{ DOWNLOAD AS PDF }
 ABOUT AUTHORS
ABOUT AUTHORS
Devender Sharma*1, Aashiya Aara E. Ali 2, Leena R. Trivedi2
1Hi – Tech College of pharmacy, Chandrapur Maharashtra (India)
2Kamla Nehru College of pharmacy, Nagpur, Maharashtra (India)
*sdevender350@gmail.com
ABSTRACT
Liposomes are most placed acquiring in pharma industries and very useful in the various drug delivery system used to target the drug to particular tissue. Because of structural similarity between lipid bilayer (two layer) and cell membrane, liposome can easily penetrate effectively deliver drug to such that a free drug would not easily penetrate. Liposomes can be also encapsulate in both hydrophilic and hydrophobic materials, and are utilized as drug carriers in drug delivery. This technology is very useful for the treatment of certain diseases. Now a day’s most of the researcher attraction and interest will increase for that technology i.e. Liposomes. Main object of this review this technology i.e. Liposomes very useful in certain disease and easily prepare and also give various advantages other than. Liposomes are highly biocompatible, with applications ranging from delivering enzymes, antibacterial, antiviral drugs, antiparasite drugs, transdermal transporters, fungicides, diagnostic tools and adjuvant for vaccines. This paper mainly focus on exclusively scalable techniques and also focus on strength, respectively, limitations in respect to industrial applicability and regulatory requirements concerning liposomal drug formulations based on FDA and EMEA documents.
[adsense:336x280:8701650588]
Reference Id: PHARMATUTOR-ART-2565
|
PharmaTutor (Print-ISSN: 2394 - 6679; e-ISSN: 2347 - 7881) Volume 6, Issue 2 Received On: 02/11/2017; Accepted On: 06/12/2017; Published On: 01/02/2018 How to cite this article: Sharma D, Ali AAE, Trivedi LR; An Updated Review on: Liposomes as drug delivery system; PharmaTutor; 2018; 6(2); 50-62; http://dx.doi.org/10.29161/PT.v6.i2.2018.50 |
INTRODUCTION
Liposomes
Liposomes consist of vesicles composed of bilayers or multilayers that contain or have phospholipids and cholesterol surrounding an aqueous compartment. Drug is entrapped within the liposome and is released from the liposome for absorption at the intestinal membrane surface. This dosage form received considerable and this may well relate to their absorption enhancing ability, the feasibility of their use to promote drug absorption is uncertain drugs or chemical entities. Advances in combinatorial chemistry have led to the discovery of a wide number of new chemical entities (NCE) or drugs that have a potential therapeutic action on the biological systems. But most of the NCEs or drugs being discovered provide a challenge or produce most difficulties to the formulation scientist because of their physicochemical properties like poor solubility and permeability. Even though, above problems or difficulties could be addressed, but most of the molecules do not show or they fail their desired therapeutic action in vivo, which leads to lack of in vitro – in vivo correlation. A majority of anti-neoplastic agents, which are highly cytotoxicity to tumor cells in vitro, affect the normal cells also. This is due to their low therapeutic index (TI), i.e., the dose required to produce anti-tumor effect is toxic to normal cells. Such drugs have to be targeted to a specific site (diseased site) in order to reduce their toxic effects to normal tissues. Hence, an efficient drug delivery system is required to present the maximum fraction of administered dose at the target site or valuable for targeted sites. (Amidon et al., 1995)
Various carriers like nanoparticles, microparticles, polysaccharides, lectins and liposomes can be used to target the drug to a specific site. Liposomal drug delivery is gaining interest due to its contribution to varied areas like drug delivery, cosmetics, and structure of biological membrane. Liposomes very useful because act as a carrier for a variety of drugs, having a potential therapeutic action or other properties. Liposomes are colloidal carriers, having a size range of 0.01–5.0μm in diameter. Indeed these are bilayer vesicles that are formed when phospholipids are hydrated in excess of aqueous medium or aqueous solution. Liposomes have got a potential advantage of encapsulating hydrophilic as well as hydrophobic drugs and targeting them to the phospholipids are hydrated in excess of aqueous medium. Liposomes have got a potential advantage of encapsulating hydrophilic as well as hydrophobic drugs and targeting them to the required diseased site in the body. (Sunil et al., 2005)
Structure of liposome
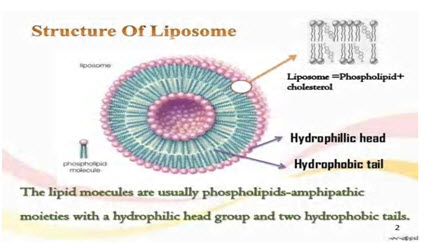
Table: Advantages and disadvantages of liposome (Sharma et al., 1997)
|
Advantages of liposome |
Disadvantages of liposome |
|---|---|
|
Stability increased if liposome prepared via encapsulation |
Short half-life
|
|
Liposomes increased efficacy and therapeutic index of drug (actinomycin-D) |
Low solubility |
|
Liposomes reduce the toxicity of the encapsulated agent (amphotericin B, Taxol) |
Leakage and fusion of encapsulated drug/molecules |
|
Liposomes help reduce the exposure of sensitive tissues to toxic drugs |
Production cost is high |
|
Site avoidance effect |
Fewer stables |
|
Liposomes are flexible, non-toxic, biocompatible, completely biodegradable, and non-immunogenic for systemic and non-systemic administrations |
Sometimes phospholipids undergoes oxidation and hydrolysis-like reaction |
|
Flexibility to couple with site-specific ligands to achieve active targeting |
|
Mechanism of liposome formation:
The basic or important part of liposome is formed by phospholipids, which are amphiphilic molecules (having a hydrophilic head and hydrophobic tail). The hydrophilic part is important it is mainly phosphoric acid bound to a water soluble molecule, whereas, the hydrophobic part consists of two fatty acid chains with 10-24 carbon atoms and 0-6 double bonds in each chain. When these phospholipids are disseminate or dispersed in aqueous medium, they form lamellar sheets by organizing in such a way that, the polar head group faces outwards to the aqueous region while the fatty acid groups face each other and finally form spherical/vesicle like structures called as liposomes. The polar portion remains or residue part in contact or touch with aqueous region along with shielding or keep safe of the non-polar part (which is oriented at an angle to the membrane surface). (Gregoriadis., 1979)
When phospholipids are hydrated in water, along with the input of energy like Sonication, shaking, heating, homogenization, etc. it is the hydrophilic/ hydrophobic interactions between lipid–lipid, lipid–water molecules that lead or show to the formation of bilayer vesicles in order to achieve or arrive at a thermodynamic equilibrium in the aqueous phase. (Gregoriadis., 1979) The main cause for bilayer formation includes:
1. The unfavorable interaction generated between hydrophilic and hydrophobic phase can be minimized or decreased by folding into closed concentric vesicles.
