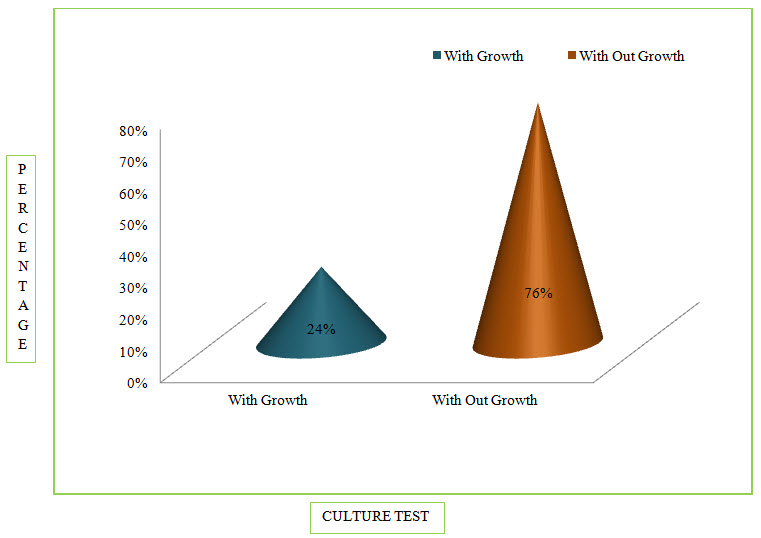
Figure 6: Column Presentation Of Percentage Of Culture Sensitivity Test In COPD (%)
Among the study population, 24 patients were found positive for Culture Test.

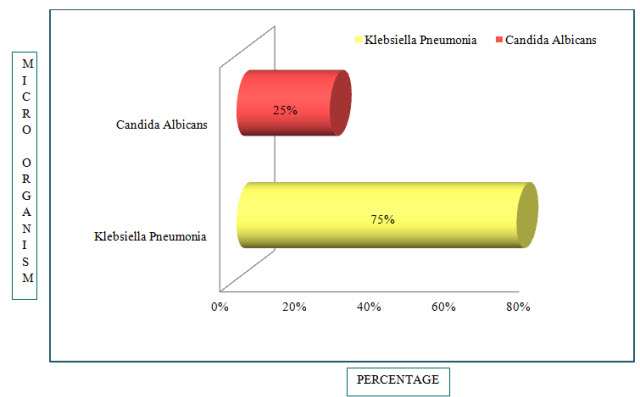
Figure 7: Bar Presentation of Percentages of Microbes in COPD(%)
Out of 24 patients, Klebsiella Pneumonia was found in 18 patients (75%) and Candida Albicans was found in 6 patients (25%).
Table 9: Percentage of Drugs Used in COPD (%)
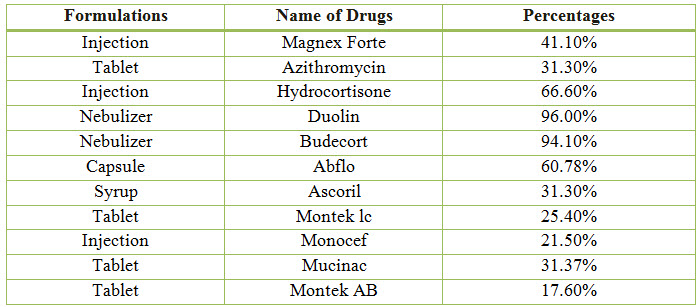
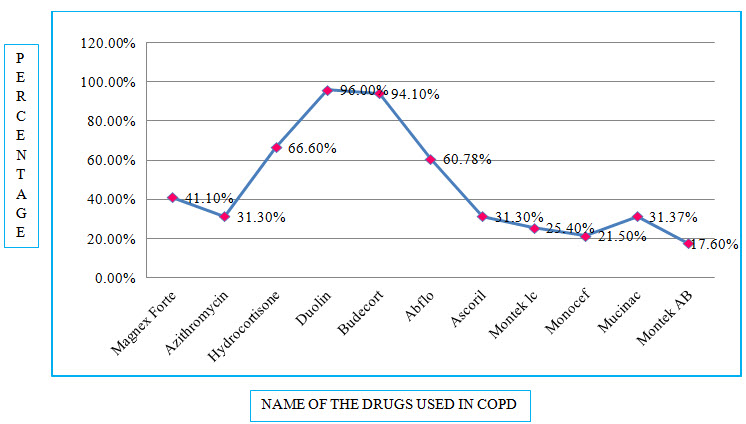
Figure 8: Line presentation of percentage of drugs used in COPD
Among the study population, the most commonly used Bronchodilator is Duolin with a percentage of 96.00% and the most commonly used Antibiotic is Magnex Forte with a percentage of 41.10%.
