
About Author
SHIV KUMAR MANDAL*SHASHI DAKSH
Department of Pharmacy,Mewar University,Chittorgarh (312901),
Rajasthan,India
shivmandal097@gmail.com
ABSTRACT
Dengue fever is the most common arthropod-borne self-limiting viral disease with clinical spectrum ranging from asymptomatic infection to life threatening shock. It is called as dengue shock syndrome. Hemoconcentration and thrombocytopenia are the distinctive features of dengue hemorrhagic fever. Supportive fluid replacement therapy and vigilant monitoring for the successful management of the condition. Vector control measures are the most important preventive methods. As the outbreaks of Dengue fever increasing in India, one state after other getting affected, it is very essential to know more about this disease and prevalence, any change in the viral strain, severity of the disease pattern, early detection of the virus and early management of the disease resulting in good recovery . Population growth, rapid urbanization, increase in international travel from endemic areas and global warming are playing a major role in disease spread. Measures should be taken to control the aforementioned causes to prevent disease spread and reduce epidemic flare up.
INTRODUCTION
Dengue is an infectious disease caused by female mosquito AEDES AEGYPTI. It is a mosquito-borne infection found in tropical and sub-tropical regions around the world[1]. Infection with dengue virus results in various conditions like pathological ranging from mild asymptomatic dengue fever to severe dengue hemorrhagic fever and dengue shock syndrome that causes to death[2].
Epidemiology: Dengue has become one of the widest spreadable diseases globally. In India, outbreak of dengue was recorded in 1812. A recent dengue distribution model has estimated 390 million dengue infections annually, out of which 96 million cases occurred.
A double peak hemorrhagic fever epidemic occurred in India for the first time in Calcutta between July 1963 and March 1964. Thus, there is an urgent need of improvement in surveillance to enable the authorities to prepare adequately for record of outbreak[3].
Dengue Virus: This virus is also known as Flavivirus and they are in spherical and 40-60 mm in diameter. It is RNA virus that is enveloped contains three structural polypeptides two are glycosylated and replication in cytoplasm[4]. Most potent vector having epidemic potential A. aegypti and other species are Aedes albopictus, A. stegomyia, A. polnesiensis, A. scutellaris, A. finalaya but in India A. tigris is very common. Four dengue viruses (types 1-4) within the genus flavivirus and family flaviviridae, are the causative agents[5]. All four subtypes are found in India. Dengue virions are small particles with lipoprotein envelope and nucleocapsid of single stranded RNA genome with positive polarity. There is a close antigenic similarity between the four serotypes but the cross protection in humans is at best partial and transient[6].
SYMPTOMS
Many people specially children and teens feel no signs or symptoms during a mild case of dengue fever. When symptoms do occur, they usually begin 4 to 7 days after you are bitten by an infected mosquito[7].
Dengue fever causes a high fever 104 F and these are following symptoms:
• Headache
• Muscle, bone and joint pain
• Nausea
• Vomiting
• Pain behind the eyes
• Swollen glands
• Rash
Most people recover within a week. In some cases, symptoms worsen and can become life-threatening. Blood vessels often become damage and leaky and the number of clot-forming cells (platelets) in your bloodstream drops. This can cause a severe form of dengue fever, called dengue hemorrhagic fever, severe dengue or dengue shock syndrome.
Signs and symptoms of dengue hemorrhagic fever or severe dengue a life-threatening emergency include:
• Severe abdominal pain
• Persistent vomiting
• Bleeding from your gums or nose
• Blood in your urine, stools or vomit
• Bleeding under the skin, which might look like bruising
• Difficult or rapid breathing
• Cold or clammy skin (shock)
• Fatigue
• Irritability or restlessness
DIAGNOSIS
Efficient and accurate diagnosis of dengue is of primary importance for clinical care (i.e. early detection of severe cases, case confirmation and differential diagnosis with other infectious diseases), surveillance activities, outbreak control, pathogenesis, academic research, vaccine development, and clinical trials[8].
A range of laboratory diagnostic methods has been developed to support patient management and disease control. The choice of diagnostic method depends on the purpose for which the testing is done (e.g. clinical diagnosis, epidemiological survey, vaccine development), the type of laboratory facilities and technical expertise available, costs, and the time of sample collection[9].
Below figure reprsent Approximate time-line of primary and secondary dengue virus infections and the diagnostic methods that can be used to detect infection.

CONSIDERATIONS IN THE CHOICE OF DIAGNOSTIC METHODS
Clinical management
Dengue virus infection produces a broad spectrum of symptoms, many that is non-specific. Thus, a diagnosis based only on clinical symptoms is unreliable. Early laboratory confirmation of clinical diagnosis may be valuable because some patients progress over a short period from mild to severe disease and sometimes to death. Early intervention may be life-saving[10].