2. Large bilayer vesicle formation assists the reduction of large free energy difference present between the hydrophilic and hydrophobic environment.
3. Maximum stability to super molecular self assembled structure can be attained by forming into vesicles.
Classification of liposomes: (Gregoriadis., 1979)
Various classes of liposomes have been reported in various research and review paper. They are classified based on their size, number of bilayer, composition and method of preparation. Based on the size and number of bilayers, liposomes are classified as multilamellar vesicles (MLV), large unilamellar vesicles (LUV) and small unilamellar vesicles (SUV) as depicted in Fig. 2. Based on composition, they are classified as conventional liposomes (CL), pH-sensitive liposomes, cationic liposomes, long circulating liposomes (LCL) and immuno-liposomes. Based on the method of preparation, they are classified as reverse phase evaporation vesicles (REV), French press vesicles (FPV) and ether injection vesicles (EIV). In this context, the classification based on size and number of bilayer is discussed below. (Albertsson et al., 1985) (Donaruma et al., 1985)
Size range of liposome
|
Types of liposome |
Size range (nm) |
|
Small unilamellar vesicles |
20-40 |
|
Medium unilamellar vesicles |
40-80 |
|
Large unilamellar vesicles |
100-1000 |
Multilamellar vesicles (MLV)
MLV have a size greater than 0.1μm and consists of two or more bilayer. Their method of formulation is simple and very easy to carry which includes thin–film hydration method or hydration of lipids in excess of organic solvent. They are mechanically stable on long storage. Due to the large size, they are cleared early or rapidly by the reticulo-endithelial system (RES) cells and hence can be beneficial for various targeting the organs of RES. MLV have a moderate trapped volume, i.e., amount of aqueous volume to lipid ratio. The drug entrapment or corporate into the vesicles can be enhanced by slower rate of hydration and gentle mixing. Hydrating thin films of dry lipids can also easily enhance encapsulation efficiency. Subsequent lyophilization and rehydration after mixing with the aqueous phase (containing the drug) can yield MLV with good encapsulation efficiency i.e. 40%.
Large unilamellar vesicles (LUV) (Donaruma et al., 1985)
This class of liposomes particularly large unilamellar vesicles consists of a single bilayer and has a size greater than 0.1μm. They have higher encapsulation efficiency, since they can hold a large volume of solution in their cavity.
They have high trapped volume and can be useful for encapsulating hydrophilic drugs. Most useful advantage of LUV is that less amount of lipid is required for encapsulating large quantity of drug. Similar to MLV, they are rapidly cleared by RES cells, due to their larger size. LUV can be formulated by various methods like ether injection, detergent dialysis and reverse phase evaporation techniques. Apart from these methods, freeze thawing of liposomes, dehydration/ rehydration of SUV and slow swelling of lipids in non-electrolyte solution can a prepare LUV.
Small unilamellar vesicles (SUV) (Abra et al., 1981)
SUV are smaller in size (less than 0.1 μm) when compared to MLV and LUV, and have a single bilayer. They have a low entrapped aqueous volume to lipid ratio and characterized by having long circulation half life. SUV can be prepared by using solvent injection method (ethanol or ether injection methods) or alternatively by reducing the size of MLV or LUV using sonication or extrusion process under an inert atmosphere like nitrogen or Argon. The sonication can be performed using either a bath or probe type sonicator. SUV can also be achieved by passing MLV through a narrow orifice under high pressure. These SUV are susceptible to aggregation and fusion at lower or negligible/ no charge.
Methods of preparation: (Abra et al., 1981)
The conventional methods for preparing liposomes include solubilizing the lipids in organic solvent, drying down the lipids from organic solution, dispersion of lipids in aqueous media, purification of resultant liposomes and analysis of the final product. Of all the methods used for preparing liposomes, thin-film hydration method is the most simple and widely used one. MLV are produced by this method within a size range of 1–5μm. If the drug is hydrophilic it is included in the aqueous buffer and if the drug is hydrophobic, it can be included in the lipid film. But the drawback of this method is poor encapsulation efficiency (5-15% only) for hydrophobic drugs. By hydrating the lipids in presence of organic solvent, the encapsulation efficiency of the MLV can be increased. LUV can be prepared by solvent injection, detergent dialysis; calcium induced fusion and reverse phase evaporation techniques. SUV can be prepared by the extrusion or sonication of MLV or LUV. All these preparation methods involve the usage of organic solvents or detergents whose presence even in minute quantities can lead to toxicity. In order to avoid this, other methods like polyol dilution, bubble method and heating method have been developed without using any organic solvents or detergents. Detailed procedures for liposome preparation can be obtained from literature. (Tirrell et al., 1971) (Sahoo et al., 2003)
General methods of preparation (Gabizon et al., 1998)
All the methods of preparing the liposomes or formulating the liposomes involve four basic stages:
