About Authors: Manoj Kumar Jadia1*, Dr. U. L. Narayan2
1. Department of Pharmaceutical Chemistry,
Indira Gandhi institute of Pharmaceautical Sciences,
IRC village, Bhubaneswar, Odhisa, India
2. Principal, Department of Pharmaceutical Chemistry,
Indira Gandhi institute of Pharmaceautical Sciences,
IRC village, Bhubaneswar, Odhisa, India
Abstract
The two methods are described for the simultaneous determination of Paracetamol and Etodolac in binary mixture. The first method was based on UV-spectrophotometric determination of both of the drugs, using simultaneous equation method. It involves absorbance measurement at 256.0 nm (λmax of Paracetamol) and 226.0 nm (λmax of Etodolac) in methanol; linearity was obtained in the range of 5 – 25 μg.mL-1 for both the drugs. The second method was based on HPLC separation of the two drugs in reverse phase mode using Promosil C18 column. Linearity was obtained in the concentration range of 30-70μg.mL-1 for Paracetamol and 20-60 μg.mL-1 for Etodolac. The LOD and LOQ value of UV-Spectrophotometric determination was found to be 167.43 ng mL-1, 507.37 ng mL-1 and for HPLC determination was found to be 1653.12 ng mL-1, 5009.48 ng mL-1.Both these methods have beensuccessively applied to pharmaceutical formulation and were validated according to ICH guidelines.
[adsense:336x280:8701650588]
Reference ID: PHARMATUTOR-ART-1090
1. Introduction:
Paracetamol chemically N- (4-hydroxyphenyl)-acetamide[1], is a widely used over-the-counter analgesic (pain reliever) and antipyretic (fever reducer). It is commonly used for the relief of headaches, other minor aches and pains, and is a major ingredient in numerous cold and flu remedies. In combination with opioid analgesics, paracetamol can also be used in the management of more severe pain such as post surgical pain and providing palliative care in advanced cancer patients.[2] The onset of analgesia is approximately 11 minutes after oral administration of paracetamol,[3] and its half-life is 1–4 hours. Its therapeutic effects are similar to salicylates, but it lacks anti-inflammatory, antiplatelet, and gastric ulcerative effects.[4] It is the active metabolite of phenacetin, once popular as an analgesic and antipyretic in its own right, but unlike phenacetin and its combinations, paracetamol is not considered to be carcinogenic at therapeutic doses .[1] Paracetamol increases the risk of liver damage in chronic alcoholics. Increased risk of toxicity with other hepatotoxic drugs or drugs which induce microsomal enzymes e.g. barbiturates, carbamazepine, hydantoins, rifampicin and sulfinpyrazone.[5] side effects of recommended doses (up to 8 tablets/capsules a day in adults) are rare, but rashes or other allergic reactions may rarely occur; pancreatitis has been reported after prolonged use. In higher than recommended doses paracetamol causes liver damage, which may not be apparent for two days or more and can be fatal unless treated immediately with the antidote (methionine or acetylcysteine).[6] The chemical structure of Paracetamol is shown in Fig 1.[1] The recommended adult dose of paracetamol is two 500 mg tablets, with four hours between doses, and no more than eight tablets in 24 hours.[7] Literature survey reveled that various analytical methods such as HPLC, GC-MS , LC-MS HPLC-Electrospray tendem mass spectrometry and HPTL have been reported for estimation of Paracetamol from its formulations and biological fluids.