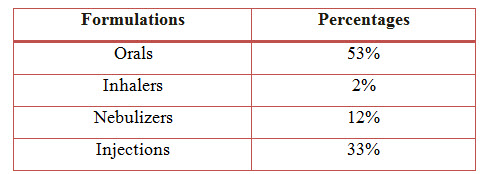
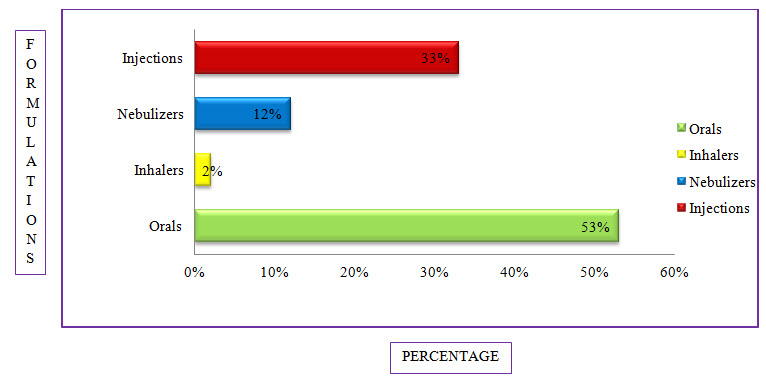
Figure 9: Bar Presentation Of Percentage Of Formulations Used In COPD (%)
Among the study population, the most commonly used formulation is Orals with a percentage of 53% followed by Injections with a percentage of 33%, Nebulizers with a percentage of 12% and the least common is Inhalers with a percentage of 2%.
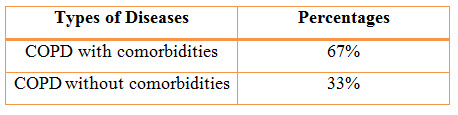
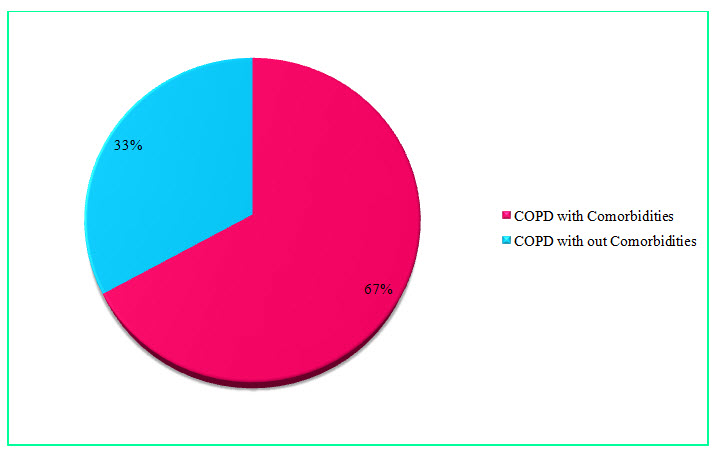
Figure 10: Pie Chart presentation of results of percentages of COPD with or without co-morbidity (%)
Among the study population, 68 individuals suffering from COPD with comorbidities resulted in a percentage of 67% and 34 individuals suffering from COPD without comorbidities resulted in a percentage of 33%.
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT editor-in-chief@pharmatutor.org
Subscribe to Pharmatutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE
Table 12: Changes in O2 Saturation Before and After Nebulization therapy in COPD (%)
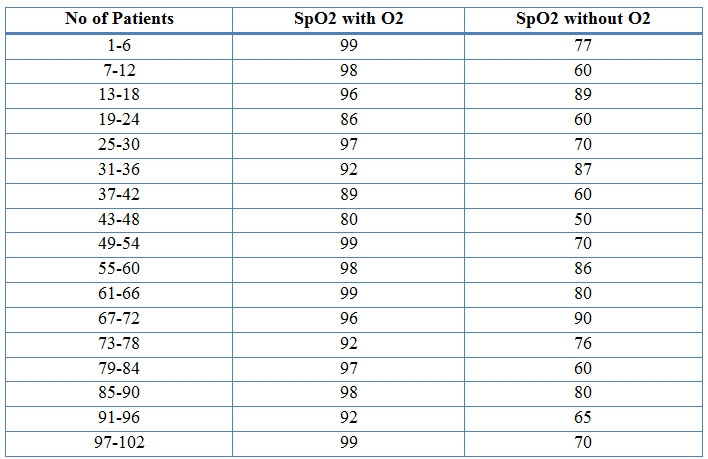
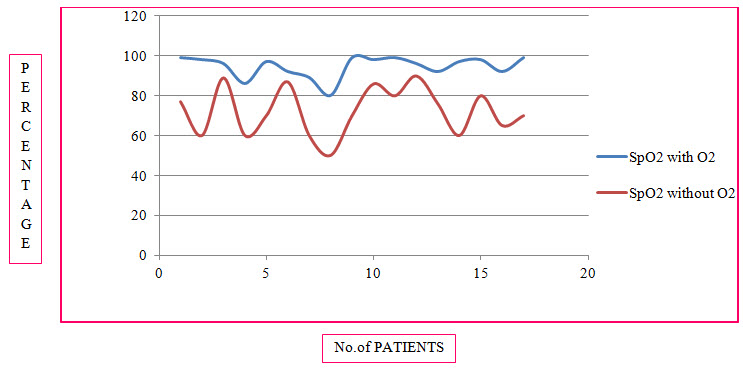
Figure 11: Line Presentation Of Changes In O2 Saturation Before And After Nebulization In COPD (%)
Table 13: One-Sample Statistics of SpO2 with O2

Table 14: One-Sample Statistics of SpO2 with out O2

Among the study population, 94 individuals had effect on SpO2 levels whereas 8 individuals had no effect on SpO2 levels. 92% of individuals responde in a positive way after the nebulization therapy with oxygen therapy provided to them, as their SpO2 levels with oxygen therapy changed to normal range.