Differential diagnosis
Dengue fever can easily be confused with non-dengue illnesses, particularly in nonepidemic situations. Depending on the geographical origin of the patient, other etiologies including non-dengue flavivirus infections should be ruled out[11]. These include yellow fever, Japanese encephalitis, St Louis encephalitis, Zika, and West Nile, alphaviruses (such as Sinbis and chikungunya), and other causes of fever such as malaria, leptospirosis, typhoid, Rickettsial diseases (Rickettsia prowazeki, R. mooseri, R. conori, R. rickettsi, Orientia tsutsugamushi, Coxiella burneti, etc.), measles, enteroviruses, influenza and influenza-like illnesses, haemorrhagic fevers (Arenaviridae: Junin, etc.; Filoviridae: Marburg, Ebola; Bunyaviridae: hantaviruses, Crimean-Congo haemorrhagic fever, etc.).
Outbreak investigations
During outbreaks some patients may be seen presenting with fever with or without rash during the acute illness stage. some others may present with signs of plasma leakage while still others may be observed during the convalescent phase[12].
One of the priorities in a suspected outbreak is to identify the causative agent so that appropriate public health measures can be taken and physicians can be encouraged to initiate appropriate acute illness management[13]. In such cases, the rapidity and specificity of diagnostic tests is more important than test sensitivity. Samples collected from febrile patients could be tested by nucleic acid methods in a well-equipped laboratory or a broader spectrum of laboratories using an ELISA-based dengue antigen detection kit. Serological assays may be used to determine the extent of outbreaks[14].
Vaccine trials
Vaccine trials are performed in order to measure vaccine safety and efficacy in vaccinated persons. The plaque reduction and neutralization test (PRNT) and the microneutralization assays are commonly used to measure protection correlates[15].
The assay is based on the principle that neutralizing antibodies inactivate the virus so that it is no longer able to infect and replicate in target cells.
After a second dengue virus infection, high-titre neutralizing antibodies are produced against at least two, and often all four, dengue viruses as well as against non-dengue flaviviruses[16]. During the early convalescent stage following sequential dengue infections, the highest neutralizing antibody titre is often directed against the first infecting virus and not the most recent one[16].
CURRENT DENGUE DIAGNOSTIC METHODS
Virus isolation
Specimens for virus isolation should be collected early in the course of the infection, during the period of viremia (usually before day 5). Virus may be recovered from serum, plasma and peripheral blood mononuclear cells and attempts may be made from tissues collected at autopsy (e.g. liver, lung, lymph nodes, thymus, bone marrow). Because dengue virus is heat-labile, specimens awaiting transport to the laboratory should be kept in a refrigerator or packed in wet ice. For storage up to 24 hours, specimens should be kept at between +4 °C and +8 °C[17].
Nucleic acid detection
RNA is heat-labile and therefore specimens for nucleic acid detection must be handled and stored according to the procedures described for virus isolation[18].
RT-PCR
Since the 1990s, several reverse transcriptase-polymerase chain reaction (RT-PCR) assays have been developed. They offer better sensitivity compared to virus isolation with a much more rapid turnaround time. In situ RT-PCR offers the ability to detect dengue RNA in paraffin-embedded tissues[19]. All nucleic acid detection assays involve three basic steps: nucleic acid extraction and purification, amplification of the nucleic acid, and detection and characterization of the amplified product. Extraction and purification of viral RNA from the specimen can be done by traditional liquid phase separation methods (e.g. phenol, chloroform) but has been gradually replaced by silica-based commercial kits (beads or columns) that are more reproducible and faster, especially since they can be automated using robotics systems. A combination of the four serotype-specific oligonucleotide primers in a single reaction tube (one-step multiplex RT-PCR) is an interesting alternative to the nested RT-PCR[20].
Real-time RT-PCR
The real-time RT-PCR assay is a one-step assay system used to quantitate viral RNA and using primer pairs and probes that are specific to each dengue serotype. The use of a fluorescent probe enables the detection of the reaction products in real time, in a specialized PCR machine, without the need for electrophoresis. Many real-time RT-PCR assays have been developed employing TaqMan or SYBR Green technologies[21].
The TaqMan real-time PCR is highly specific due to the sequence-specific hybridization of the probe. Nevertheless, primers and probes reported in publications may not be able to detect all dengue virus strains: the sensitivity of the primers and probes depends on their homology with the targeted gene sequence of the particular virus analyzed. The SYBR green real-time RT-PCR has the advantage of simplicity in primer design and uses universal RT-PCR protocols but is theoretically less specific[22].
