About Authors:
Keyur B.ahir, Emanual M. Patelia*, Falgun A.Mehta
Department of Pharmaceutical Chemistry and Analysis,
Indukaka Ipcowala College of Pharmacy,
New Vallabh Vidyanagar – 388121, Gujarat, India
*ricky.emanual@gmail.com
Abstract:
A simple, precise, rapid, selective, and economic high-performance-thin-layer chromatography (HPTLC) method has been established for simultaneous analysis of Domperidone (DMP), Paracetamol (PCM) and Tramadol Hcl (TMD) in tablet dosage forms. The chromatographic separations were performed on precoated silica gel 60254 plates with toluene-ethylacetate-butanol-ammonia 5:4:1:0.2(v/v) as mobile phase. The plates were developed in a 7.0 cm at ambient temperature. The developed plates were scanned and quantified at their single wavelength of maximum absorption at approximately 278 nm for DMP and PCM, respectively. Experimental conditions such as chamber size, chamber saturation time, migration of solvent front, slit width, etc. were critically studied and the optimum conditions were selected. The drugs were satisfactorily resolved with Rf 0.18 ± 0.02 for DMP, Rf 0.25 ± 0.02 for PCM and for TMD Rf 0.50 ± 0.02. The method was validated for linearity, accuracy, precision, and specificity. The calibration plot was linear between 100-600 ng / band for DMP, 3250-19500 ng / band based for PCM and 375-2250 ng / band based for TMD. The limits of detection and quantification for DMP were 9.95 and 30.16ng / band, respectively; for PCM they were 64.30 ng and 194.87 ng / band and for TMD 5.51 and 16.70/ band. This HPTLC procedure is economic, sensitive, and less time consuming than other chromatographic procedures. It is a user-friendly and importance tool for analysis of combined tablet dosage forms.
Reference Id: PHARMATUTOR-ART-1367
Introduction
PCM (PCM; N-[4-hydroxyphenyl] ethanamide; Figure 1b) is a widely used analgesic and antipyretic for the relief of fever, headaches, and other minor aches and pains, and is a major ingredient in numerous cold and flu remedies. In combination with nonsteroidal anti-inflammatory drugs (NSAIDs) and opioid analgesics, paracetamol is used also in the management of severe pain (such as postoperative pain) [1]. Tramadol Hcl (TMD; (+/-) cis-2-[(Dimethylamino) methyl]-1-(3-methoxyphenyl) cyclohexanol hydrochloride Figure 1c) is a centrally acting analgesic, having agonist actions at the μ-opioid receptor and affects reuptake at the noradrenergic and serotonergic systems. TMD is a compound with mild and delayed μ-agonist activity [2]. Domperidone (DMP; 5-chloro-1-[1-[3-(2-oxo-2, 3-dihydro-1H-benzimidazol-1- yl) propyl]-piperidin-4-yl]-1, 3-dihydro-2H-benzimidazol-2-one; Figure 1a) used as antiemetic drug.
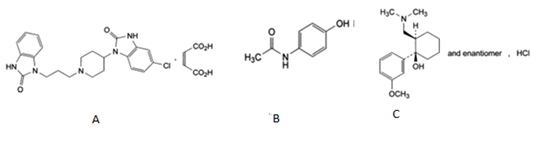
Figure 1 Structure of domperidone (a)paracetamol (b) and (c) tramadol HCl
PCM is official in Indian Pharmacopoeia. This pharmacopoeia suggests titrimetric and UV spectrophotometric assay method for PCM in bulk and tablet formulations. DMP is official in Indian Pharmacopoeia where assay is described by titrimetric method. Tramadol is official in Indian Pharmacopoeia. This pharmacopoeia suggests titrimetric (potentiometric) assay method for tramadol in bulk. Literature survey revealed that various analytical methods like spectrophotometric [3-6], HPLC [7-14], GC [15] and HPTLC [16-19] have been reported for the determination of TMD, PCM and either individually or combination with some other drugs, but no HPTLC method was reported for simultaneous estimation of TMD and PCM and domperidol in combined dosage forms. Many methods [20], [21], [22], [23], [24], [25], [26], [27] have been described in the literature for the determination of domperidol and pracetamol, individually. The analytical methods like HPLC [28] and HPTLC [29] for determination of domperidol and PCM in combined dosage form has been reported. The RP HPLC [30] method has been reported for estimation of TMD, PCM and Domperidol in tablet formulation. The review of literature prompted us to develop an accurate, selective and precise simultaneous method for the estimation of TMD, PCM and Domperidol in combined dosage forms.