DISCUSSION
The rationality of prescription was evaluated by using the WHO core drug Prescribing indicators which was developed and published by WHO and INRUD (Quick JD et al., 1997). It resulted as the average number of drugs per encounter were found to be 11 drugs.
• Percentage encounters with an Antibiotic was 25.2%.
• Percentage of encounters with an Injection was 30.3%.
• Percentage of encounters with Nebulizers was 17.7%.
Thus, it was found to be irrational use of the drugs as two drugs per encounter is the rational use of drugs as per Prescription studies.
The age group between 21-90 years were admitted during the period of study. The age group between 21-30 years were found to be 4% where as in the study conducted previously only above 40 years of age were admitted during their period of study. The majority of patients were between 71-80 years age group (30%).
The sex wise distribution resulted that majority of the patients was of male sex which was as high as 84%. The females accounted for around 16%. Male to female ratio observed in our study is 5:1. This confirms the finding of the previous study conducted that males accounted for the majority of the disease burden which was as high as 87.5% (Sajesh Kalkandi Veettil et al., 2014).
The Alcoholic status distribution resulted that majority of the patients were Non-Alcoholic 73% and Alcoholics accounted for around 27%.
As per the survey done in the previous study, the common risk factors into focus were smoking, alcoholism and tobacco consumption. It was seen that 37% of the patients had smoking status (Maryam Mahmoodan et al., 2017). Where as In present study, the smokers accounted for around 41% and non-smokers accounted for 59% making smoking as a potential risk hazard for COPD patients.
In present study, the culture sensitivity test was performed in COPD patients. Among the study population, 24% were positive towards culuture sensitivity test, Out of which Candida Albicans was found in 6 patients (25%) and Klbesiella Pneumonia was found in 18 patients (75%). Whereas Candida albicans (16%) and Pseudomonas aeruginosa (4.6%) were the most common microorganisms isolated from sputum samples in previous study (Aswathy Unni et al., 2015).
Since, COPD requires extensive treatment, different classes of drugs used in the treatment were also assessed in the present study. Among them, Bronchodilators were mostly prescribed (96%). When compared to the study conducted by Maryam Mahmoodan et.al, our results were not in accordance, as the use of Corticosteroids was higher in that study (21.9%). Deriphylline was the most prescribed methylxanthine (13.9%).
Among the combination therapies, salbutamol with ipratropium bromide was the most preferred combination (96%). Where in previous studies salbutamol with ipratropium bromide combination was used in 54% of the cases. Salbutamol with ipratropium bromide was mostly preferred in older patients as it prevents cardiovascular complications and also one can avoid high doses of β2-agonist drugs (Maryam Mahmoodan et al., 2017).
Antibiotics were used in 92.1% of patients which may be an indicator of the prevalence of infections among the patients admitted with acute exacerbation of COPD in different demographic areas (Sajesh Kalkandi Veettil et al., 2014). The most commonly used antibiotic is Magnex Forte accounting for about 41.1%.
Corticosteroids remain a standard of care for Acute exacerbation of COPD, secondary to their documented efficacy in improving air flow and gas exchange (increasing FEV1 and PaO2) as well as improving dyspnea, hasten recovery, and reducing length of hospitalization and rate of relapse at 30 days. In the previous study, the Systemic Corticosteroids are efficacious in the treatment of COPD and is considered as the standard care for COPD patients (Woods JA et al., 2014). The Corticosteroids accounted for about 66.6% which included the IV injection of Hydrocortisone and relatively the same percentage of Prednisolone. Therefore in our study, the use of Systemic Corticosteroid is used as an additional therapy to treat COPD along with different medications.
The majority of the drugs are taken as Orals than the Inhalers, Nebulizers and Injections which accounted for 53%, 2%, 12% and 33% respectively. In the previous study the most preffered Route of Administration is Injections, and they state that, the possible reason for the use of high amount of injectables could be beliefs and attitudes of patients and physicians about the high efficacy of injections as compared to oral medications (Dr. R. Kothai et al., 2017). The use of Nebulization therapy was preferred over Inhalational therapy as the formulation of Nebulizers account around 12% and Inhalers around 2%. Despite several known drawbacks associated with Nebulized therapy it is recommended that maintenance therapy with Nebulizers should employed in elderly patients and those with physical (or) cognitive limitations (Dhand R et al., 2012) as it is easy to use and results in a positive manner than Inhalational therapy.