Real-time RT-PCR assays are either “singleplex” (i.e. detecting only one serotype at a time) or “multiplex” (i.e. able to identify all four serotypes from a single sample). The multiplex assays have the advantage that a single reaction can determine all four serotypes without the potential for introduction of contamination during manipulation of the sample[23]. However the multiplex real-time RT-PCR assays, although faster, are currently less sensitive than nested RT-PCR assays. An advantage of this method is the ability to determine viral titre in a clinical sample, which may be used to study the pathogenesis of dengue disease[24].
Isothermal amplification methods
The NASBA (nucleic acid sequence-based amplification) assay is an isothermal RNA specific amplification assay that does not require thermal cycling instrumentation[25]. The initial stage is a reverse transcription in which the single-stranded RNA target is copied into a double-stranded DNA molecule that serves as a template for RNA transcription[26]. Detection of the amplified RNA is accomplished either by electrochemiluminescence or in real-time with fluorescent-labelled molecular beacon probes. NASBA has been adapted to dengue virus detection with sensitivity near that of virus isolation in cell cultures and may be a useful method for studying dengue infections in field studies[27].
Detection of antigens
Detection of dengue antigens in acute-phase serum was rare in patients with secondary infections because such patients had pre-existing virus-IgG antibody immune complexes[28]. New developments in ELISA and dot blot assays directed to the envelop/membrane (E/M) antigen and the non-structural protein 1 (NS1) demonstrated that high concentrations of these antigens in the form of immune complexes could be detected in patients with both primary and secondary dengue infections up to nine days after the onset of illness[29].
Serological tests
MAC-ELISA
For the IgM antibody-capture enzyme-linked immunosorbent assay (MAC-ELISA) total IgM in patients’ sera is captured by anti-µ chain specific antibodies (specific to human IgM) coated onto a microplate. Dengue-specific antigens, from one to four serotypes (DEN-1, -2, -3, and -4), are bound to the captured anti-dengue IgM antibodies and are detected by monoclonal or polyclonal dengue antibodies directly or indirectly conjugated with an enzyme that will transform a non-coloured substrate into coloured products. The optical density is measured by spectrophotometer[30].
Principle of a MAC-ELISA test

IgG ELISA
The IgG ELISA is used for the detection of recent or past dengue infections. This assay uses the same antigens as the MAC-ELISA. The use of E/M-specific capture IgG ELISA (GAC) allows detection of IgG antibodies over a period of 10 months after the infection. IgG antibodies are lifelong as measured by E/M antigen-coated indirect IgG ELISA, but a fourfold or greater increase in IgG antibodies in acute and convalescent paired sera can be used to document recent infections. Test results correlate well with the haemagglutination-inhibition test[31].
This method can be used to detect IgG antibodies in serum or plasma and filter-paper stored blood samples and permits identification of a case as a primary or secondary dengue infection. In general, IgG ELISA lacks specificity within the flavivirus sero complex groups.
IgM/IgG ratio
A dengue virus E/M protein-specific IgM/IgG ratio can be used to distinguish primary from secondary dengue virus infections. IgM capture and IgG capture ELISAs are the most common assays for this purpose[32]. In some laboratories, dengue infection is defined as primary if the IgM/IgG OD ratio is greater than 1.2 (using patient’s sera at 1/100 dilution) or 1.4 (using patient’s sera at 1/20 dilutions). The infection is secondary if the ratio is less than 1.2 or 1.4.
This algorithm has also been adopted by some commercial vendors. However, ratios may vary between laboratories, thus indicating the need for better standardization of test performance.
IgA
Positive detection for serum anti-dengue IgA as measured by anti-dengue virus IgA capture ELISA (AAC-ELISA) often occurs one day after that for IgM. The IgA titre peaks around day 8 after onset of fever and decreases rapidly until it is undetectable by day 40. No differences in IgA titres were found by authors between patients with primary or secondary infections. Even though IgA values are generally lower than IgM, both in serum and saliva, the two methods could be performed together to help in interpreting dengue serology[33]. This approach is not used very often and requires additional evaluation.