Etodolac chemically (RS)-2-(1,8-Diethyl-4,9-dihydro-3H-pyrano[3,4-b]indol-1-yl)acetic acid[8], is a Non-steroidal anti-inflammatory drug [10], Other members of this class include aspirin, ibuprofen (Motrin, Advil, Nuprin, etc.), naproxen (Aleve, Naprosyn), indomethacin (Indocin), nabumetone (Relafen) and numerous others.[8] These drugs are used for the management of mild to moderate pain, fever, and inflammation. They work by reducing the levels of prostaglandins, which are chemicals that are responsible for pain and the fever and tenderness that occur with inflammation. Etodolac blocks the enzyme that makes prostaglandins (cyclooxygenase), resulting in lower concentrations of prostaglandins. As a consequence, inflammation, pain and fever are reduced. The FDA approved etodolac in January 1991.[9] Etodolac is a non-steroidal anti-inflammatory drug (NSAID) with anti-inflammatory, analgesic and antipyretic properties. Its therapeutic effects are due to its ability to inhibit prostaglandin synthesis. It is indicated for relief of signs and symptoms of rheumatoid arthritis and osteoarthritis.[10] Additional studies in human indicated that Etodolac does not affect serum fat – soluble vitamins. The recommended dose of Etodolac for acute pain relief is 200 mg to 400 mg, taken every six to eight hours. Capsules: 200 and 300 mg; Tablets: 400 and 500 mg; Extended Release: 400, 500 and 600 mg. The maximum recommended daily dosage of etodolac is 1000 mg. However, some patients have been found to benefit from daily dosage of 1200 mg. Daily dosages of Etodolac exceeding 1200 mg have not been evaluated.[9]. Non-steroidal anti-inflammatory drug (NSAID) that selectively inhibits cyclooxygenase-2 (COX-2) (IC 50 values are 53 and >100 μ M for COX-2 and COX-1 respectively). Displays anti-inflammatory effects in both adjuvant arthritic and normal rats.[11] The structure of Etodolac is shown in Fig 2.[8] There are very few methods reported for estimation of Etodolac in pharmaceutical dosage form, which includes a validated RP –HPLC, spectrophotometric method.
At present no HPLC and UV spectrophotometric methods are reported for the simultaneous estimation of Paracetamol and Etodolac in tablet formulation.
Therefore, it was thought worthwhile to develop simple, precise, accurate UVspectrophotometric and HPLC methods for simultaneous determination of Paracetamol and Etodolac in tablets.
[adsense:468x15:2204050025]
2. Experimental:
2.1. Materials:
Pharmaceutical grade Paracetamol and Etodolac were kindly supplied by Burgeon Pharmaceauticals PVT LTD.Puducherry (INDIA), used without further purification and certified to contain 99.53 % (w/w) and 99.66% (w/w), respectively on dried basis. All chemicals are of HPLC grade and were purchased from Merck Ltd. And India Rankem Laboratories, India.
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT articles@pharmatutor.org
Subscribe to Pharmatutor Job Alerts by Email
2.2. UV- spectrophotometry:
A Thermospectronic model is Systronics-108 (Double beam) Spectrophotometer with 1cm. matched quartz cells was used. Standard stock solutions of Paracetamol of 1000 μg.mL-1 were prepared by dissolving 100 mg of paracetamol in 100 mL of methanol, From these stock solutions, working standard solutions having concentration 20,30,40,50,60 μg.mL-1 and Standard stock solutions of Etodolac of 1000 μg.mL-1 were prepared by dissolving 100 mg of Etodolac in 100 mL of methanol, From these stock solutions, working standard solutions having concentration 30,40,50,60,70 μg.mL-1each were prepared by appropriate dilutions. They were scanned in the wavelength range of 400−200 nm and the overlain spectrum was obtained (Fig 3). Two wavelengths 256.0 nm (λmax of Paracetamol) and 226.0 nm (λmax of Etodolac) were selected for the formation of simultaneous equation. The calibration curves were found to be linear in the concentration range of 20-60 μg.mL-1 for Paracetamol and 30-70 μg.mL-1 for Etodolac. The absorptivity coefficients of each drug at both wavelengths were determined. The concentration of two drugs in the mixture were calculated using equations [12],
CPCM = A2 ay1 – A1 ay2/ ax2 ay1 – ax1ay2 ……………… … (1)
CETO = A1 ax2 – A2 ax2/ ax2 ay1 – ax1ay2…………………... (2)
Where, A1 and A2 are absorbance of mixture at 256.0 nm and 226.0 nm; ax1 and ax2,
absorptivities of Paracetamol at 256.0 nm and 226.0 nm, respectively; ay1 and ay2
absorptivities of Etodolac at 256.5 nm and 226.0 nm, respectively. CPCM and CETO are
concentration of Paracetamol and Etodolac in mixture. The absorptivities reported are the
mean of six independent determinations (Table 1).