Majority of the patients in the study had co-morbid conditions accounting for about 67% and patients without co-morbid conditions accounted for 33% where in the previous study 55% of the patients presented with COPD alone and 45% of patients presented COPD along with certain co-morbidities (Maryam Mahmoodan et al., 2017). The most commonly observed co-morbid condition was Hypertension. The other co-morbid conditions found were Diabetes Mellitus, Congestive Cardiac Failure, Alcoholic Hepatitis, Acute Renal Failure etc.
In the previous study, there was no significant change in the SpO2 levels before and after the ODN was given (Wu WW et al., 2014). In the present study, there was a significant difference between the value of SpO2 with O2 and SpO2 without O2. The levels of SpO2 with the oxygenation were changed in the normal range after the Nebulization therapy was given.
CONCLUSION
In our present study we performed a Randomized, Prospective and Observational study on patient cohort in Gleneagles Global Hospital to evaluate the following:
• The Observational study was conducted in 102 patients to assess the efficacy of Nebulization and Antibiotic therapy in COPD patients. During the study, the SpO2 levels were monitored and recorded as the parameter to assess the efficacy of the Nebulization therapy. After the study, it was founded that 92% of individuals showed a positive response to the Nebulization therapy as their SpO2 levels recovered in the normal ranges.
• During study, the culture sensitivity test was performed in COPD patients. Among the study population, 24% were positive towards culture sensitivity test, Out of which Candida Albicans was found in 6 patients (25%) and Klbesiella Pneumonia was found in 18 patients (75%).
• Poverty and illiteracy lead to increase number of COPD patients as they use OTC and Non-Pharmacological methods to avoid the economic burden of hospitals. During the study, we counseled the patients and provided some knowledge about the disease so as to avoid irrational use of drugs and increase risk of COPD.
• Further study can be done using Spirometry as a diagnostic tool. The study related to adverse effects, associated with prescribed drugs, for treating COPD can be done for optimization of the therapy.
• Duolin belonging to the class of Bronchodilator was found to be the most commonly used Nebulizer in COPD patients. Magnex Forte which is a combinational antibiotic was most commonly used Antibiotic in In-patients with COPD.
• The Alcoholic status distribution resulted that majority of the patients were Non-Alcoholic 73% and Alcoholics accounted for around 27% smokers accounted for around 41% and non-smokers accounted for 59% making smoking as a potential risk hazard for COPD patients.
Thus from our study we have concluded that the Nebulization therapy had more clinical improvements. The addition of corticosteroids had decreased the patient stay in hospital. Antibiotics added to the treatment decreased the frequency of exacerbations. The patient should be educated about the rational use of the drug to decrease risk of COPD.
REFERENCES
1. Albert RK., Connett J., Bailey WC., Casaburi R., Cooper JA Jr., Criner GJ., Curtis JL., Dransfield MT., Han MK., Lazarus SC., Make B., Marchetti N., Martinez FJ., Madinger NE., McEvoy C., Niewoehner DE., Porsasz J., Price CS., Reilly J., Scanlon PD., Sciurba FC,. Scharf SM., Washko GR., Woodruff PG. and Anthonisen NR (2011); Azithromycin for prevention of exacerbations of COPD; N Engl J Med; 365(8); 689-98.
2. Anthonisen NR., Connett JE., Kiley JP., Altose MD., Bailey WC., Buist AS., Conway WA Jr., Enright PL., Kanner RE. and O'Hara P (1994); Effects of smoking intervention and the use of an inhaled anticholinergic bronchodilator on the rate of decline of FEV1. The Lung Health Study; JAMA; 272(19); 1497-505.
3. Anthonisen NR., Manfreda J., Warren CP., Hershfield ES., Harding GK. and Nelson NA (1987); Antibiotic therapy in exacerbations of chronic obstructive pulmonary disease; Ann Intern Med;106(2); 196-204.
4. Aswathy Unni., Akshaya K Jayaprakash., Yadukrishnan Mc. and Uma Devi P (2015); Drug Utilization Pattern In Chronic Obstructive Pulmonary Disease Inpatients At A Tertiary Care Hospital; International Journal of Pharmacy and Pharmaceutical Sciences; 7(11); 389-391.
5. Barbara G. Wells., Joseph T. DiPiro., Terry L. Schwinghammer. and Cecily V. DiPiro (2009), “Chronic Obstructive Pulmonary Disease”, in Dennis M. Williams and Sharya V. Bourdet, Handbook of Pharmacotherapy-A Pathophysiologic Approach, McGraw-Hill, New York, pp. 924-928.
6. Barbera JA., Reyes A., Roca J., Montserrat JM., Wagner PD. and Rodriguez-Roisin R (1992); Effect of intravenously administered aminophylline on ventilation/perfusion inequality during recovery from exacerbations of chronic obstructive pulmonary disease; Am Rev Respir Dis; 145(6); 1328-33.
7. Barnes PJ (2010); Chronic obstructive pulmonary disease: effects beyond the lungs; PLOS Med; 7(3); e1000220.
8. Barnes PJ (2014); Preface. Chronic obstructive pulmonary disease; Clin Chest Med; 35; xiii.
9. Behrendt CE (2005); Mild and moderate-to-severe COPD in nonsmokers. Distinct demographic profiles; Chest; 128; 1239-44.