Haemagglutination-inhibition test
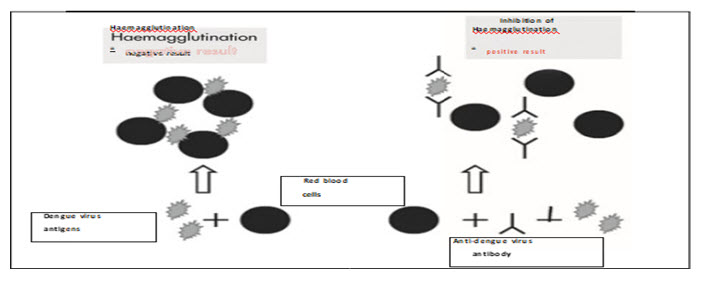
The haemagglutination-inhibition (HI) test is based on the ability of dengue antigens to agglutinate red blood cells (RBC). Anti-dengue antibodies in sera can inhibit this agglutination and the potency of this inhibition is measured in an HI test. Serum samples are treated with acetone or kaolin to remove non-specific inhibitors of haemagglutination, and then adsorbed with gander to remove non-specific agglutinins[34].
Figure : Haemagglutination-inhibition assay
Haematological tests
Platelets and haematocrit values are commonly measured during the acute stages of dengue infection. These should be performed carefully using standardized protocols, reagents and equipment.A drop of the platelet count below 1000 per µL may be observed in dengue fever but it is a constant feature of dengue haemorrhagic fever[35]. Thrombocytopaenia is usually observed in the period between day 3 and day 8 following the onset of illness.
Haemoconcentration, as estimated by an increase in haematocrit of 20% or more compared with convalescent values, is suggestive of hypovolaemia due to vascular permeability and plasma leakage[36].
TREATMENT
There is no specific therapy available for dengue virus infections.it is important to exclude other treatable diagnoses. Patients at risk for dengue can acquire other diseases with similar clinical features, such as malaria, typhoid fever, and leptospirosis[37]. Symptoms in patients with dengue virus infections resolve in five to seven days. Supportive treatments are available for the specific disease manifestations of dengue virus infection[38].
Dengue Fever: Patients with dengue fever should be cautioned to maintain their intake of oral fluid to avoid dehydration[39]. Fever and myalgias can be managed as needed with acetaminophen. Aspirin or nonsteroidal anti-inflammatory agents should generally be avoided because of the risk of bleeding complications and in children because of the potential risk of Reye's syndrome[40]. The most important measure to assist the patient with dengue fever is to carefully evaluate the patient for impending complications.
Dengue virus infection with significant Bleeding: Gastrointestinal bleeding or menorrhagia in patients with DHF, and occasionally in patients with dengue fever as well, can be severe enough to require blood transfusion[41]. Factors that contribute to bleeding include thrombocytopenia due to decreased platelet survival and, in severe cases, frank disseminated intravascular coagulation. Platelet transfusions are rarely given, but may be warranted in patients with severe thrombocytopenia (<10,000/mm3) and active bleeding[42].
Dengue Hemorrhagic Fever: Plasma leakage in DHF is important to manage with aggressive intravascular volume repletion to prevent or reverse hypovolemic shock[43]. In mild cases, particularly when medical attention is received early, oral rehydration may be sufficient[44]. However, in patients with established intravascular fluid loss, intravenous fluid administration is recommended. Blood transfusion is appropriate in patients with significant bleeding[45].
Treatment of Shock: A protocol for intravenous fluid therapy has been developed by the World Health Organization (WHO) based upon clinical experience mainly in children from Southeast Asia[46]. For patients with shock, an initial bolus of five percent dextrose in normal saline or Ringer's lactate (10 to 20 mL per kg of body weight) infused rapidly is recommended, followed by continuous infusion (10 to 20 mL/kg per hour) until vital signs and urine output normalize. The infusion rate can then be gradually reduced until it matches plasma fluid losses[47].
AS A HERBAL TREATMENT
Role of Papaya in Dengue fever
Botanical Name: Carica papaya
Family Name: Caricaceae
Common Name: Papaya, Paw Paw, Kates, Papaw
Part Used: Leaves, Fruits, bark
With the rising number of people catching dengue fever, the demand for papaya leaf juice has soared. Thrombocytopenia is one of the clinical manifestations in dengue fever and contributes to the plasma leakage and haemorrhage in the presence of enhanced vascular permeability[48]. Thrombocytopenia in dengue is considered to be an immune related, molecular mimicry involving dengue viral particles and the platelet leads to auto-destruction of the platelets by immunoglobulin M(IgM) antibodies[49]. Interestingly, C. papaya leaves juice have demonstrated a positive effect on increasing platelet count in healthy mice. C. papaya leaves extract prepared in water has been tested against dengue fever. After the administration of aqueous extract in dengue infected patient, the platelet count increased from 55x103/μL to 168x103/μL, White blood cells from 3.7x103/μL to 7.7x103/μL and neutrophils from 46% to 78%. Carica papaya leaf juice showed a significant inhibition of haemolysis invitro and could have a potential therapeutic effect on disease processes causing destabilization of biological membranes may effectively enhance the survival of platelets[50]. Thus, carica papya can be used to target dengue fever.