2 Experimental
2.1 Chemicals and Materials
TMD, PCM and DMP were procured from Cadila pharmaceuticals, Ahmedabad. Ethyl Acetate, Toluene, Ammonia and n-Butanol were used as solvents to prepare the mobile phase. All the reagents used were of Analytical reagent grade (CHEMDYES CORPORATION, Ahmedabad, india) and used without further purification. Tablet formulation A (Tramazac - PD, Zydus Cadila Healthcare Ltd., Ahmedabad,India) and Tablet formulation B (RAMCET-D sundyota numandis pharma. Ahmedabad., India) containing labeled amount of 325 mg PCM, 37.5 mg TMD, and 10 mg DMP were procured from local market.
2.2 Chromatographic Conditions
The samples were spotted in the form of bands of width 6mm with a camag 100 µL sample (Hamilton, Bonaduz, Switzerland) syringe on precoated silica gel 60254 plates (10×10) with 250 µm thickness; (E MERCK, Darmstadt, Germany), supplied by using a camag Linomat V( Switzerland). The plates were prewashed by methanol and activated at 1100C for 5 minutes, prior to chromatography. A constant application rate of 0.1µL /spot was employed and space between two bands was 5 mm. The slit dimension was kept at 6 mm × 0.45 mm and 10 mm/spot scanning speed was employed. The monochromator band width was set at 20 nm and 320 cut off filter; each track was scan thrice and base line correction was used. The mobile phase consists of n-Butanol – ethylacetate - toluene – ammonia (1:4:5:0.1 v/v/v/v). Linear ascending development was carried out in 10 cm × 10 cm twin trough glass chamber (Camag, Muttenz, Switzerland). The optimized chamber saturation time for mobile phase was 20 min; at room temp. (250C± 2); at relative humidity 60% ± 5. The length of chromatogram run was 7 cm and approximately 40 min., subsequent to the development. TLC plates were dried in a current of air with the help of an air drier. Densitometric scanning was performed on Camag TLC scanner III in the reflectance-absorbance mode at 278 nm for all measurements and operated by WINCATS software version 1.3.0. The source of radiation utilized was deuterium lamp, continues emits UV spectrum between 200 nm to 400 nm.
2.3 Sample Preparation
Twenty tablets of brand A were weighed and average weight is calculated. Powder equivalent to approximately 325 mg PCM, 10 mg DMP and 37.5 mg TMD was weighed accurately, transferred to a 100-mL volumetric flask, and methanol (100 mL) was added. The solution was sonicated for 15 min, then diluted to volume with methanol and filtered through a Whatman filter paper No.41.
NOW YOU CAN ALSO PUBLISH YOUR ARTICLE ONLINE.
SUBMIT YOUR ARTICLE/PROJECT AT articles@pharmatutor.org
Subscribe to PharmaTutor Alerts by Email
FIND OUT MORE ARTICLES AT OUR DATABASE
Preparation of standard solution
A Combined standard stock solution containing 100µg/mLDMP, 3250 µg/m PCM and 375 µg/ml TMD was prepared in methanol. Calibration was done by applying mix standard solution ranging from 1-6 µl by micro liter syringe with the help of automatic sample applicator Linomat V on TLC Plate, which gives concentration of 100-600 ng/spot of DMP , 3250-19500 ng/spot of PCM and 375-2250 ng/spot of TMD. Each concentration was spotted six times on the TLC plates. The plate was developed on previously described mobile phase. The peak areas plotted against the corresponding concentrations to obtain the calibration graphs.
2.4 Method Validation
The developed method was validated for linearity and range, specificity, accuracy, precision, Limit of detection, Limit of quantitation, robustness and solution stability as per ICH guidelines.
2.4.1 Linearity and Range
Aliquots of standards 1-6µl TMD, PCM & DMP solutions were loaded in the 10 x 10 Silica gel 60F254 TLC plate using 100μl Hamilton syringe and Camag – Linomat -5 instrument.
2.4.2 Specificity
The specificity of the method was ascertained by analyzing PCM, DMP and TMD in presence of excipients commonly used for tablet formulations. The bands of PCM, DMP and TMD were confirmed by comparing RF values and respective spectra of sample with those of standards. The peak purity of PCM, DMP and TMD was assured by comparing the spectra at three different levels, that is, peak start, peak apex and peak end positions, as listed in Table 1.
2.4.3 Accuracy (% Recovery)
The recovery study was car











.png)