10. Bianchi R., Gigliotti F., Romagnoli I., Lanini B., Castellani C., Grazzini M. and Scano G (2004); Chest wall kinematics and breathlessness during pursed-lip breathing in patients with COPD; Chest; 125(2); 459-65.
11. Bousquet J., Kiley J., Bateman ED., Viegi G., Cruz AA., Khaltaev N., Ait Khaled N., Baena-Cagnani CE., Barreto ML., Billo N., Canonica GW., Carlsen KH., Chavannes N., Chuchalin A., Drazen J., Fabbri LM., Gerbase MW., Humbert M., Joos G., Masjedi MR., Makino S., Rabe K., To T. and Zhi L (2010); Prioritised research agenda for prevention and control of chronic respiratory diseases; Eur Respir J; 36(5); 995–1001.
12. Boutin-Forzano S., Moreau D., Kalaboka S., Gay E., Bonnefoy X., Carrozzi L., Viegi G., Charpin D. and Annesi-Maesano I (2007); Reported prevalence and co-morbidity of asthma, chronic bronchitis and emphysema: a pan-European estimation; Int J Tuberc Lung Dis; 11(6); 695–702.
13. Burge S. and Wedzicha JA (2003); COPD exacerbations: definitions and classifications; Eur Respir J Suppl; 21(41s); 46s-53s.
14. Burrows B., Knudson RJ., Cline MG. and Lebowitz MD (1977); Quantitative relationships between cigarette smoking and ventilatory function; Am Rev Respir Dis; 115(2); 195-205.
15. Cazzola M. and Matera MG (2014); Bronchodilators: current and future; Clin Chest Med; 35(1); 191–201.
16. Cazzola M. and Page C (2014); Long-acting bronchodilators in COPD: where are we now and where are we going?; Breathe; 10; 110 20.
17. Celli BR. and MacNee W (2004); Standards for the diagnosis and treatment of patients with COPD: a summary of the ATS/ERS position paper; Eur Respir J; 23(6); 932-46.
18. Celli BR., Halbert RJ., Isonaka S. and Schau B (2003); Population impact of different definitions of airway obstruction; Eur Respir J; 22(2); 268-73.
19. Celli BR., Halbert RJ., Nordyke RJ. and Schan B (2005); Airway obstruction in never smokers: results from the Third National Health and Nutrition Examination Survey; Am J Med; 118(2); 1364-72.
20. Chapman KR (2004); Chronic obstructive pulmonary disease: are women more susceptible than men?; Clin Chest Med; 25(2); 331-41.
21. Clinical indications for noninvasive positive pressure ventilation in chronic respiratory failure due to restrictive lung disease, COPD, and nocturnal hypoventilation; Chest 1999; 116(2); 521-34.
22. Davies L., Angus RM. and Calverley PM (1999); Oral corticosteroids in patients admitted to hospital with exacerbations of chronic obstructive pulmonary disease: a prospective randomised controlled trial; Lancet; 354(9177); 456-60.
23. Definition and classification of chronic bronchitis for clinical and epidemiological purposes. A report to the Medical Research Council by their Committee on the Aetiology of Chronic Bronchitis; (1965); Lancet; 1(7389); 775-779.
24. deKoning HW., Smith KR. and Last JM (1985); Biomass fuel combustion and health; Bull World Health Organ; 63(1); 11-26.
25. Dhand R., Dolovich M., Chipps B., Myers TR., Restrepo R. and Farrar JR (2012); The role of nebulized therapy in the management of COPD: evidence and recommendations COPD; 9(1); 58-72.
26. Diaz-Guzman E. and Mannino DM (2014); Epidemiology and prevalence of chronic obstructive pulmonary disease; Clin Chest Med; 35(1); 7–16.
27. Dr. R. Kothai., Dr. Rangabashayan., Dr. B. Arul., Punniya Mariam Sunny., Reshma R. Nair. and Rinku Eliza Mathew (2017); Analysis Of Prescribing Pattern Of COPD Patients In A Tertiary Care Hospital Salem; World Journal Of Pharmacy And Pharmaceutical Sciences; Issue 6(10);1111-1117.
28. Elliott MW., Adams L., Cockcroft A., MacRae KD., Murphy K. and Guz A (1991); The language of breathlessness. Use of verbal descriptors by patients with cardiopulmonary disease; Am Rev Respir Dis; 144(4); 826-32.
29. Fein A. and Fein AM (2000); Management of acute exacerbations in chronic obstructive pulmonary disease; Curr Opin Pulm Med; 6(2); 122–126.
30. Fishman A., Martinez F., Naunheim K., Piantadosi S., Wise R., Ries A., Weinmann G. and Wood DE (2003); A randomized trial comparing lung-volume reduction surgery with medical therapy for severe emphysema; N Engl J Med; 348(21); 2059-2073.
