Scientists identified an over-the-counter antihistamine as a treatment for multiple sclerosis
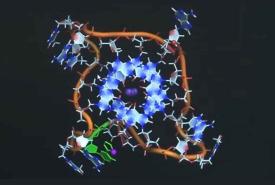
A decade after UC San Francisco scientists identified an over-the-counter antihistamine as a treatment for multiple sclerosis, researchers have developed an approach to measure the drug’s effectiveness in repairing the brain, making it possible to also assess future therapies for the devastating disorder.
















.png)


