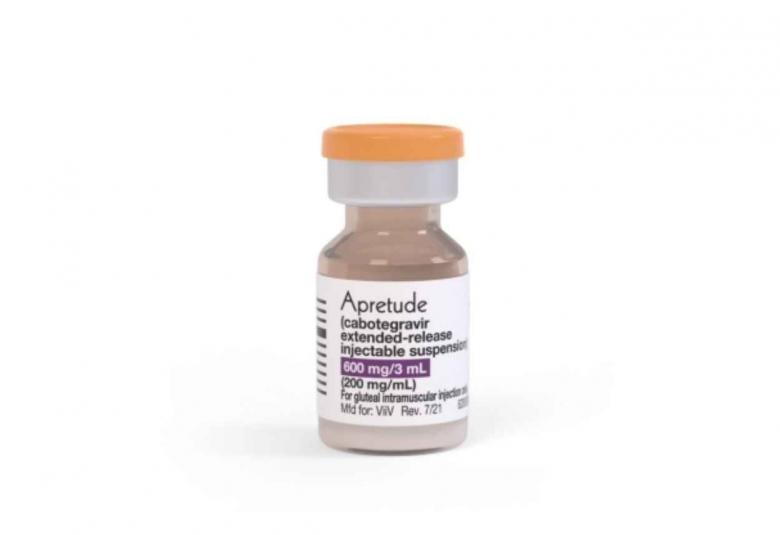
ViiV Healthcare announces USFDA approval of Apretude
ViiV Healthcare, the global specialist HIV company majority owned by GlaxoSmithKline plc (GSK), with Pfizer Inc and Shionogi Limited (Shionogi) as shareholders, today announced that the US Food and Drug Administration (FDA) approved Apretude, the first and only long-acting injectable pre-exposure prophylaxis (PrEP) option to reduce the risk of sexually acquired HIV-1. The long-acting injectable was approved for use in adults and adolescents weighing at least 35 kg who are at risk of sexually acquiring HIV and who have a negative HIV-1 test prior to initiation.

















.png)