31. Georgopoulas D. and Anthonisen NR (1991); Symptoms and signs of COPD. In: Cherniack NS, ed. Chronic obstructive pulmonary disease. Toronto; WB Saunders Co; 357-63.
32. Halbert RJ., Isonaka S., George D. and Iqbal A (2003); Interpreting COPD prevalence estimates: what is the true burden of disease?; Chest; 123(5); 1684-1692.
33. Halbert RJ., Natoli JL., Gano A., Badamgarav E., Buist AS. and Mannino DM (2006); Global burden of COPD: systematic review and meta-analysis; Eur Respir J; 28(3); 523-32.
34. Hill AT., Bayley D. and Stockley RA (1999); The interrelationship of sputum inflammatory markers in patients with chronic bronchitis; Am J Respir Crit Care Med; 160(3); 893-8.
35. Hnizdo E., Glindmeyer HW., Petsonk EL., Enright P. and Buist AS (2006); Case Definitions for Chronic Obstructive Pulmonary Disease; J COPD; 3(2); 95-100.
36. International Institute of Population Sciences (IIPS) and Macro International (2007); National Family Health Survey NFHS-3, 2005-2006: India: Volume II, Mumbai: IIPS 2007.
37. Jarvis S., Ind PW. and Shiner RJ (2007); Inhaled therapy in elderly COPD patients: time for re-evaluation?; Age Ageing; 36(2); 213-218.
38. Jemal A., Ward E., Hao Y. and Thun M (2005); Trends in the leading causes of death in the United States, 1970-2002; JAMA; 294(10); 1255-1259.
39. Jindal SK., Aggarwal AN., Chaudhry K., Chhabra SK., D'Souza GA., Gupta D., Katiyar SK., Kumar R., Shah B. and Vijayan VK (2006); A multicentric study on epidemiology of chronic obstructive pulmonary disease and its relationship with tobacco smoking and environmental tobacco smoke exposure; Indian J Chest Dis Allied Sci; 48(1); 23-29.
40. Lightowler JV., Wedzicha JA., Elliott M. and Ram FS (2003); Non-invasive positive pressure ventilation to treat respiratory failure resulting from exacerbations of chronic obstructive pulmonary disease: Cochrane systematic review and meta-analysis; BMJ; 326(7382); 185.
41. Liu S., Zhou Y., Wang X., Wang D., Lu J., Zheng J., Zhong N. and Ran P (2007); Biomass fuels are the probable risk factor for chronic obstructive pulmonary disease in rural South China; Thorax; 62(10); 889–897.
42. Lopez AD., Shibuya K., Rao C., Mathers CD., Hansell AL., Held LS., Schmid V. and Buist S (2006); Chronic obstructive pulmonary disease; current burden and future projections; Eur Respir J; 27(2):397-412.
43. Loveridge B., West P., Kryger MH. and Anthonisen NR (1986); Alteration in breathing pattern with progression of chronic obstructive pulmonary disease; Am Rev Respir Dis; 134(5); 930-4.
44. Lozano R., Naghavi M., Foreman K, et al. (2012); Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010; Lancet; 380 (9859): 2095-128.
45. MacNee W (2005); Pulmonary and systemic oxidant/antioxidant imbalance in chronic obstructive pulmonary disease; Proc Am Thorac Soc; 2(1); 50-60.
46. Mannino DM. and AS Buist (2007); Global burden of COPD: risk factors, prevalence, and future trends; Lancet; 370(9589); 765–773.
47. Mary Anne Koda-Kimble., Lloyd Yee Young., Brian K. Alldredge., Robin L. Corelli., B. Joseph Guglielmo., Wayne A. Kradjan. and Bradley R. Williams (2005), “Chronic Obstructive Pulmonary Disease”, in Philip T. Diaz and Daren L. Knoell, Textbook of Applied Therapeutics-The Clinical Use of Drugs, Lippincott Williams & Wilkins, Maryland, USA, pp. 23-1.
48. Mary Anne Koda-Kimble., Lloyd Yee Young., Brian K. Alldredge., Robin L. Corelli., B. Joseph Guglielmo., Wayne A. Kradjan. and Bradley R. Williams (2005), “Chronic Obstructive Pulmonary Disease”, in Philip T. Diaz and Daren L. Knoell, Textbook of Applied Therapeutics-The Clinical Use of Drugs, Lippincott Williams & Wilkins, Maryland, USA, pp. 23-1, 23-2.
49. Mary Anne Koda-Kimble., Lloyd Yee Young., Brian K. Alldredge., Robin L. Corelli., B. Joseph Guglielmo., Wayne A. Kradjan. and Bradley R. Williams (2005), “Chronic Obstructive Pulmonary Disease”, in Philip T. Diaz and Daren L. Knoell, Textbook of Applied Therapeutics-The Clinical Use of Drugs, Lippincott Williams & Wilkins, Maryland, USA, pp. 23-2 to 23-6.
50. Maryam Mahmoodan., N. M. Mahesh. and Bandenawaz Ramdurga (2017); Drug Utilization Evaluation In Chronic Obstructive Pulmonary Disease Patients; Der Pharmacia Lettre; 9(6]; 142-151.
51. McCloskey SC., Patel BD., Hinchliffe SJ., Reid ED., Wareham NJ. and Lomas DA (2001); Siblings of patients with severe chronic obstructive pulmonary disease have a significant risk of airflow obstruction; Am J Respir Crit Care Med; 164 (8 Pt 1); 1419-24.
52. Menezes AM., Perez-Padilla R., Jardim JR., Muino A., Lopez MV., Valdivia G., Montes de Oca M., Talamo C., Hallal PC. and Victora CG (2005); Chronic obstructive pulmonary disease in five Latin American cities (the PLATINO study): a prevalence study; Lancet; 366(9500); 1875-81.
53. Morbidity & Mortality: Chart book on Cardiovascular, Lung, and Blood Diseases. Bethesda, MD: US Department. of Health and Human Services, Public Health Service, National Institutes of Health; (1998).
54. Murray CJ. and Lopez AD (1997); Alternative projections of mortality and disability by cause 1990-2020: Global Burden of Disease Study; Lancet; 349(9064); 1498-504.
55. Murray CJL. and Lopez AD (1996); The global burden of disease: a comprehensive assessment of mortality and disability from diseases, injuries and risk factors in 1990 and projected to 2020. Cambridge, MA; Harvard University Press.
56. National Institute for Clinical Excellence (NICE) (2004); Chronic obstructive pulmonary disease. National clinical guideline on management of chronic obstructive pulmonary disease in adults in primary and secondary care; Thorax; 59 Suppl 1; 1-232.
57. Niewoehner DE., Erbland ML., Deupree RH., Collins D., Gross NJ., Light RW., Anderson P. and Morgan NA (1999); Effect of systemic glucocorticoids on exacerbations of chronic obstructive pulmonary disease. Department of Veterans Affairs Cooperative Study Group; N Engl J Med; 340(25); 1941-7.
58. O'Driscoll BR., Kay EA., Taylor RJ., Weatherby H., Chetty MC. and Bernstein A (1992); A long-term prospective assessment of home nebulizer treatment; Respir Med; 86(4); 317–25.
59. O'Driscoll R (1991); Home nebulized therapy: is it effective?; Respir Med; 85(1); 1–3.
60. P Moayyedi., J Congleton., R L Page., S B Pearson. and M F Muers (1995); Comparison of nebulised salbutamol and ipratropium bromide with salbutamol alone in the treatment of chronic obstructive pulmonary disease; Thorax; 50(8); 834-837.
61. Paolo Montuschi (2006); Pharmacological treatment of chronic obstructive pulmonary disease; Int J Chron Obstruct Pulmon Dis; 1(4); 409–423.
62. Pauwels RA., Buist AS., Calverley PM., Jenkins CR. and Hurd SS (2001); GOLD Scientific Committee. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease. NHLBI/WHO Global Initiative for Chronic Obstructive Lung Disease (GOLD) Workshop summary; Am. J. Respir. Crit. Care Med; 163(5); 1256–76.
63. Pellegrino R., Viegi G., Brusasco V., Crapo RO., Burgos F., Casaburi R., Coates A., van der Grinten CP., Gustafsson P., Hankinson J., Jensen R., Johnson DC., MacIntyre N., McKay R., Miller MR., Navajas D., Pedersen OF. and Wanger J (2005); Interpretative strategies for lung function tests; Eur Respir J; 26(5); 948-68.
64. Postma DS. and Siafakas N (1998); Epidemiology of chronic obstructive pulmonary disease. Management of Chronic Obstructive Pulmonary Disease; Eur Respir; 7; 41–73.
65. Prasad R., Singh A., Garg R. and Giridhar GB (2012); Biomass fuel and respiratory disease in India; Biosci Trends; 6(5); 219-228.
66. Quick JD., Rankin JR., Laing RO., O’Connor RW., Hogerzeil HV., Dukes MNG. and Garnett A (1997); Managing drug supply. 2nd ed; West Hartford, CT, Kumarian Press.
67. Reddy KS., Shah B., Varghese C. and Ramadoss A (2005); Responding to the threat of chronic diseases in India; Lancet; 366(9498); 1744-1749.
68. Robert L. Talbert., Gary C. Yee., Gary R. Matzke., Joseph T. Dipiro., Cecily V. Dipiro., Barbara G. Wells. and L. Michael Posey (2008), “Chronic Obstructive Pulmonary Disease”, in Dennis M. Williams and Sharya V. Bourdet, Textbook of Pharmacotherapy-A Pathophysiologic Approach, McGraw-Hill, New York, pp. 502-513.
69. Roberts CM., Bugler JR., Melchor R., Hetzel MR. and Spiro SG (1993); Value of pulse oximetry in screening for long-term oxygen therapy requirement; Eur Respir J; 6(4); 559-62.
70. Roberts SD., Farber MO., Knox KS., Phillips GS., Bhatt NY., Mastronarde JG. and Wood KL(2006); FEV1/FVC ratio of 70% misclassifies patients with obstruction at the extremes of age; Chest; 130(1); 200-206.