1. Drying down lipids from organic solvent.
2. Dispersing the lipid in aqueous media.
3. Purifying the resultant liposome.
4. Analyzing the final product.
Method of liposome preparation and drug loading (Abra et al., 1981)
The following methods are used for the preparation of liposome:
1. Passive loading techniques
2. Active loading technique.
Passive loading techniques include three different methods:
1. Mechanical dispersion method.
2. Solvent dispersion method.
3. Detergent removal method (removal of non encapsulated material)
Mechanical dispersion method (Tirrell et al., 1971)
The following are types of mechanical dispersion methods:
1.1. Sonication.
1.2. French pressure cell: extrusion.
1.3. Freeze-thawed liposomes.
1.4. Lipid film hydration by hand shaking, non-hand, shaking or freeze drying.
1.5. Micro-emulsification.
1.6. Membrane extrusion.
1.7. Dried reconstituted vesicles.
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT editor-in-chief@pharmatutor.org
Subscribe to Pharmatutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE
Sonication
Sonication is helpful the most extensively used method for the preparation of SUV. Here, MLVs are sonicated either with a bath type sonicator or a probe sonicator under a passive atmosphere. The main disadvantages of this method are very low internal volume/ encapsulation efficacy, possible degradation of phospholipids and compounds to be encapsulated, elimination of large molecules, metal pollution from probe tip, and presence of MLV along with SUV. (Sunil et al., 2005)
There are two Sonication techniques: (Tirrell et al., 1971)
1. Probe Sonication.
The tip of a sonicator is directly engaged into the liposome dispersion. The energy input into lipid dispersion is very high in this method. The coupling of energy at the tip results in local hotness; therefore, the vessel must be engrossed into a water/ice bath. Throughout the sonication up to 1 h, more than 5% of the lipids can be deesterified. Also, with the probe sonicator, titanium will slough off and pollute the solution.
2. Bath sonication.
The liposome dispersion in a cylinder is placed into a bath sonicator. Controlling the temperature of the lipid dispersion or scatter is usually easier in this method, in contrast to Sonication by dispersal directly using the tip. The material being sonicate can be protected in a sterile vessel, dissimilar the probe units, or under an inert atmosphere.
French pressure cell: (Sahoo et al., 2003)
Extrusion French pressure cell involves the extrusion of MLV through a small orifice. An important feature of the French press vesicle method is that the proteins do not seem to be significantly pretentious during the procedure as they are in sonication. An interesting comment is that French press vesicle appears to recall entrapped solutes significantly longer than SUVs do, produced by sonication or detergent removal. The method involves gentle handling of unstable materials. The method has several advantages over Sonication method [25]. The resulting liposomes are rather larger than sonicated SUVs. The drawbacks of the method are that the high temperature is difficult to attain, and the working volumes are comparatively small
(about 50 mL as the maximum). Freeze-thawed liposomes SUVs are rapidly frozen and thawed slowly. The short-lived sonication disperses aggregated materials to LUV. The creation of unilamellar vesicles is as a result of the fusion of SUV throughout the processes of freezing and thawing. This type of synthesis is strongly inhibited by increasing the phospholipid concentration and by increasing the ionic strength of the medium. The encapsulation efficacies from 20% to 30% were obtained.
Solvent dispersion method (Tirrell et al., 1971)
Ether injection (solvent vaporization) A solution of lipids dissolved in diethyl ether or ether-methanol mixture is gradually injected to an aqueous solution of the material to be encapsulated at 55°C to 65°C or under reduced pressure. The consequent removal of ether under vacuum leads to the creation of liposomes. The main disadvantages of the technique are that the population is heterogeneous (70 to 200 nm) and the exposure of compounds to be encapsulated to organic solvents at high temperature.
Ethanol injection (Sahoo et al., 2003)
A lipid solution of ethanol is rapidly injected to a huge excess of buffer. The MLVs are at once formed. The disadvantages of the method are that the population is heterogeneous (30 to 110 nm), liposomes are very dilute, the removal all ethanol is difficult because it forms into azeotrope with water, and the probability of the various biologically active macromolecules to inactivate in the presence of even low amounts of ethanol is high.
Reverse phase evaporation method (Sahoo et al., 2003)
This method provided a progress in liposome technology, since it allowed for the first time the preparation of liposomes with a high aqueous space-to-lipid ratio and a capability to entrap a large percentage of the aqueous material presented. Reverse-phase evaporation is based on the creation of inverted micelles. These inverted micelles are shaped upon sonication of a mixture of a buffered aqueous phase, which contains the water-soluble molecules to be encapsulated into the liposomes and an organic phase in which the amphiphilic molecules are solubilized. The slow elimination of the organic solvent leads to the conversion of these inverted micelles into viscous state and gel form. At a critical point in this process, the gel state collapses, and some of the inverted micelles were disturbed. The excess of phospholipids in the environment donates to the formation of a complete bilayer around the residual micelles, which results in the creation of liposomes. Liposomes made by reverse phase evaporation method can be made from numerous lipid formulations and have aqueous volume-to-lipid ratios that are four times higher than hand-shaken liposomes or multilamellar liposomes. Briefly, first, the water-in-oil emulsion is shaped by brief sonication of a two-phase system, containing phospholipids in organic solvent such as isopropyl ether or diethyl ether or a mixture of isopropyl ether and chloroform with aqueous buffer. The organic solvents are detached under reduced pressure, resulting in the creation of a viscous gel. The liposomes are shaped when residual solvent is detached during continued rotary evaporation under reduced pressure. With this method, high encapsulation efficiency up to 65% can be obtained in a medium of low ionic strength for example 0.01 M NaCl. The method has been used to encapsulate small, large, and macromolecules. The main drawback of the technique is the contact of the materials to be encapsulated to organic solvents and to brief periods of sonication. These conditions may possibly result in the breakage of DNA strands or the denaturation of some proteins. Modified reverse phase evaporation method was presented by Handa et al., and the main benefit of the method is that the liposomes had high encapsulation efficiency (about 80%).