PREVENTION
The greatest risk for dengue virus infection is in individuals residing in endemic areas and not in travelers[51]. Public health efforts in endemic areas: Control of the Aedes aegypti mosquito, which transmits dengue virus, and the development of vaccines are two potential approaches in preventing dengue virus infections[52]. Mosquito Control: Mosquito control is the most effective approach to the prevention of dengue transmission. Programs targeting the Aedes aegypti mosquito as a means to eliminate urban yellow fever in the Americas from the 1940s through 1970s were quite successful[53].
These programs were also effective at reducing dengue transmission in the region. These programs were based on a "top down" approach involving aggressive mosquito surveillance and insecticide use. However, lack of attention and funding of these programs in the 1970s led to re-emergence of A. aegypti throughout its former region and the corresponding re-emergence of dengue[54].
Insecticide spraying, in response to dengue outbreaks, is not highly effective against A. aegypti mosquitoes, which frequently breed inside houses. Community-based approaches involving education of the population in efforts to reduce breeding sites, such as discarded tyres and other containers that accumulate standing water[55].
In one study, a comprehensive community and governmental control strategy, including the seeding of water vessels with Copepods (Fish) that feed on mosquito larvae, was successful in eliminating A. aegypti and dengue transmission in 32 communities in rural areas of Vietnam[56].
Vaccination : Infection with dengue provides long-term protection against the particular serotype that caused the disease, supporting the feasibility of a dengue vaccine. However, it provides only short-lived immunity to the other three dengue serotypes[57]. In view of the association of DHF with previous exposure to dengue viruses and the recognition that all four serotypes are capable of inducing DHF it is the general consensus in the scientific and public health communities that any candidate vaccine should produce protective immunity against DEN 1-4. Since waning immunity might also increase the risk for DHF in vaccines, vaccine-induced protective immunity should also be long-lived[58].
Animal studies indicate that protective immunity against dengue can be mediated by neutralizing antibodies, especially those directed against the envelope (E) glycoprotein. However, natural dengue infection induces low levels of cross-reactive antibodies that are detected in neutralization assays, but do not prevent infection with the other dengue serotypes[59].
Tetravalent vaccines that induce immunity against all four serotypes are in development. In a rhesus monkey model, one tetravalent live attenuated dengue virus vaccine demonstrated seroconversion rates of 100, 100, 90 and 70 percent against dengue serotypes 1, 2, 3, and 4. In addition, vaccination resulted in complete protection against viremia from inoculation with serotype 2; challenge with the other dengue serotypes demonstrated protection in 50 to 80 percent of animals compared to controls[60].
Recommendations for travelers: Most travelers from non-endemic countries are at exceedingly low risk for DHF because they lack previous exposure to dengue viruses[61]. Avoidance of exposure to infected A. aegypti mosquitoes is the primary approach to prevention of dengue virus infections in travelers. These mosquitoes predominantly live in urban areas in and around houses[62].
There are following some steps that taken as prevention of dengue virus infection :-
• Spray mosquito repellant on your skin and clothes.
• Wear long sleeves, long pants, and socks sprayed with mosquito repellant.
• Avoid being outdoors at dawn and dusk when mosquitoes are most active.
• Take time to look around your home and yard for mosquito-breeding places, especially areas where there is standing water. To avoid standing water around your home:
• Drain kiddie pools weekly.
• Change water in flower vases, bird baths, and animal watering pans twice a week.
• Get rid of old tires, buckets, bottles, and cans, or ensure they are empty of water.
• Repair any leaky pipes and outside faucets, and move air conditioner drain hoses frequently.
CONCLUSION
Though dengue fever is usually a self-limiting disease, lack of proper monitoring and adequate volume replacement may lead to fatal outcome. In view of emerging outbreaks of dengue fever in various states of India, it becomes imperative for primary care physicians to have an updated knowledge about early diagnosis and recent management guidelines.
Dengue has evolved as a global life-threatening public health concern, affecting around 2.5 billion individuals in more than 100 countries. The physician should be aware about the varied clinical manifestations of this condition and ensure an early and adequate treatment plan. Future directions to combat this dreadful disease aim at methods of mosquito control, development of vaccine, and antiviral drug regimen.