71. Rodriguez-Roisin R (2000); Toward a consensus definition for COPD exacerbations; Chest; 117(5 Suppl 2); 398S-401S.
72. Saint S., Bent S., Vittinghoff E. and Grady D (1995); Antibiotics in chronic obstructive pulmonary disease exacerbations. A meta-analysis; JAMA; 273(12); 957–960.
73. Sajesh Kalkandi Veettil., Kingston Rajiah. and Suresh Kumar (2014); Study of Drug Utilization Pattern for Acute Exacerbation of Chronic Obstructive Pulmonary Disease in Patients Attending a Government Hospital in Kerala, India; J Family Med Prim Care; 3(3); 250–254.
74. Salvi S. and Agarwal A (2012); India needs a national COPD prevention and Control program; J Assoc Physicians India; 60Suppl; 5-7.
75. Salvi S. and Barnes PJ (2009); Chronic obstructive pulmonary disease in non smokers; Lancet; 374(9691); 733-743.
76. Salvi S. and Barnes PJ (2010); Is exposure to biomass smoke the biggest risk factor for COPD globally?; Chest; 138(1); 3-6.
77. Schellevis FG., Van de Lisdonk EH., Van der Velden J., Hoogbergen SH., Van Eijk JT. and Van Weel C (1994); Consultation rates and incidence of intercurrent morbidity among patients with chronic disease in general practice; Br J Gen Pract; 44(383); 259-62.
78. Schols AM., Soeters PB., Dingemans AM., Mostert R., Frantzen PJ. and Wouters EF (1993); Prevalence and characteristics of nutritional depletion in patients with stable COPD eligible for pulmonary rehabilitation; Am Rev Respir Dis; 147(5); 1151-6.
79. Shohaimi S., Welch A., Bingham S., Luben R., Day N., Wareham N. and Khaw KT (2004); Area deprivation predicts lung function independently of education and social class; Eur Respir J; 24; 157–161.
80. Simon PM., Schwartzstein RM., Weiss JW., Fencl V., Teghtsoonian M. and Weinberger SE (1990); Distinguishable types of dyspnea in patients with shortness of breath; Am Rev Respir Dis; 142(5); 1009-14.
81. Soriano JB., Visick GT., Muellerova H., Payvandi N. and Hansell AL (2005); Patterns of comorbidities in newly diagnosed COPD and asthma in primary care; Chest; 128(4); 2099-107.
82. Soriano JR., Maier WC., Egger P., Visick G., Thakrar B., Sykes J. and Pride NB (2000); Recent trends in physician diagnosed COPD in women and men in the UK; Thorax; 55(9); 789-94.
83. Standards for the diagnosis and care of patients with chronic obstructive pulmonary disease. American Thoracic Society; (1995); Am J Respir Crit Care Med; 152(5 pt 2); S77–121.
84. Sterk PJ (2004); Let's not forget: the GOLD criteria for COPD are based on post-bronchodilator FEV1; Eur Respir J; 23(4); 497-498.
85. Stoller JK. and Aboussouan LS (2005); Alpha1-antitrypsin deficiency; Lancet; 365(9478); 2225-36.
86. Tirimanna PR., van Schayck CP., den Otter JJ., van Weel C., van Herwaarden CL., van den Boom G., van Grunsven PM. and van den Bosch WJ (1996); Prevalence of asthma and COPD in general practice in 1992: has it changed since 1977?; Br J Gen Pract; 46(406); 277-81.
87. US Surgeon General. The health consequences of smoking: chronic obstructive pulmonary disease. Washington, D.C. US Department of Health and Human Services; (1984).
88. van den Boom G., van Schayck CP., van Mollen MP., Tirimanna PR., den Otter JJ., van Grunsven PM., Buitendijk MJ., van Herwaarden CL. and van Weel C (1998); Active detection of chronic obstructive pulmonary disease and asthma in the general population. Results and economic consequences of the DIMCA program; Am J Respir Crit Care Med; 158(6); 1730-8.
89. Voelkel NF. and Tuder R (2000); COPD: exacerbation; Chest; 117(5 suppl 2); S376–379.
90. Woods JA., Wheeler JS., Finch CK. and Pinner NA (2014); Corticosteroids in the treatment of acute exacerbations of chronic obstructive pulmonary disease; Int J Chron Obstruct Pulmon Dis; 4(9); 421-430.
91. World Health Organization. (2008), “World Health Statistics”, available at: http://www.who.int/whosis/whostat/2008/en/index.html.
92. Wu WW., Hong HH., Shao XP., Rui L., Jing M. and Lu GD (2014); Effect of oxygen-driven nebulization at different oxygen flows in acute exacerbation of chronic obstructive pulmonary disease patients; Am J Med Sci; 347(5); 343-6.
93. Xu X., Weiss ST., Rijcken B. and Schouten JP (1994); Smoking, changes in smoking habits, and rate of decline in FEV1: new insight into gender differences; Eur Respir J; 7(6); 1056-61.
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT editor-in-chief@pharmatutor.org
Subscribe to Pharmatutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE