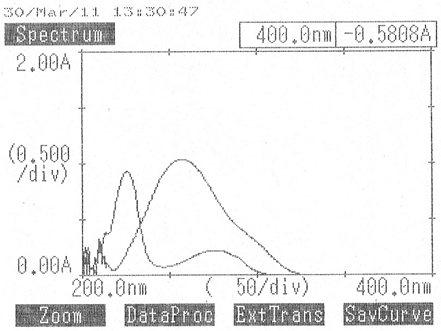
Fig 3. Overlain Spectrum of Paracetamol And Etodolac in Methanol taken on Systronics-108 (Double beam) Spectrophotometer
2.3. HPLC method:
The HPLC system consisted of a YL-9100 pump, a loop injector of injection capacity 20 μL, Detector consists of Photodiode array detector, a Promosil C18 (250 X 4.60 mm) 5μm column, a HPLC guard cartridge system and a YL Clarity software at ambient temperature.
Different mobile phases were tested in order to find the best conditions, for separating both the drugs simultaneously. The optimal composition of mobile phase was determined to be Acetonitrile: Methanol (23:77, v/v). The flow rate was set to 1.5 mL.min-1 and UV detection was carried out at 239 nm.
Stock solution was prepared by dissolving 100 mg of Paracetamol and Etodolac in 100 mL was taken in separate two100 mL volumetric flask for each drug by using Acetonitrile: Methanol (23:77, v/v) as solvent.
Table 1. Absorptivity Values at 256.0 nm (λmax of Paracetamol) and 226.0 nm (λmax of Etodolac)
|
|
Absorptivity at 226.0 nm |
Absorptivity at 256.0 nm |
||
|
|
Paracetamol |
Etodolac |
Paracetamol |
Etodolac |
|
*Mean |
362.25 |
514.23 |
472.45 |
408.28 |
|
± S.D. |
1.17 |
0.51 |
0.54 |
0.48 |
* Absorptivity values are the mean of six determinations. S.D. is standard deviation. ax1 and ax2 absorptivities of at 256.0 nm and 226.0 nm, respectively; ay1 and ay2 absorptivities of Etodolac at 226.0 nm and 256.0 nm, respectively.
All solutions were stored at + 50C in the dark; these solutions were shown to be stable during the period of study.
From the above stock solutions, dilutions were made in the concentration range of 20–60 μg.mL–1 of Paracetamol and 30-70 μg.mL–1 Etodolac. A volume of 20 μL of each sample was injected into column. All measurements were repeated three times for each concentration and calibration curve was constructed by plotting the peak area ratios of analyte to the corresponding drug concentration.
2.4. Analysis of Pharmaceutical Dosage Forms
To determine the content of Paracetamol and Etodolac simultaneously in tablets (label claim: 100 mg Paracetamol and 100 mg Etodolac, film coated); twenty tablets were weighed; their average weight determined and were finely powdered. The correct amount of powder was dissolved in Acetonitrile: Methanol (23:77, v/v). by stirring for 30 min. The excipients were separated by filtration. After filtration, an appropriate amount of internal standard was added and diluted up to mark with Acetonitrile: Methanol (23:77, v/v). Appropriate aliquots were subjected to above methods and the amount of Paracetamol and Etodolac were determined. The results are reported in Table 2.
Table 2. Analysis data of tablet formulations
|
Parameters |
UV – spectrophotometry |
HPLC |
||
|
Paracetamol |
Etodolac |
Paracetamol |
Etodolac |
|
|
Label Claim |
500 |
400 |
500 |
400 |
|
*Drug content |
99.96 |
99.37 |
100.3 |
100.0 |
|
± S. D. |
0.450 |
0.625 |
0.624 |
1.000 |
|
% R.S.D. |
0.451 |
0.628 |
0.622 |
1.000 |
* Value for Drug content (%) are the mean of five estimations; S.D. is standard deviation and R.S.D. is relative standard deviation
2.5. Recovery studies
To check the accuracy of the developed methods and to study the interference of formulation additives, analytical recovery experiments were carried out by standard addition method, at 80, 100 and 120 % level. From the total amount of drug found, the percentage recovery was calculated. The results are reported in Table 3.
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT articles@pharmatutor.org
Subscribe to Pharmatutor Job Alerts by Email
3. Results and Discussion
Both, UV spectrophotometric and HPLC methods were found to be simple, accurate, economic and rapid for routine simultaneous estimation of Paracetamol and Etodolac, in tablet dosage forms. For UV spectrophotometric method, linearity were obtained in concentration range of 20 – 60 μg .mL-1, for the drug Paracetamol with regression 0.9982 and 30 – 70 μg .mL-1, for the drug Etodolac with regression 0.998, intercept + 0.0133 and + 0.008 and slope 0.0122 and 0.0013 for Paracetamol and Etodolac, respectively. Recovery was in the range of 99 – 101 %; the value of standard deviation and % R.S.D. were found to be < 2 %; shows the high precision of the method.
Table 3. Recovery studies
|
UV – spectrophotometry |
HPLC |
||||
|
Excess Drug |
*Recovery |
% R.S.D. |
Excess Drug |
*Recovery |
% R.S.D. |
|
Paracetamol |
|||||
|
80 100 120 |
99.83 99.72 99.07 |
0.2953 0.2026 0.0672 |
80 100 120 |
99.37 100.11 100.58 |
0.9405 0.0115 0.9702 |
|
Etodolac |
|||||
|
80 100 120 |
100.69 100.43 99.52 |
0.2953 0.1036 0.1165 |
80 100 120 |
100.32 99.33 98.80 |
1.1238 0.0232 1.1243 |
* Recovery is mean of three estimations.
In HPLC method, HPLC conditions were optimized to obtain, an adequate separation of eluted compounds. Initially, various mobile phase compositions were tried, to separate drugs and internal standard. Mobile phase and flow rate selection was based on peak parameters (height, tailing, theoretical plates, capacity factor), run time etc. The system with Acetonitrile: Methanol (23:77, v/v) with 1.5 mL.min-1 flow rate is quite robust.
A typical chromatogram for Paracetamol and Etodolac is shown in fig 4. The optimum wavelength for detection was 239 nm at which better detector response for drugs was obtained. The average retention times for Paracetamol and Etodolac were found to be 11.76 ± 0.03 and 15.59 ± 0.03 min, respectively. According to USP XXIV (621), system suitability tests are an integral part of chromatographic method. They are used to verify the reproducibility of the chromatographic system. To ascertain its effectiveness, system suitability tests were carried out on freshly prepared stock solutions. The parameters obtained are shown in Table 4. The calibration linearity were obtained in concentration range of 20 – 60 μg .mL-1, for the drug Paracetamol with regression 0.998 and 30 – 70 μg .mL-1, for the drug Etodolac with regression 0.998, intercept – 92159 and + 98894 and slope 10050 and 11853 for Paracetamol and Etodolac, respectively. The low values of % R.S.D. indicate the method is precise and accurate. The mean recoveries were found in the range of 98 – 100 %.
Table 4. System suitability parameters
|
Parameters |
Paracetamol |
Etodolac |
|
Tailing factor |
1.68 |
1.43 |
|
Theoretical Plates |
2579 |
2753 |
|
Resolution factor |
2.16 |
2.81 |
|
Capacity factor |
2.67 |
2.5 |
Sample – to sample precision and accuracy were evaluated using, three samples of three different concentrations, which were prepared and analyzed on same day. Day – to day variability was assessed using three concentrations analyzed on three different days, over a period of one week.
These results show the accuracy and reproducibility of the assay. Thus, it was concluded that there was no significant difference on the assay, which was tested on an intra – day and inter – day basis.
The % R.S.D. values reported in Table 5, shows that proposed methods provides acceptable intra – day and inter – day variation of Paracetamol and Etodolac.
Ruggedness of the proposed methods was determined by analysis of aliquots from homogeneous slot in different laboratories, by different analysts, using similar operational and environmental conditions; the % R.S.D. reported in Table 5 was found to be less than 2 %.
The proposed methods are accurate, simple, rapid and selective for the simultaneous estimation of Paracetamol and Etodolac in tablet dosage form by internal standardization method.
Hence, it can be conveniently adopted for the routine quality control analysis in the combination formulations. As the drug combination is available in market, hence, work is toward development of an analysis.
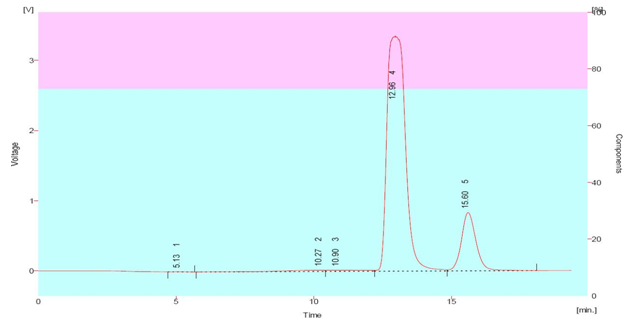
Fig 4. Chromatogram of Standard Paracetamol (50 μg.mL-1); (Rt 12.96) and Etodolac (40 μg.mL-1); (Rt 11.761) measured at 239nm, mobile phase Acetonitrile: Methanol (23:77, v/v)
Table 5. Summary of repeatability, precision and ruggedness
|
Parameter |
UV – spectrophotometry |
HPLC |
||
|
Paracetamol |
Etodolac |
Paracetamol |
Etodolac |
|
|
Repeatability |
0.89 |
0.53 |
0.67 |
0.62 |
|
Precision Intra-day Inter-day |
0.653 1.155 |
0.810 1.662 |
0.682 1.132 |
0.825 1.716 |
|
Ruggedness Analyst 1 Analyst 2 |
0.049 0.089 |
0.045 0.087 |
0.052 0.093 |
0.097 0.095 |
n is the number of repetitions.
Reference
1-en.wikipedia. org/wiki/Paracetamol/
2-Scottish Intercollegiate Guidelines Network (SIGN) (November 2008). Guideline 106: Control of pain in adults with cancer. Scotland: National Health Service (NHS). ISBN 978 1 905813 38 4
3-Moller, P.; Sindet-Pedersen, S.; Petersen, C.; Juhl, G.; Dillenschneider, A.; Skoglund, L. (2005). "Onset of acetaminophen analgesia: comparison of oral and intravenous routes after third molar surgery". British journal of anaesthesia 94 (5): 642–648. doi:10.1093/bja/aei109. PMID 1579067
4-drugbank.ca/drugs/DB00316/
5-mims.com/USA/drug/info/paracetamol/
6-jrank.org/health/pages/2881/paracetamol.html
7-pharmweb.net/pwmirror/pwy/paracetamol/pharmwebpic3.html
8-en.wikipedia.org/wiki/Etodolac.
9-medicinenet.com/etodolac/article.htm
10-drugbank.ca/drugs/DB00749
11-chemicalbook.com/ProductChemicalPropertiesCB3216064_EN.htm.
12-Beckett, A. H. and Stanlake, J. B., Practical Pharmaceutical Chemistry, (1997), 4 th Edition, part 2, CBS Publishers and Distributors, New Delhi (India), p 285.
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT articles@pharmatutor.org