Detergent removal method (removal of non-encapsulated material)
Dialysis the detergents at their critical micelle concentrations (CMC) have been used to solubilize lipids. As the detergent is detached, the micelles become increasingly better-off in phospholipid and lastly combine to form LUVs. The detergents were removed by dialysis. A commercial device called LipoPrep (Diachema AG, Switzerland), which is a version of dialysis system, is obtainable for the elimination of detergents. The dialysis can be performed in dialysis bags engrossed in large detergent free buffers (equilibrium dialysis).
Detergent (cholate, alkyl glycoside, Triton X-100) removal of mixed micelles (absorption)
Detergent absorption is attained by shaking mixed micelle solution with beaded organic polystyrene adsorbers such as XAD-2 beads (SERVA Electrophoresis GmbH, Heidelberg, Germany) and Bio-beads SM2 (Bio-Rad Laboratories, Inc., Hercules, USA). The great benefit of using detergent adsorbers is that they can eliminate detergents with a very low CMC, which are not entirely depleted.
Gel-permeation chromatography
In this method, the detergent is depleted by size special chromatography. Sephadex G-50, Sephadex G-l 00 (Sigma-Aldrich, MO, USA), Sepharose 2B-6B, and Sephacryl S200-S1000
(General Electric Company, Tehran, Iran) can be used for gel filtration. The liposomes do not penetrate into the pores of the beads packed in a column. They percolate through the inter-bead spaces. At slow flow rates, the separation of liposomes from detergent monomers is very good. The swollen polysaccharide beads adsorb substantial amounts of amphiphilic lipids; therefore, pretreatment is necessary. The pre-treatment is done by pre-saturation of the gel filtration column by lipids using empty liposome suspensions.
Dilution
Upon dilution of aqueous mixed micellar solution of detergent and phospholipids with buffer, the micellar size and the polydispersity increase fundamentally, and as the system is diluted beyond the mixed micellar phase boundary, a spontaneous transition from poly-dispersed micelles to vesicles occurs.
Stealth liposomes and conventional liposomes
Although liposomes are like biomembranes, they are still foreign objects of the body. Therefore, liposomes are known by the mononuclear phagocytic system (MPS) after contact with plasma proteins. Accordingly, liposomes are cleared from the blood stream. These stability difficulties are solved through the use of synthetic phospholipids, particle coated with amphipathic polyethylene glycol, coating liposomes with chitin derivatives, freeze drying, polymerization, micro-encapsulation of gangliosides. Coating liposomes with PEG reduces the percentage of uptake by macrophages and leads to a prolonged presence of liposomes in the circulation and, therefore, make available abundant time for these liposomes to leak from the circulation through leaky endothelium. A stealth liposome is a sphere-shaped vesicle with a membrane composed of phospholipid bilayer used to deliver drugs or genetic material into a cell. A liposome can be composed of naturally derived phospholipids with mixed lipid chains coated or steadied by polymers of PEG and colloidal in nature. Stealth liposomes are attained and grown in new drug delivery and in controlled release. This stealth principle has been used to develop the successful doxorubicin-loaded liposome product that is presently marketed as Doxil (Janssen Biotech, Inc., Horsham, USA) or Caelyx (Schering- Plough Corporation, Kenilworth, USA) for the treatment of solid tumors. Recently impressive therapeutic improvements were described with the useof corticosteroid loaded liposome in experimental arthritic models. The concerning on the application of stealth liposomes has been on their potential to escape from the blood circulation. However, long circulating liposome may also act as a reservoir for prolonged release of a therapeutic agent. Pharmacological action of vasopressin is formulated in long circulating liposome.
Drug loading in liposomes
Drug loading can be attained either passively (i.e., the drug is encapsulated during liposome formation) or actively (i.e., after liposome formation). Hydrophobic drugs, for example amphotericin B taxol or annamycin, can be directly combined into liposomes during vesicle formation, and the amount of uptake and retention is governed by drug-lipid interactions. Trapping effectiveness of 100% is often achievable, but this is dependent on the solubility of the drug in the liposome membrane.
Passive encapsulation of water-soluble drugs depends on the ability of liposomes to trap aqueous buffer containing a dissolved drug during vesicle formation. Trapping effectiveness (generally <30%) is limited by the trapped volume delimited in the liposomes and drug solubility. On the other hand, water-soluble drugs that have protonizable amine functions can be actively entrapped by employing pH gradients, which can result in trapping effectiveness approaching 100%.