REFERENCES
1. R. Chen and N. Vasilakis, “Dengue-Quo Tu et Quo Vadis? Viruses”, Vol.3, no. 9
2. B.R. Murphy and S.S. Whitehead, “Immune response to dengue virus and prospects for a vaccine,” Annual Review of Immunology, Vol.29
3. World Health Organiazation, Dengue: Guideline for Diagnosis, Treatment, Prevention, and, Control, WHO, Geneva, 2009
4. S. Bhatt, P. W. Gething, O. J. Brady et al., “The global distribution and burden of dengue,” Nature, vol. 496, no. 7446, pp. 504–507, 2013.
5. O. J. Brady, P. W. Gething, S. Bhatt et al., “Refining the global spatial limits of dengue virus transmission by evidence-based consensus,” PLoS Neglected Tropical Diseases, vol. 6, no. 8, Article ID e1760, 2012.
6. G. N. Malavige, S. Fernando, D. J. Fernando, and S. L. Seneviratne, “Dengue viral infections,” Postgraduate Medical Journal, vol. 80, no. 948, pp. 588–601, 2004.
7. https://www.mayoclinic.org/diseases-conditions/dengue-fever/symptoms-causes/syc-20353078
8. Vorndam V, Kuno G. Laboratory diagnosis of dengue virus infections. In: Gubler DJ, Kuno G, eds. Dengue and dengue hemorrhagic fever. New York, CAB International, 1997:313–333.
9. Chanama S et al. Analysis of specific IgM responses in secondary dengue virus infections: levels and positive rates in comparison with primary infections. Journal of Clinical Virology, 2004, 31:185–189.
10. Shu PY et al. Comparison of a capture immunoglobulin M (IgM) and IgG ELISA and non-structural protein NS1 serotype-specific IgG ELISA for differentiation of primary and secondary dengue virus infections. Clinical and Diagnostic Laboratory Immunology, 2003, 10:622–630.
11. Hunsperger EA et al. Evaluation of commercially available anti–dengue virus immunoglobulin M tests. Emerging Infectious Diseases (serial online), 2009, March (date cited). Accessible at http://www.cdc.gov/EID/content/15/3/436.htm
12. Morens DM et al. Simplified plaque reduction neutralization assay for dengue viruses by semimicro methods in BHK-21 cells: comparison of the BHK suspension test with standard plaque reduction neutralization. Journal of Clinical Microbiology, 1985, 22(2):250–254.
13. Alvarez M et al. Improved dengue virus plaque formation on BHK21 and LLCMK2 cells: evaluation of some factors. Dengue Bulletin, 2005, 29:1–9.
14. Halstead SB, Rojanasuphot S, Sangkawibha N. Original antigenic sin in dengue. American Journal of Tropical Medicine and Hygiene, 1983, 32:154–156.
15. Lanciotti RS et al. Rapid detection and typing of dengue viruses from clinical samples by using reverse transcriptase-polymerase chain reaction. Journal of Clinical Microbiology, 1992, 30:545–551.
16. Harris E et al. Typing of dengue viruses in clinical specimens and mosquitoes by single tube multiplex reverse transcriptase PCR. Journal of Clinical Microbiology, 1998, 36:2634–2639.
17. Vaughn DW et al. Dengue viremia titer, antibody response pattern and virus serotype correlate with disease severity. Journal of Infectious Diseases, 2000, 181:2–9.
18. Shu PY, Huang JH. Current advances in dengue diagnosis. Clinical and Diagnostic Laboratory Immunology, 2004, 11(4):642–650.
19. Parida MM et al. Rapid detection and differentiation of dengue virus serotypes by a real-time reverse transcription-loop-mediated isothermal amplification assay. Journal of Clinical Microbiology, 2005, 43:2895–2903 (doi: 10.1128/JCM.43.6.28952903.2005).
20. Vazquez S et al. Serological markers during dengue 3 primary and secondary infections. Journal of Clinical Virology, 2005, 33(2):132–137.
21. Fernandez RJ, Vazquez S. Serological diagnosis of dengue by an ELISA inhibition method (EIM). Memórias do Instituto Oswaldo Cruz, 1990, 85(3):347–351.
22. Vazquez S, Fernandez R, Llorente C. Usefulness of blood specimens on paper strips for serologic studies with inhibition ELISA. Revista do Instituto de Medicina Tropical de São Paulo, 1991, 33(4):309–311.
23. Vazquez S et al. Kinetics of antibodies in sera, saliva, and urine samples from adult patients with primary or secondary dengue 3 virus infections. International Journal of Infectious Diseases, 2007, 11:256–262.
24. Nawa M. Immunoglobulin A antibody responses in dengue patients: a useful marker for serodiagnosis of dengue virus infection. Clinical and Vaccine Immunology, 2005, 12:1235–1237.
25. Lemmer K et al. External quality control assessments in PCR diagnostics of dengue virus infections. Journal of Clinical Virology, 2004, 30:291–296.
26. WHO. Laboratory biosafety manual, 3rd ed. Geneva, World Health Organization, 2004 (ISBN 92 4 154650 6, WHO/CDS/CSR/LYO/2004.11,
27. http://www.who.int/csr/resources/publications/biosafety/Biosafety7.pdf).
28. Ramos MM et al. Dengue Serosurvey Working Group. Epidemic dengue and dengue hemorrhagic fever at the Texas-Mexico border: results of a household-based seroepidemiologic survey, December 2005. American Journal of Tropical Medicine and Hygiene, 2008, 78(3):364--369.
29. Centers for Disease Control and Prevention. Dengue hemorrhagic fever -- U.S.Mexico border, 2005. Morbidity and Mortality Weekly Report, 2007, 56(31):785-789. Erratum in: Morbidity and Mortality Weekly Report, 2007, 56(32):822.
30. PAHO. Dengue and dengue hemorrhagic fever. Regional Committee resolution CD43.R4, adopted at the 53rd session of the Regional Committee for the Americas, 43rd Directing Council. Washington, DC, Pan American Health Organization, 2001 (http://www.paho.org/english/hcp/hct/vbd/new-generation-resolutions.pdf).
31. PAHO. Dengue. Regional Committee resolution CD44.R9, adopted at the 55th session of the Regional Committee for the Americas, 44th Directing Council. Washington, DC, Pan American Health Organization, 2003
32. (http://www.paho.org/english/gov/cd/cd44-r9-e.pdf).
33. PAHO. Grupo de Trabajo sobre Dengue. Estrategia de Gestión Integrada para la prevención y control del dengue en la Región de las Américas. 2.ª versión. Santa Cruz de la Sierra, Bolivia. Pan American Health Organization, 2003 (Document OPS/ HDM/CD/440.07, in Spanish).
34. PAHO. Pan American Health Organization. Prevención y control del dengue en las Américas. Resolución CSP27.R15. 27. a Conferencia Sanitaria Panamericana CSP27. R15 (Esp.) 1--5 de octubre de 2007 (in Spanish).
35. Nathan MB, Dayal-Drager R. Recent epidemiological trends, the global strategy and public health advances in dengue. Working paper 3.1 in: Report of the Scientific Working Group meeting on Dengue, Geneva, 1–5 October 2006. Geneva, World Health Organization, Special Programme for Research and Training in Tropical Diseases, 2007 (pp 29--34) (Document TDR/SWG/07).
36. Kokernot RH, Smithburn KC, Weinbren MP. Neutralising antibodies to arthropodborne viruses in human and animals in the Union of South Africa. Journal of Immunology, 1956, 77:313–322.
37. Blackburn NK, Rawal R. Dengue fever imported from India: a report of 3 cases. South African Medical Journal, 1987, 21:386–287.
38. Boisier P et al. Dengue 1 epidemic in the Grand Comoro Island (Federal Islamic Republic of the Comores), March-May 1993. Annales de la Société Belge de Médecine Tropicale, 1993, 74:217–229.
39. Gubler DJ et al. Dengue 3 Virus Transmission in Africa. American Journal of Tropical Medicine and Hygiene, 1986, 35(6):1280--1284.
40. Carey DE et al. Dengue virus from febrile patients in Nigeria 1964–68. Lancet, 1971, 1:105–106.
41. Gonzalez JP et al. Dengue in Burkina Faso: seasonal epidemics in the urban area of Ouagadougu. Bulletin de la Société de pathologie exotique et de ses filiales, 1985, 78:7–14.
42. Diallo M et al. Amplification of the sylvatic cycle of dengue virus type 2, Senegal, 1999--2000: entomologic findings and epidemiologic considerations. Emerging Infectious Diseases (serial online), 2003, March (date cited). Accessible at http:// www.cdc.gov/ncidod/EID/vol9no3/02-0219.htm.
43. WHO/EMRO. World Health Organization, Regional Office for the Eastern Mediterranean, Division of Communicable Disease Control, Newsletter, I2005, 6:7–8. (http://www.emro.who.int/pdf/dcdnewsletter6.pdf).
44. Hyams KC et al. Evaluation of febrile patients in Port Sudan, Sudan: isolation of dengue virus. American Journal of Tropical Medicine and Hygiene, 1986, 35:860– 865.
45. Rodier GR et al. Epidemic dengue 2 in the city of Djibouti 1991–1992. Transactions of the Royal Society of Tropical Medicine and Hygiene, 1996, 90:237–240.
46. Bushra J. Dengue Virus Serotype 3, Karachi, Pakistan. Emerging Infectious Diseases, 2007, 13(1).
47. European Centre for Disease Prevention and Control. Dengue worldwide: an overview of the current situation and implications for Europe, Eurosurveillance, 2007, 12(25). (http://www.eurosurveillance.org/ViewArticle.aspx?PublicationType=W&Vol ume=12&Issue=25&OrderNumber=1).
48. WHO. Dengue haemorrhagic fever: diagnosis, treatment, prevention and control, 2nd ed. Geneva, World Health Organization, 1997.
49. Guha-Sapir D, Schimmer B. Dengue fever: new paradigms for a changing epidemiology. Emerging Themes in Epidemiology, 2005 (Open access journal, http:// www.ete-online.com/content/2/1/1).
50. L. Hermida, L. J. Bernardo, M. Mart´?n et al., “A recombinant fusion protein containing the domain III of the dengue-2 envelope protein is immunogenic and protective in nonhuman primates,” Vaccine, vol. 24, no. 16, pp. 3165–3171, 2006.
51. J. R. Danko, C. G. Beckett, and K. R. Porter, “Development of dengue DNA vaccines,” Vaccine, vol. 29, no. 42, pp. 7261–7266, 2011
52. C. G. Beckett, J. Tjaden, T. Burgess et al., “Evaluation of a prototype dengue-1 DNA vaccine in a Phase 1 clinical trial,” Vaccine, vol. 29, no. 5, pp. 960–968, 2011.
53. K. R. Porter, D. Ewing, L. Chen et al., “Immunogenicity and protective efficacy of a vaxfectin-adjuvanted tetravalent dengue DNA vaccine,” Vaccine, vol. 30, no. 2, pp. 336–341, 2012.
54. K. Raviprakash, T. Luke, J. Doukas et al., “A dengue DNA vaccine formulated with Vaxfectin® is well tolerated, and elicits strong neutralizing antibody responses to all four dengue serotypes in New Zealand white rabbits,” Human Vaccines and Immunotherapeutics, vol. 8, no. 12, pp. 1764–1768, 2012.
55. US Army Medical Research and Materiel Command, Evaluation of the Safety and the Ability of a DNA Vaccine to Protect Against Dengue Disease (TVDV), ClinicalTrials.gov, Bethesda, Md, USA, National Library of Medicine (US), NLM Identifier: NCT01502358, 2000, https://clinicaltrials.gov/ct2/show/ NCT01502358.
56. K. Raviprakash, D. Wang, D. Ewing et al., “A tetravalent dengue vaccine based on a complex adenovirus vector provides significant protection in rhesus monkeys against all four serotypes of dengue virus,” Journal of Virology, vol. 82, no. 14, pp. 6927–6934, 2008.
57. L. J. White, M. M. Parsons, A. C. Whitmore, B. M. Williams, A. de Silva, and R. E. Johnston, “An immunogenic and protective alphavirus replicon particle-based dengue vaccine overcomes maternal antibody interference in weanling mice,” Journal of Virology, vol. 81, no. 19, pp. 10329–10339, 2007.
58. S. Brandler, M. Lucas-Hourani, A. Moris et al., “Pediatric measles vaccine expressing a dengue antigen induces durable serotype-specific neutralizing antibodies to dengue virus,” PLoS Neglected Tropical Diseases, vol. 1, no. 3, article e96, 2007.
59. D. H. Holman, D. Wang, K. Raviprakash et al., “Two complex, adenovirus-based vaccines that together induce immune responses to all four dengue virus serotypes,” Clinical and Vaccine Immunology, vol. 14, no. 2, pp. 182–189, 2007.
60. N. U. Raja, D. H. Holman, D. Wang et al., “Induction of bivalent immuneresponsesbyexpressionofdenguevirustype1andtype 2 antigens from a single complex adenoviral vector,” American Journal of Tropical Medicine and Hygiene, vol. 76, no. 4, pp. 743– 751, 2007.
61. WHO. Dengue hemorrhagic fever: diagnosis, treatment, prevention, and control. 2nd ed. Geneva: World Health Organization, 2002.
62. Nimmannitya, S, Thisyakorn, U, Hemsrichart, V. Dengue haemorrhagic fever with unusual manifestations. Southeast Asian J Trop Med Public Health 1987; 18: 398.
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT admin@pharmatutor.org
FIND OUT MORE ARTICLES AT OUR DATABASE