Freeze-protectant for liposomes
Natural excerpts are usually degraded because of oxidation and other chemical reactions before they are delivered to the target site. Freeze-drying has been a standard practice employed to the production of many pharmaceutical products. The overwhelming majority of these products are lyophilized from simple aqueous solutions. Classically, water is the only solvent that must be detached from the solution using the freeze-drying process, but there are still many examples where pharmaceutical products are manufactured via a process that requires freeze-drying from organic co-solvent systems. Freeze-drying (lyophilization) involves the removal of water from products in the frozen state at tremendously low pressures. The process is normally used to dry products that are thermo-labile and would be demolished by heat-drying. The technique has too much potential as a method to solve long-term stability difficulties with admiration to liposomal stability. Studies showed that leakage of entrapped materials may take place during the process of freeze-drying and on reconstitution. Newly, it was shown that liposomes when freeze-dried in the presence of adequate amounts of trehalose (a carbohydrate commonly found at highconcentrations in organism) retained as much as 100% of their original substances. It shows that trehalose is an excellent cryoprotectant (freeze-protectant) for liposomes. Freeze-driers range in size from small laboratory models to large industrial units available from pharmaceutical equipment suppliers.
Mechanism of transportation through liposome (Andreas et al., 2011)
The limitations and benefits of liposome drug carriers lie critically on the interaction of liposomes with cells and their destiny in vivo after administration. In vivo and in vitro studies of the contacts with cells have shown that the main interaction of liposomes with cells is either simple adsorption (by specific interactions with cell-surface components, electrostatic forces, or by nonspecific weak hydrophobic) or following endocytosis (by phagocytic cells of the reticuloendothelial system, for example macrophages and neutrophils). Fusion with the plasma cell membrane by insertion of the lipid bilayer of the liposome into the plasma membrane, with simultaneous release of liposomal content into the cytoplasm, is much rare. The fourth possible interaction is the exchange of bilayer components, for instancecholesterol, lipids, and membrane-bound molecules with components of cell membranes. It is often difficult to determine what mechanism is functioning, and more than one may function at the same time.
Fusogenic liposomes and antibody-mediated liposomes in cancer therapy (Andreas et al., 2011)
It has been infrequently well-known that a powerful anticancer drug, especially one that targets the cytoplasm or cell nucleus, does not work due to the low permeability across a plasma membrane, degradation by lysosomal enzymes through an endocytosis-dependent pathway, and other reasons. Thus, much attention on the use of drug delivery systems is focused on overcoming these problems, ultimately leading to the inductionof maximal ability of anti-cancer drug. In this respect, a new model for cancer therapy using a novel drug delivery system, fusogenic liposome, was developed. Fusogenic liposomes are poised of the ultraviolet inactivated Sendai virus and conventional liposomes. Fusogenic liposomes effectively and directly deliver their encapsulated contents into the cytoplasm using a fusion mechanism of the Sendai virus, whereas conventional liposomes are taken up by endocytosis by phagocytic cells of the reticuloendothelial system, for example macrophages and neutrophils. Thus, fusogenic liposome is a good candidate as a vehicle to deliver drugs into the cytoplasm in an endocytosis-independent manner. Liposomal drug delivery systems provide steady formulation, provide better pharmacokinetics, and make a degree of ‘passive’ or ‘physiological’ targeting to tumor tissue available. However, these transporters do not directly target tumor cells. The design modifications that protect liposomes from unwanted interactions with plasma proteins and cell membranes which differed them with reactive carriers, for example cationic liposomes, also prevent interactions with tumor cells. As an alternative, after extravasation into tumor tissue, liposomes remain within tumor stroma as a drug-loaded depot. Liposomes ultimately become subject to enzymatic degradation and/or phagocytic attack, leading to release of drug for subsequent diffusion to tumor cells. The next generation of drug carriers under development features directs molecular targeting of cancer cells via antibody-mediated or other ligand-mediated interactions.
Characterization of liposomes:
Liposomes produced by different methods have varying physicochemical characteristics, which leads to differences in their in vitro (sterilization and shelf life) and in vivo (disposition) performances. Rapid, precise and reproducible quality control tests are required for characterizing the liposomes after their formulation and upon storage for a predictable in vitro and in vivo behavior of the liposomal drug product. A liposomal drug product can be characterized for some of the parameters that are discussed below. (Chrai et al., 2001)
Size and size distribution
When liposomes are intended for inhalation or parenteral administration, the size distribution is of primary consideration, since it influences the in vivo fate of liposomes along with the encapsulated drug molecules. Various techniques of determing the size of the vesicles include microscopy (optical microscopy, negative stain transmission electron microscopy, cryo-transmission electron microscopy, freeze fracture electron microscopy and scanning electron microscopy, diffraction and scattering techniques (laser light scattering and photon correlation spectroscopy) andhydrodynamic techniques (field flow fractionation, gel permeation and ultracentrifugation).
Percent drug encapsulation
The amount of drug encapsulated/ entrapped in liposome vesicle is given by percent drug encapsulation. Column chromatography can be used to estimate the percent drug encapsulation of liposomes. The formulation consists of both free (unencapsulated) and encapsulated drug. So as to know the exact amount of drug encapsulated, the free drug is separated from the encapsulated one. Then the fraction of liposomes containing the encapsulated drug is treated with a detergent, so as to attain lysis, which leads to the discharge of the drug from the vesicles into the surrounding medium. (Andreas et al., 2011) This exposed drug is assayed by a suitable technique which gives the percent drug encapsulated from which encapsulation efficiency can be calculated. Trapped volume per lipid weight can also give the percent drug encapsulated in a liposome vesicle. It is generally expressed as aqueous volume entrapped per unit quantity of lipid, μl/μmol or μg/mg of total lipid. In order to determine the trapped volume, various materials like radioactive markers, fluorescent markers and spectroscopically inert fluid can be used. Radioactive method is mostly used for determining trapped volume. It is determined by dispersing lipid in an aqueous medium containing a non-permeable radioactive solute like [22Na] or [14C] inulin. Alternatively, water soluble markers like 6-carboxyfluorescein, 14C or 3H-glucose or sucrose can be used to determine the trapped volume. A novel method of determining intravesicular volume by salt entrapment was also reported in literature.
Surface charge (Chrai et al., 2001)
Since the charge on the liposome surface plays a key role in the in vivo disposition, it is essential to know the surface charge on the vesicle surface. Two methods namely, free-flow electrophoresis and zeta potential measurement can be used to estimate the surface charge of the vesicle. The surface charge can be calculated by estimating the mobility of the liposomal dispersion in a suitable buffer (determined using Helmholtz–Smolochowski equation).
Vesicle shape and lamellarity
Various electron microscopic techniques can be used to assess the shape of the vesicles. The number of bilayers present in the liposome, i.e., lamellarity can be determined using freeze fracture electron microscopy and 31P-Nuclear magnetic resonance analysis64. Apart from knowing the shape and lamellarity, the surface morphology of liposomes can be assessed using freeze-fracture and freeze-etch electron microscopy.
Phospholipid identification and assay (Chrai et al., 2001)
The chemical components of liposomes must be analyzed prior to and after the preparation. Barlett assay, Stewart assay and thin layer chromatography can be used to estimate the phospholipid concentration in the liposomal formulation. A spectrophotometric method to quantify total phosphorous in a sample was given in literature, which measure the intensity of blue color developed at 825 nm against water. Cholesterol oxidase assay or ferric perchlorate method and Gas liquid chromatography techniques can be used to determine the cholesterol concentration.
Stability of liposomes:
During the development of liposomal drug products, the stability of the developed formulation is of major consideration. The therapeutic activity of the drug is governed by the stability of the liposomes right from the manufacturing steps to storage to delivery. A stable dosage forms is the one which maintains the physical stability and chemical integrity of the active molecule during its developmental procedure and storage. A well designed stability study includes the evaluation of its physical, chemical and microbial parameters along with the assurance of product’s integrity throughout its storage period. Hence a stability protocol is essential to study the physical and chemical integrity of the drug product in its storage. (Johnston et al., 2007)
Physical stability
Liposomes are bilayered vesicles that are formed when phospholipids are hydrated in water. The vesicles obtained during this process are of different sizes. During its storage, the vesicles tend to aggregate and increase in size to attain thermodynamically favorable state. (Andreas et al., 2011) During storage, drug leakage from the vesicles can occur due to fusion and breaking of vesicles, which deteriorates the physical stability of the liposomal drug product. Hence morphology, size and size distribution of the vesicles are important parameters to assess the physical stability. In order to monitor this, a variety of techniques like light scattering and electron microscopy can be used to estimate the visual appearance (morphology) and size of the vesicles.
Chemical stability
Phospholipids are chemically unsaturated fatty acids that are prone to oxidation and hydrolysis, which may alter the stability of the drug product. Along with this, pH, ionic strength, solvent system and buffered species also play a major role in maintaining a liposomal formulation. Indeed chemical reaction can be induced even by light, oxygen, temperature and heavy metal ions. Oxidation deterioration involves the formation of cyclic peroxides and hydroxyperoxidases due to the result of free radical generation in the oxidation process. Liposomes can be prevented from oxidative degradation by protecting them from light, by adding anti-oxidants such as alpha – tocopherol or butylated hydroxyl toluene (BHT), producing the product in an inert environment (presence of nitrogen or Argon) or by adding EDTA to remove trace heavy metals. Hydrolysis of the ester bond at carbon position of the glycerol moiety of phospholipids leads to the formation of lyso-phosphatidylcholine (lysoPC), which enhances the permeability of the liposomal contents. Hence, it becomes necessary to control the limit of lysoPC within the liposomal drug product. This can be achieved by formulating liposomes with phosphatidylcholine free from lysoPC. (Johnston et al., 2007)
In vivo behavior of liposomes:
During the optimization of liposomal formulation, various physico-chemical parameters are altered in order to achieve a desired bio-distribution and cellular uptake of drugs. Those parameters which affect the in vivo (biological) performance of liposomes are described below.
Liposome size (Allen. 1997)
The size of the vesicle governs the in vivo fate of liposomes, because it determines the fraction cleared by RES. The rate of uptake of liposome by RES increases with the vesicle size. Liposomes larger than 0.1μm are taken up (opsonized) more rapidly by RES, when compared to liposomes smaller than 0.1μm. The size of the vesicle also determines the extravasations of liposomes. Tumor capillaries are more permeable than normal capillaries. Due to such leaky vasculature, fluids along with small sized liposomes can pass through the gaps leading to increased accumulation of drug loaded liposomes in the tumor tissue. The difference between intravascular hydrostatic and interstitial pressure acts as a driving force for the extravasations of small sized liposomes.
Surface charge (Andreas et al., 2011)
The lipid – cell interaction can be governed by the nature and density of charge on the liposome surface. Charging the lipid composition can alter the nature and charge on the liposome. Lack of charge in the SUV liposomes can lead to their aggregation and thereby reducing the stability of the liposome; whereas, the interaction of neutrally charged liposome with the cell is almost negligible. High electrostatic surface charge on the liposome may provide useful results in promoting lipid – cell interaction. Negatively charged density influences the extent of lipid – cell interactions and increase the intracellular uptake of liposomes by target cells. But positively charged liposomes are cleared more rapidly after systemic administration. Unlike negatively charged liposomes, cationic liposomes deliver the contents to cells by fusion with cell membrane. (Johnston et al., 2007)
Surface hydration (Andreas et al., 2011)
Liposomes with hydrophilic surface coatings are less prone to opsonization, hence reducing its uptake by RES cells. This can be attributed to the hydrophilic surface coating, which reduces the interaction of liposomes with cell and blood components. These sterically stabilized liposomes are more stable in the biological environment and exhibit high circulation half lives, when compared to liposomes coated with hydrophobic coatings. Monogangliosides, hydrogenated phosphotidyl inositol, polyethylene glycol are some of the hydrophilic groups responsible for steric stabilization of liposomes.
Bilayer fluidity (Chrai et al., 2001)
Lipid exists in different physical states above and below the phase transition temperature (Tc). They are rigid and well ordered below Tc but are in fluid like liquid – crystalline state above Tc. indicates the phase transition temperatures of various phospholipids. Liposomes with low Tc (less than 37°C) are fluid like and are prone to leakage of the drug content at physiological temperature. But, the liposomes with high Tc (greater than 37°C) are rigid and less leaky at physiological temperature. The phase transition temperature also governs the liposomal cell interaction. Liposomes with low Tc lipids have high extent of uptake by RES when compared to those with high Tc lipids. Incorporation of cholesterol in the bilayer can decrease the membrane fluidity at a temperature greater than phase transition temperature, which gives stability to liposomes.
Therapeutic applications of liposomes: (Chrai et al., 2001)
When a conventional dosage form fails to provide a desired therapeutic effect, then new drug delivery systems are developed. Liposomes are among such systems which provide a superior therapeutic efficacy and safety in comparison to existing formulations. Some of the major therapeutic applications of liposomes in drug delivery include: (Johnston et al., 2007)
Site-avoidance delivery (Chrai et al., 2001)
The cytotoxicity of anti-cancer drugs to normal tissues can be attributed to their narrow therapeutic index (TI). Under such circumstances, the TI can be improved by minimizing the delivery of drug to normal cells by encapsulating in liposomes. Free doxorubicin has a severe side effect of cardiac toxicity, but when formulated as liposomes, the toxicity was reduced without any change in the therapeutic activity.
Site specific targeting (Chrai et al., 2001)
Delivery of larger fraction of drug to the desired (diseased) site, by reducing the drug’s exposure to normal tissues can be achieved by site specific targeting. Encapsulating the drug in liposomes can be used for both active and passive targeting of drugs in order to achieve a safer and efficacious therapy. On systemic administration, long circulating immunoliposomes are able to recognize and bind to target cells with greater specificity. In patients with recurrent osteosarcoma, there was an enhanced tumoricidal activity of monocytes, when muramyl peptide derivatives were formulated as liposomes and administered systemically. (Benech et al., 2002)
Intracellular drug delivery (Atrooz., 2011)
Increased delivery of potent drugs to the cytosol (in which drug’s receptors are present), can be accomplished using liposomal drug delivery system. N-(phosphonacetyl)-L-aspartate (PALA) is normally poorly taken up into cells. Such drugs when encapsulated within liposomes, showed greater activity against ovarian tumor cell lines in comparison to free drug [76].
Sustained release drug delivery (Benech et al., 2002)
Liposomes can be used to provide a sustained release of drugs, which require a prolonged plasma concentration at therapeutic levels to achieve the optimum therapeutic efficacy. Drugs like cytosine Arabinoside can be encapsulated in liposomes for sustained release and optimized drug release rate in vivo.
IntraperitoneaI administration (Shehata et al., 2008)
Tumors that develop in the intra-peritoneal (I.P.) cavity can be treated by administering the drug to I.P. cavity. But the rapid clearance of the drugs from the I.P. cavity results in minimized concentration of drug at the diseased site. However, liposomal encapsulated drugs have lower clearance rate, when compared to free drug and can provide maximum fraction of drug in a prolonged manner to the target site.
Immunological adjuvants in vaccines (Shehata et al., 2008)
Immune response can be enhanced by delivering antigens encapsulated within liposomes. Depending on the lipophilicity of antigens, the liposome can accommodate antigens in the aqueous cavity or incorporate within the bilayers. In order to enhance the immune response to diphtheria toxoid, liposomes were first used as immunological adjuvants.
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT editor-in-chief@pharmatutor.org
Subscribe to Pharmatutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE
Liposomes in anticancer therapy (Johnston et al., 2007)
Numerous or various type of liposome formulations of numerous anticancer agents were shown to be less toxic than the free drug. Anthracyclines are drugs which stop the growth of dividing cells by intercalating into the DNA and, thus, kill mainly rapidly dividing cells. These cells are not only in tumors but are also in hair, gastrointestinal mucosa, and blood cells; therefore, this class of drug is very toxic. The most used and studied is Adriamycin (commercial name for doxorubicin HCl; Ben Venue Laboratories, Bedford, Ohio). In addition to the above-mentioned acute toxicities, its dosage is limited by its increasing cardio toxicity. Numerous diverse formulations were tried. In most cases, the toxicity was reduced to about 50%. These include both acute and chronic toxicities because liposome encapsulation reduces the delivery of the drug molecules towards those tissues. For the same reason, the efficiency was in many cases compromised due to the reduced bioavailability of the drug, especially if the tumor was not phagocyte or located in the organs of mononuclear phagocyte system. In some cases, such as systemic lymphoma, the effect of liposome encapsulation showed enhanced efficacy due to the continued release effect, i.e., longer presence or long time of therapeutic concentrations in the circulation, while in several other cases, the sequestration of the drug into tissues of mononuclear phagocyte system actually reduced its efficacy. Applications in man showed, in general, reduced toxicity and better tolerability of administration with not too encouraging efficacy. Several different formulations are in different phases of clinical studies and show mixed results which are very useful.
Table: Benefits of drug load in liposomes (Johnston et al., 2007)
|
Benefits of drug load in liposome |
Examples |
|---|---|
|
Improved solubility of lipophilic and amphiphilic drugs |
Minoxidil, amphotericin B, porphyrins, some peptides, and anthracyclines, respectively; hydrophilic drugs, such as anticancer agent doxorubicin or acyclovir |
|
Sustained release system of systemically or locally administered liposomes |
cortisones , Doxorubicin, cytosine arabinoside, biological proteins or peptides such as vasopressin |
|
Passive targeting to the cells of the immune system, especially cells of the mononuclear phagocytic system |
Antimonials, amphotericin B, porphyrins, vaccines, immunomodulators |
|
Site-avoidance mechanism |
Doxorubicin andamphotericin B |
|
Site-specific targeting |
Anti-inflammatory drugs, anti-cancer, anti-infection |
|
Improved penetration into tissues |
,Anesthetics, corticosteroids and insulin |
|
Improved transfer of hydrophilic, charged molecules |
Antibiotics, chelators, plasmids, and genes |
CONCLUSION:
A number of drug candidates or chemical molecules which are highly potent and have low therapeutic indication can be targeted to the required diseased site using the liposomal drug delivery system. Liposomes have been used in a broad range of pharmaceutical applications. Drugs encapsulated in liposomes can have a significantly altered pharmacokinetics. The efficacy of the liposomal formulation depends on its ability to deliver the drug molecule to the targeted site over a prolonged period of time, simultaneously reducing its (drug’s) toxic effects. The drugs are encapsulated within the phospholipids bilayer and are expected to diffuse out from the bilayer slowly. Various factors like drug concentration, drug to lipid ratio, encapsulation efficiency and in vivo drug release must be considered during the formulation of liposomal drug delivery systems. Finally, liposomal drugs exhibit reduced toxicities and retain enhanced efficacy compared with free complements. The development of deformable liposomes and ethosomes along with the administration of drug loaded liposomes through inhalation and ocular route are some of the advances in the technology and very useful in pharma Industries. Thus liposomal approach can be successfully utilized to improve the pharmacokinetics and therapeutic efficacy, simultaneously reducing the toxicity of various highly potent drugs. However, property of liposome based on the pharmaceutical applications and available products, we can say that liposomes have not definitely but surely most acquire space in pharma Industries and also established their position in modern delivery systems.
REFERENCES:
1. Abra, R.M., Hunt, C.A. Liposome disposition in vivo. III. Dose and vesicle size effects, Biochim Biophys Acta. 1981; 666: 493-503.
2. Albertsson, A.C., Donaruma, L.G., Vogl, O. Synthetic polymers as drugs, In Tirrell, D.A., Donaruma, L.G., Turek, A.B., (Eds.), Macromolecules as drugs and drug as carriers for biologically active material. Ann NY Acad Sci. 1985; 446: 105-115.
3. Allen TM: Liposomes. Opportunities in drug delivery. Drugs 1997, 54(Suppl 4):8–14.
4. Amidon, G.L., Lennernas, H., Shah, V.P., Crison, J.R. A theoretical basis for a biopharmaceutic drug classification: the correlation of in vitro drug product dissolution and in vivo bioavailability. Pharm Res. 1995; 12: 413-420.
5. Andreas W, Karola VU: Liposome technology for industrial purposes. J Drug Delivery 2011, 2011:9.
6. Atrooz OM: Effects of alkylresorcinolic lipids obtained from acetonic extract of Jordanian wheat grains on liposome properties. Int J Biol Chem 2011, 5(5):314–321.
7. Benech RO, Kheadr EE, Laridi R, Lacroix C, Fliss I: Inhibition of Listeria innocua in cheddar cheese by addition of nisin Z in liposomes or by in situ production in mixed culture. Applied Environ Microbiol 2002, 68:3683–3690.
8. Chrai SS, Murari R, Imran A: Liposomes: a review. Bio Pharm 2001,14(11):10–14.
9. Donaruma, L.G., Warner, R.J. Some biologically active (thiosemicarbazides). In Tirrell, D.A., Donaruma, L.G. and Turek, A.B. (Eds.), Macromolecules as drugs and drug as carriers for biologically active materials. Ann NY Acad Sci. 1985; 446: 116-133.
10. Gabizon A, Goren D, Cohen R, Barenholz Y: Development of liposomal anthracyclines: from basics to clinical applications. J Control Release 1998, 53:275–279.
11. Gregoriadis, G. Liposomes, In Gregoriadis, G., (Ed.), Drug Carriers in Biology and Medicine. Academic Press, New York. 1979; Ch. 14. Page 287- 341.
12. Johnston MJ, Semple SC, Klimuk SK, Ansell S, Maurer N, Cullis PR: Characterization of the drug retention and pharmacokinetic properties of liposomal nanoparticles containing dihydrosphingomyelin. Biochim Biophys Acta 2007, 1768:1121–1127.
13. Sahoo SK, Labhasetwar V: Nanotech approaches to drug delivery and imaging. DDT 2003, 8:24.
14. Sharma, A., Sharma, U.S. Liposomes in drug delivery: progress and limitations. Int J Pharm. 1997; 154: 123-140.
15. Shehata T, Ogawara K, Higaki K, Kimura T: Prolongation of residence time of liposome by surface-modification with mixture of hydrophilic polymers. Int J Pharm 2008, 359:272–279.
16. Sunil, P., Maru, O., Chan, M. Novel lipid-based formulations enhancing the in vitro dissolution and permeability characteristics of a poorly water-soluble model drug, piroxicam. Int J Pharm. 2005; 301(1-2): 209-216.
17. Tirrell, D.A., Heath, T.D., Colley, C.M., Ryman, B.E. New aspects of liposomes, Biochim Biophys Acta. 1976; 457: 259.
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT editor-in-chief@pharmatutor.org
Subscribe to Pharmatutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE









